Abstract
Objectives
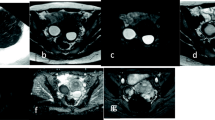
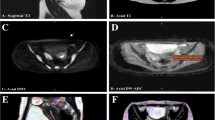
The endometrium undergoes morphological and functional changes during menstrual cycles, regulated by female hormones. MRI T2 weighted imaging (T2WI) plays an essential role in endometrial disease evaluation. This study aims to define the cyclic course of endometrial T2 values and thickness and explore their relationship with basal female sex hormones.
Methods
We prospectively recruited 38 healthy volunteers (20–30 years:22; 30–40 years:16), 3.0 T MR were performed during the menstrual phase (MP), follicular phase (FP), peri-ovulation phase (OP), and luteal phase (LP). Basal serum oestradiol (E), progesterone (P), luteinizing hormone (LH) and follicle stimulating hormone (FSH) hormone was tested. Endometrium thicknesses, T2 values were obtained in each phase. Pearson correlation test, Fisher's least significant difference (LSD) test, the analysis of variance (ANOVA) test, and the paired sample t test were applied.
Results
The endometrium thickness was significantly thinner in the younger age group (p < 0.001). The thickness was significantly thinner in first half compared with that in the second half in both groups (p < 0.001). Endometrial T2 values were significantly shorter in menstrual cycles compared with the other three phases (p ≤ 0.001). Both endometrial T2 value and thickness were correlated to female hormones. Basal FSH and P were correlated with T2 values in the menstrual phase in the 30–40 year group. Serum FSH was correlated with endometrial thickness.
Conclusions
T2 mapping and endometrium thickness distinguish in different cyclic phases, provided background information for evaluation pathologic conditions. Moreover, with their correlations with female basal sex hormones, we found the T2 mapping sequence can provide a potential biomarker for quantifying the endometrium's physiological status.
Advances in knowledge
The current study is the first to investigate the relationship between T2 values and basal sex hormones. This new technique may find a role in determining endometrium functions such as receptivity and might have potential clinical significance in the diagnosis of endometrium-related gynecology diseases.



Similar content being viewed by others
References
Hagen CP, Mouritsen A, Mieritz MG, Tinggaard J, Wohlfahrt-Veje C, Fallentin E, et al. Uterine volume and endometrial thickness in healthy girls evaluated by ultrasound (3-dimensional) and magnetic resonance imaging. Fertil Steril. 2015;104(2):452-9.e2.
Pierson RA. Imaging the endometrium: are there predictors of uterine receptivity? J Obstet Gynaecol Can. 2003;25(5):360–8.
Bazot M, Darai E. Diagnosis of deep endometriosis: clinical examination, ultrasonography, magnetic resonance imaging, and other techniques. Fertil Steril. 2017;108(6):886–94.
Nougaret S, Reinhold C, Alsharif SS, Addley H, Arceneau J, Molinari N, et al. Endometrial cancer: combined MR volumetry and diffusion-weighted imaging for assessment of myometrial and lymphovascular invasion and tumor grade. Radiology. 2015;276(3):797–808.
Sahdev A. Imaging the endometrium in postmenopausal bleeding. BMJ. 2007;334(7594):635–6.
Zondervan KT, Becker CM, Koga K, Missmer SA, Taylor RN, Vigano P. Endometriosis. Nat Rev Dis Primers. 2018;4(1):9.
Fernandez-Jimenez R, Barreiro-Perez M, Martin-Garcia A, Sanchez-Gonzalez J, Aguero J, Galan-Arriola C, et al. Dynamic edematous response of the human heart to myocardial infarction: implications for assessing myocardial area at risk and salvage. Circulation. 2017;136(14):1288–300.
Lee KH, Park HS, Park CH, Kim KH, Chung H, Kim TH, et al. Extracellular volume imaging and quantitative T2 mapping for the diagnosis of mitochondrial cardiomyopathy. Circulation. 2014;130(20):1832–4.
Bonner F, Spieker M, Haberkorn S, Jacoby C, Flogel U, Schnackenburg B, et al. Myocardial T2 mapping increases noninvasive diagnostic accuracy for biopsy-proven Myocarditis. JACC Cardiovasc Imaging. 2016;9(12):1467–9.
Crouser ED, Ono C, Tran T, He X, Raman SV. Improved detection of cardiac sarcoidosis using magnetic resonance with myocardial T2 mapping. Am J Respir Crit Care Med. 2014;189(1):109–12.
