Abstract
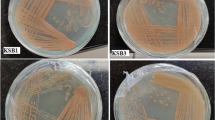
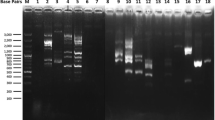
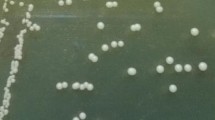
Beta-glucan is a natural polysaccharide, abundantly present in the cell walls of several fungi, majorly in mushrooms, yeasts and many bacterial species. It can be used in the cosmetics industry as it has good anti-aging, wound healing and antioxidant properties. Marine yeasts were isolated from the backwater samples derived from Andaman-Nicobar Islands, India and screened for beta-glucan production. Among 38 yeast isolates, 5 isolates could produce beta-glucan and they were further chosen for UV mutagenesis at different time intervals for strain improvement. Mutated marine yeast isolate DAMB1 was found to be producing the highest amount of beta-glucan (1.67 gm/100 ml). Further, potential isolates were characterized microscopically and checked for their antioxidant activity and anti-bacterial activity against bacterial pathogens. The beta-glucan compounds exhibited good antioxidant activity as well as good antimicrobial activity against all the pathogens and highest against Staphylococcus aureus (23 ± 0.7 mm). The yeast-derived compound was structurally characterized using GCMS analysis and represented the presence of Ethylthiophosphonamide Acid, O-methyl Ester, and 1,2,4-Trithiolane which are highly pure fatty acid methyl esters. The extracted beta-glucan compound from potential yeast isolate was used to formulate beta-glucan almond scrub consisting of sweet almond oil and almond powder. The beta-glucan almond scrub showed no side effects or infection on the skin after a period of usage (10 weeks) rather it smoothened out the rough skin to an extent. Finally, the yeast was further characterized and identified as Rhodotorula sp. DAMB1 (Acc. No. MK968443) through 18s rRNA sequencing analysis.







Similar content being viewed by others
Data Availability
All the necessary data is available in the manuscript.
References
Adjimani JP, Asare P (2015) Antioxidant and free radical scavenging activity of iron chelators. Toxicol Rep 2:721–728. https://doi.org/10.1016/j.toxrep.2015.04.005
Akramienė D, Kondrotas A, Didžiapetrienė J, Kėvelaitis E (2007) Effects of ß-glucans on the immune system. Medicina 43(8):597. https://doi.org/10.3390/medicina43080076
Alam MN, Bristi NJ, Rafiquzzaman M (2013) Review on in vivo and in vitro methods evaluation of antioxidant activity. Saudi Pharm J 21(2):143–152. https://doi.org/10.1016/j.jsps.2012.05.002
Anusha SU, Sundar SK, Williams PG (2014) Studies on the isolation and characterisation of marine yeast, β-glucan production and immunostimulatory activity on Carassius auratus. Int J Curr Microbiol Appl Sci 3(9):230–240
Banerjee S, Pandey K, Mazumdar S, Sarkar A, Bhaskara Rao KV (2019) Characterization and applications of protease inhibitor compound from marine yeast Candida orthopsilosis KSSB3. Res J Biotechnol 14(9):43–52
Barsanti L, Passarelli V, Evangelista V, Frassanito AM, Gualtieri P (2011) Chemistry, physico-chemistry and applications linked to biological activities of β-glucans. Nat Prod Rep 28(3):457–466. https://doi.org/10.1039/C0NP00018C
Bauer AW (1966) Antibiotic susceptibility testing by a standardised single disc method. Am J Clin Pathol 45:149–158
Berdal M, Appelbom HI, Eikrem JH, Lund Å, Zykova S, Busund LT, Seljellid R, Jenssen T (2007) Aminated β-1, 3‐d‐glucan improves wound healing in diabetic db/db mice. Wound Repair Regen 15(6):825–832. https://doi.org/10.1111/j.1524-475X.2007.00286.x
Bohn JA, BeMiller JN (1995) (1→ 3)-β-d-Glucans as biological response modifiers: a review of structure-functional activity relationships. Carbohydr Polym 28(1):3–14. https://doi.org/10.1016/0144-8617(95)00076-3
Brusick DJ (1970) The mutagenic activity of ICR-170 in Saccharomyces cerevisiae. Mutat Research/Fundamental Mol Mech Mutagen 10(1):11–19. https://doi.org/10.1016/0027-5107(70)90141-7
Byrtusová D, Shapaval V, Holub J, Šimanský S, Rapta M, Szotkowski M, Kohler A, Márová I (2020) Revealing the potential of lipid and β-glucans coproduction in Basidiomycetes yeast. Microorganisms 8(7):1034. https://doi.org/10.3390/microorganisms8071034
Chamidah A, Hardoko H, Prihanto AA (2017) Antibacterial activities of β-glucan (laminaran) against gram-negative and gram-positive bacteria. In AIP Conference Proceedings 1844(1) AIP Publishing. https://doi.org/10.1063/1.4983422
Chan GCF, Chan WK, Sze DMY (2009) The effects of β-glucan on human immune and cancer cells. J Hematol Oncol 2(1):1–11. https://doi.org/10.1186/1756-8722-2-25
Choromanska A, Kulbacka J, Rembialkowska N, Pilat J, Oledzki R, Harasym J, Saczko J (2015) Anticancer properties of low molecular weight oat beta-glucan–An in vitro study. Int J Biol Macromol 80:23–28. https://doi.org/10.1016/j.ijbiomac.2015.05.035
Cong L, Chi Z, Li J, Wang X (2007) Enhanced carotenoid production by a mutant of the marine yeast Rhodotorula sp. hidai. J Ocean Univ China 6(1):66–71. https://doi.org/10.1007/s11802-007-0066-x
Delatte SJ, Evans J, Hebra A, Adamson W, Othersen HB, Tagge EP (2001) Effectiveness of beta-glucan collagen for treatment of partial-thickness burns in children. J Pediatr Surg 36(1):113–118. https://doi.org/10.1053/jpsu.2001.20024
Ding J, Wang Y, Xiong S, Zhao S, Huang Q (2013) Optimised methodology for carboxymethylation of (1→3)-β‐d‐glucan from yeast (Saccharomyces cerevisiae) and promotion of mechanical activation. Int J Food Sci Technol 48(2):253–259. https://doi.org/10.1111/j.1365-2621.2012.03181.x
Dinis TC, Madeira VM, Almeida LM (1994) Action of phenolic derivatives (acetaminophen, salicylate, and 5-aminosalicylate) as inhibitors of membrane lipid peroxidation and as peroxyl radical scavengers. Arch Biochem Biophys 315(1):161–169. https://doi.org/10.1006/abbi.1994.1485
Du B, Bian Z, Xu B (2014) Skin health promotion effects of natural beta-glucan derived from cereals and microorganisms: A review. Phytother Res 28(2):159–166. https://doi.org/10.1002/ptr.4963
Fang J, Wang Y, Lv X, Shen X, Ni X, Ding K (2012) Structure of a β-glucan from Grifola frondosa and its antitumor effect by activating Dectin-1/Syk/NF-κB signaling. Glycoconj J 29(5):365–377. https://doi.org/10.1007/s10719-012-9416-z
Fell JW (2001) Collection and identification of marine yeasts. In: Paul JP (ed) Methods in Microbiology. Oxford University Press, New York, pp 347–356. https://doi.org/10.1016/S0580-9517(01)30052-1
Gientka I, Bzducha-Wróbel A, Stasiak-Różańska L, Bednarska AA, Błażejak S (2016) The exopolysaccharides biosynthesis by Candida yeast depends on carbon sources. Electron J Biotechnol 22:31–37. https://doi.org/10.1016/j.ejbt.2016.02.008
Haefner K, Howrey L (1967) Gene-controlled UV-sensitivity in Schizosaccharomyces Pombe. Mutat Res/Fund Mol Mech Mutagen 4(2):219–221. https://doi.org/10.1016/0027-5107(67)90075-9
Hamidi M, Okoro OV, Ianiri G, Jafari H, Rashidi K, Ghasemi S, Castoria R, Palmieri D, Delattre C, Pierre G, Mirzaei M (2023) Exopolysaccharide from the yeast papiliotrema terrestris PT22AV for skin wound healing. J Adv Res 46:61–74. https://doi.org/10.1016/j.jare.2022.06.012