Puntmann VO, Nagel E. T1 and T2 mapping in nonischemic cardiomyopathies and agreement with endomyocardial biopsy. J Am Coll Cardiol. 2016;68(17):1923–4.
Shitano F, Kido A, Kataoka M, Fujimoto K, Kiguchi K, Fushimi Y, et al. MR appearance of normal uterine endometrium considering menstrual cycle: differentiation with benign and malignant endometrial lesions. Acta Radiol. 2016;57(12):1540–8.
He YL, Ding N, Qi YF, Li Y, Xiang Y, Qian TY, et al. Visualising the boundary sharpness of uterine zonal structures using high-resolution T2-weighted images during the menstrual cycle. Clin Radiol. 2019;74(1):81.e19-81.e24.
Sarvi F, Arabahmadi M, Alleyassin A, Aghahosseini M, Ghasemi M. Effect of increased endometrial thickness and implantation rate by granulocyte colony-stimulating factor on unresponsive thin endometrium in fresh in vitro fertilization cycles: a randomized clinical trial. Obstet Gynecol Int. 2017;2017:3596079.
Maekawa R, Taketani T, Mihara Y, Sato S, Okada M, Tamura I, et al. Thin endometrium transcriptome analysis reveals a potential mechanism of implantation failure. Reprod Med Biol. 2017;16(2):206–27.
He Y, Ding N, Li Y, Li Z, Xiang Y, Jin Z, et al. 3-T diffusion tensor imaging (DTI) of normal uterus in young and middle-aged females during the menstrual cycle: evaluation of the cyclic changes of fractional anisotropy (FA) and apparent diffusion coefficient (ADC) values. Br J Radiol. 2015;88(1049):20150043.
Hoad CL, Raine-Fenning NJ, Fulford J, Campbell BK, Johnson IR, Gowland PA. Uterine tissue development in healthy women during the normal menstrual cycle and investigations with magnetic resonance imaging. Am J Obstet Gynecol. 2005;192(2):648–54.
Kido A, Kataoka M, Koyama T, Yamamoto A, Saga T, Togashi K. Changes in apparent diffusion coefficients in the normal uterus during different phases of the menstrual cycle. Br J Radiol. 2010;83(990):524–8.
Hauth EA, Jaeger HJ, Libera H, Lange S, Forsting M. MR imaging of the uterus and cervix in healthy women: determination of normal values. Eur Radiol. 2007;17(3):734–42.
Haynor DR, Mack LA, Soules MR, Shuman WP, Montana MA, Moss AA. Changing appearance of the normal uterus during the menstrual cycle: MR studies. Radiology. 1986;161(2):459–62.
de Bazelaire CM, Duhamel GD, Rofsky NM, Alsop DC. MR imaging relaxation times of abdominal and pelvic tissues measured in vivo at 3.0 T: preliminary results. Radiology. 2004;230(3):652–9.
Ghosh A, Singh T, Bagga R, Srinivasan R, Singla V, Khandelwal N. T2 relaxometry mapping in demonstrating layered uterine architecture: parameter optimization and utility in endometrial carcinoma and adenomyosis: a feasibility study. Br J Radiol. 2018;91(1081):20170377.
He YL, Ding N, Li Y, Li Z, Xiang Y, Jin ZY, et al. Cyclic changes of the junctional zone on 3 T MRI images in young and middle-aged females during the menstrual cycle. Clin Radiol. 2016;71(4):341–8.
Nakai A, Togashi K, Yamaoka T, Fujiwara T, Ueda H, Koyama T, et al. Uterine peristalsis shown on cine MR imaging using ultrafast sequence. J Magn Reson Imaging. 2003;18(6):726–33.
Hall JE, Schoenfeld DA, Martin KA, Crowley WF Jr. Hypothalamic gonadotropin-releasing hormone secretion and follicle-stimulating hormone dynamics during the luteal-follicular transition. J Clin Endocrinol Metab. 1992;74(3):600–7.
Acknowledgements
We thank Hui Liu for MRI parameters setting of this study; Hao Zhang for serum female hormone measurements; and Wei Han for the design of local SAS evaluation programs.
Funding
No competing financial interests exist. Funded by Basic Scientific Research Program of Chinese Academy of Medical Sciences (2019PT320008 and 2018PT32003).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have nothing to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ding, N., He, Y., Qi, Y. et al. Endometrial T2 values and thickness measured during the spontaneous menstrual cycle: potential imaging biomarker related to female physiological hormones. Chin J Acad Radiol 4, 98–104 (2021). https://doi.org/10.1007/s42058-021-00063-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42058-021-00063-5