Horn P, Wilkie D (1966) Use of Magdala red for the detection of auxotrophic mutants of Saccharomyces cerevisiae. J Bacteriol 91(3):1388
Hussain PR, Rather SA, Suradkar PP (2018) Structural characterization and evaluation of antioxidant, anticancer and hypoglycemic activity of radiation degraded oat (Avena sativa) β-glucan. Radiat Phys Chem 144:218–230. https://doi.org/10.1016/j.radphyschem.2017.08.018
Jiao G, Yu G, Zhang J, Ewart HS (2011) Chemical structures and bioactivities of sulfated polysaccharides from marine algae. Mar Drugs 9(2):196–223. https://doi.org/10.3390/md9020196
Kagimura FY, da Cunha MAA, Theis TV, Malfatti CR, Dekker RF, Barbosa AM, Teixeira SD, Salomé K (2015) Carboxymethylation of (1→ 6)-β-glucan (lasiodiplodan): Preparation, characterization and antioxidant evaluation. Carbohydr Polym 127:390–399. https://doi.org/10.1016/j.carbpol.2015.03.045
Kanlayavattanakul M, Lourith N (2009) Carboxymethylglucan in cosmetics. Thai Pharmaceut Health Sci J 3(3)
Karthik L, Kumar G, Rao KVB (2013) Antioxidant activity of newly discovered lineage of marine actinobacteria. Asian Pac J Trop Med 6(4):325–332. https://doi.org/10.1016/S1995-7645(13)60065-6
Kumar SRS, Rao KVB (2016) Postprandial anti-hyperglycemic activity of marine Streptomyces coelicoflavus SRBVIT13 mediated gold nanoparticles in streptozotocin induced diabetic male albino Wister rats. IET Nanobiotechnol 10(5):308–314. https://doi.org/10.1049/iet-nbt.2015.0094
Lavova B, Urminska D (2013) Total antioxidant activity of yeast Saccharomyces cerevisiae. J Microbiol Biotechnol Food Sci 2(1):1927–1933
Liang J, Melican D, Cafro L, Palace G, Fisette L, Armstrong R, Patchen ML (1998) Enhanced clearance of a multiple antibiotic resistant Staphylococcus aureus in rats treated with PGG-glucan is associated with increased leukocyte counts and increased neutrophil oxidative burst activity. Int J Immunopharmacol 20(11):595–614. https://doi.org/10.1016/S0192-0561(98)00007-1
Magnani M, Calliari CM, de Macedo FC Jr, Mori MP, de Syllos Cólus IM, Castro-Gomez RJ (2009) Optimized methodology for extraction of (1→ 3)(1→ 6)-β-D-glucan from Saccharomyces cerevisiae and in vitro evaluation of the cytotoxicity and genotoxicity of the corresponding carboxymethyl derivative. Carbohydr Polym 78(4):658–665. https://doi.org/10.1016/j.carbpol.2009.05.023
Mata-Gómez LC, Montañez JC, Méndez-Zavala A, Aguilar CN (2014) Biotechnological production of carotenoids by yeasts: an overview. Microb Cell Factories 13(1):1–11. https://doi.org/10.1186/1475-2859-13-12
Medina-Córdova N, Reyes-Becerril M, Ascencio F, Castellanos T, Campa-Córdova AI, Angulo C (2018) Immunostimulant effects and potential application of β-glucans derived from marine yeast Debaryomyces hansenii in goat peripheral blood leucocytes. Int J Biol Macromol 116:599–606. https://doi.org/10.1016/j.ijbiomac.2018.05.061
Munn CB (2004) Marine Microbiology: Ecology and application. Garland Science/Bios Scientific Publishers, New York
Oktay M, Gülçin İ, Küfrevioğlu Öİ (2003) Determination of in vitro antioxidant activity of fennel (Foeniculum vulgare) seed extracts. LWT-Food Sci Technol 36(2):263–271. https://doi.org/10.1016/S0023-6438(02)00226-8
Pillai R, Redmond M, Röding J (2005) Anti-wrinkle therapy: significant new findings in the non‐invasive cosmetic treatment of skin wrinkles with beta‐glucan. Int J Cosmet Sci 27(5):292–292. https://doi.org/10.1111/j.1463-1318.2005.00268_3.x
Pillemer L, Ecker EE (1941) Anti-complementary factor in fresh yeast. J Biol Chem 137:139–142
Priyanka S, Jayashree M, Shivani R, Anwesha S, Rao KVB (2018) A study on marine lipolytic yeasts: optimization and characterization of lipase enzyme. Res J Pharm Technol 11(2):593–598. https://doi.org/10.5958/0974-360X.2018.00109.9
Safaee-Ardakani MR, Hatamian-Zarmi A, Sadat SM, Alvandi H, Ebrahimi-Hosseinzadeh B, Mokhtari-Hosseini ZB, Mohammadi M (2022) In vivo study of beta-glucan-based biogenic synthesis of silver nanocomposite using Schizophyllum commune for wound dressings in a rat burn model. Adv Nat Sci Nanosci Nanotechnol 13(3):035001. https://doi.org/10.1088/2043-6262/ac79b6
Samuelsen ABC, Schrezenmeir J, Knutsen SH (2014) Effects of orally administered yeast-derived beta‐glucans: a review. Mol Nutr Food Res 58(1):183–193. https://doi.org/10.1002/mnfr.201300338
Santipanichwong R, Suphantharika M (2009) Influence of different β-glucans on the physical and rheological properties of egg yolk stabilized oil-in-water emulsions. Food Hydrocoll 23(5):1279–1287. https://doi.org/10.1016/j.foodhyd.2008.10.006
Sarkar Anwesha, Rao KVB (2016) Marine yeast: a potential candidate for biotechnological applications–A review. Asian J Microbiol Biotechnol Environ Sci 18(3):627–634
Sarkar A, Philip AM, Thakker DP, Wagh MS, Rao KVB (2020) In vitro antioxidant activity of extracellular L-glutaminase enzyme isolated from marine yeast Rhodotorula Sp DAMB1. Res J Pharm Technol 13(1):209–215. https://doi.org/10.5958/0974-360X.2020.00042.6
Sarlin PJ, Philip R (2011) Efficacy of marine yeasts and baker’s yeast as immunostimulants in Fenneropenaeus indicus: a comparative study. Aquaculture 321(3–4):173–178. https://doi.org/10.1016/j.aquaculture.2011.08.039
Schepetkin IA, Quinn MT (2006) Botanical polysaccharides: macrophage immunomodulation and therapeutic potential. Int Immunopharmacol 6(3):317–333. https://doi.org/10.1016/j.intimp.2005.10.005
Schwartz B, Vetvicka V (2021) Review: β-glucans as Effective Antibiotic Alternatives in Poultry. Molecules 26(12):3560. https://doi.org/10.3390/molecules26123560
Shimada K, Fujikawa K, Yahara K, Nakamura T (1992) Antioxidative properties of xanthan on the autoxidation of soybean oil in cyclodextrin emulsion. J Agric Food Chem 40(6):945–948. https://doi.org/10.1021/jf00018a005
Singhal M, Paul A, Singh HP (2014) Synthesis and reducing power assay of methyl semicarbazone derivatives. J Saudi Chem Soc 18(2):121–127. https://doi.org/10.1016/j.jscs.2011.06.004
Song J, Chen H, Wei Y, Liu J (2020) Synthesis of carboxymethylated β-glucan from naked barley bran and its antibacterial activity and mechanism against Staphylococcus aureus. Carbohydr Polym 242:116418. https://doi.org/10.1016/j.carbpol.2020.116418
Tanngoen PI, Lamlertthon S, Tiyaboonchai W (2019) Effects of alpha-mangostin and citronella oil against acne-causing bacteria. Int J Pharm Sci 11:45–49
Thenmozhi M, Sindhura S, Kannabiran K (2010) Characterization of antioxidant activity of Streptomyces species VITTK3 isolated from Puducherry Coast, India. J Adv Sci Res 1(02):46–52
Thompson IJ, Oyston PC, Williamson DE (2010) Potential of the β-glucans to enhance innate resistance to biological agents. Expert Rev Anti Infect Ther 8(3):339–352. https://doi.org/10.1586/eri.10.10
Tohamy AA, El-Ghor AA, El-Nahas SM, Noshy MM (2003) β-glucan inhibits the genotoxicity of cyclophosphamide, adriamycin and cisplatin. Mutat Res Genet Toxicol Environmen Mutagen 541(1–2):45–53. https://doi.org/10.1016/S1383-5718(03)00184-0
Vetvicka V, Vetvickova J (2011) β (1–3)-D-glucan affects adipogenesis, wound healing and inflammation. Orient Pharm Exp Med 11(3):169–175. https://doi.org/10.1007/s13596-011-0024-4
Volman JJ, Ramakers JD, Plat J (2008) Dietary modulation of immune function by β-glucans. Physiol Behav 94(2):276–284. https://doi.org/10.1016/j.physbeh.2007.11.045
Wang L, Chi Z, Wang X, Liu Z, Li J (2007) Diversity of lipase-producing yeasts from marine environments and oil hydrolysis by their crude enzymes. Ann Microbiol 57(4):495–501
Wang Y, Yang Z, Wei X (2012) Antioxidant activities potential of tea polysaccharide fractions obtained by ultra filtration. Int J Biol Macromol 50(3):558–564. https://doi.org/10.1016/j.ijbiomac.2011.12.028
Williams DL, Pretus HA, McNamee RB, Jones EL, Ensley HE, Browder IW (1992) Development of a water-soluble, sulfated (1→ 3)-β-D-glucan biological response modifier derived from Saccharomyces cerevisiae. Carbohydr Res 235:247–257. https://doi.org/10.1016/0008-6215(92)80093-G
Xin Y, Ji H, Cho E, Roh KB, You J, Park D, Jung E (2022) Immune-enhancing effect of water-soluble beta-glucan derived from enzymatic hydrolysis of yeast glucan. Biochem Biophys Rep 30:101256. https://doi.org/10.1016/j.bbrep.2022.101256
Zaky AS, Tucker GA, Daw ZY, Du C (2014) Marine yeast isolation and industrial application. FEMS Yeast Res 14(6):813–825. https://doi.org/10.1111/1567-1364.12158
Zhu F, Du B, Xu B (2016) A critical review on production and industrial applications of beta-glucans. Food Hydrocoll 52:275–288. https://doi.org/10.1016/j.foodhyd.2015.07.003
Zulli F, Suter F, Biltz H, Nissen HP (1998) Improving skin function with CM-glucan, a biological response modifier from yeast. Int J Cosmet Sci 20(2):79–86. https://doi.org/10.1046/j.1467-2494.1998.171740.x
Acknowledgements
The authors are highly grateful to the whole management of Vellore Institute of Technology, Vellore, Tamil Nadu for delivering the essential support and facilities including GCMS and SEM (DST-FIST) for the completion of the work.
Funding
The authors declare that no fund and financial support was received during the preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
All persons who meet the authorship criteria are listed as authors, and all authors certify that they have participated sufficiently in the work to take public responsibility for the content, including participation in the concept, design, acquisition and analysis of the data, writing, or revision of the manuscript. All the authors have read and approved the manuscript. Furthermore, all authors certify that this material or similar material has not been and will not be submitted to or published in any other publication before its appearance in International Microbiology. Conceptualization: K. V. Bhaskara Rao, Anwesha Sarkar; Methodology: K.V. Bhaskara Rao, Anwesha Sarkar; Formal analysis and investigation: Anwesha Sarkar, Angelin Mary Philip, Darshali P. Thakker; Data curation: Angelin Mary Philip and Darshali P. Thakker; Writing- Original draft preparation: Anwesha Sarkar; Writing- Review and editing: K. V. Bhaskara Rao and Anwesha Sarkar; Resources: K. V. Bhaskara Rao; Supervision: K. V. Bhaskara Rao.
Corresponding author
Ethics declarations
Ethical Approval
Not applicable.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sarkar, A., Philip, A.M., Thakker, D.P. et al. Extraction and Characterization of Beta-glucan Methyl Esters Derived From UV-mutated Marine Yeast and their Applications in Cosmetics. Thalassas 40, 721–733 (2024). https://doi.org/10.1007/s41208-024-00661-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41208-024-00661-x