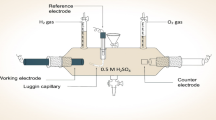
Abstract
A thin layer of platinum coating (0.5 µgcm−2) on stainless steel surface was deposited by direct current (DC) and pulse current (PC) electrodeposition method for hydrogen evolution reaction (HER) application. Scanning electron microscopy, atomic force microscopy, and X-ray diffraction analysis were used to characterize the coatings. Linear sweep voltammetry and cyclic voltammetry studies were carried out to know the overpotential values for hydrogen evolution reaction (HER) on these coatings. The optimization of catalytic activity for hydrogen evolution using different coating methods helps in reducing the overall cost. Tafel polarization experiments were conducted for DC and PC platinum coating to know hydrogen generation trend. Cathodic slope and HER current values revealed that, coatings obtained at 75% duty cycle by PC method exhibit lower cathodic slope, high current density of 150 mA/cm2 and more corrosion current with highest hydrogen evolution. Chronopotentiometry experiments showed that 20 ml of hydrogen was collected from 75% sample.









Similar content being viewed by others
Data Availability
The raw/processed data required to reproduce these findings are available from the corresponding author on reasonable request.
References
Ahmed NS, Menzel R, Wang Y, Garcia- GA, Bawaked SM, Obaid AY, Basahel SN, Mokhtar M (2017) Graphene-oxide-supported CuAl and CoAl layered double hydroxides as enhanced catalysts for carbon-carbon coupling via Ullmann reaction. J Solid State Chem 246:130–137. https://doi.org/10.1016/J.JSSC.2016.11.024
Aliaksandr Bandarenka S, Koper MTM (2013) Structural and electronic effects in heterogeneous electrocatalysis: toward a rational design of electrocatalysts. J Catal 308:11–24. https://doi.org/10.1016/j.jcat.2013.05.006
Bicakova O, Straka P (2012) Production of hydrogen from renewable resources and its effectiveness. Int J Hydrog Energy 37:11563–11578. https://doi.org/10.1016/j.ijhydene.2012.05.047
Carmo M, Fritz DL, Mergel J, Stolten D (2013) A comprehensive review on PEM water electrolysis. Int J Hydrog Energy 38:4901–4934. https://doi.org/10.1016/j.ijhydene.2013.01.151
El-Shobaky GA, Ahmad AS, Mokhtar M (1997) Effect of gamma-irradiation on surface and catalytic properties of CuO–ZnO/Al2O3 system. J Radioanal Nucl Chem 219:89–94. https://doi.org/10.1007/bf02040271
Holewinski A, Xin H, Nikolla E, Linic S (2013) Identifying optimal active sites for heterogeneous catalysis by metal alloys based on molecular descriptors and electronic structure engineering. Curr Opin Chem Eng 2:312–319. https://doi.org/10.1016/j.coche.2013.04.006
Hussain S, Akbar K, Vikraman D, Arslan A, Song W, An Ki-Seok, Farooq A, Jun-Young P, Seung-Hyun C, Jongwan J (2018) WS(1–x)sex nanoparticles decorated three-dimensional graphene on nickel foam: a robust and highly efficient electrocatalyst for the hydrogen evolution reaction. Nanomaterials. 8:929. https://doi.org/10.3390/nano8110929
Jakub T, Calle-Vallejo F, Wolfgang S, Bandarenka AS (2016) Making the hydrogen evolution reaction in polymer electrolyte membrane electrolysers even faster. Nat Commun. https://doi.org/10.1038/ncomms10990
Jeong Eun Y, Abdulohsen Ali A, Shanshan Q, Salem Mohamed B, Mohamed Mokhtar MM, Narasimharao K, Dominik F, Jochen S, Anca M, Nikita D, Gihoon G, Karsten M, Patrik S (2020) Establishing high photocatalytic H2 evolution from multiwalled titanate nanotubes. ChemCatChem. 12:2951–2956. https://doi.org/10.1002/cctc.202000281
Jeong Eun Y, Marco A, Mohamed M, Abdulmohsen AA, Al-Thabaiti SA, Anca M, Schmuki P (2016) Photocatalytic H2 generation using dewetted Pt-decorated TiO2 nanotubes: optimized dewetting and oxide crystallization by a multiple annealing process. J Phys Chem C. https://doi.org/10.1021/acs.jpcc.5b12050
Koper MTM (2011) Structure sensitivity and nanoscale effects in electrocatalysis. Nanoscale 3:2054–2073. https://doi.org/10.1039/C0NR00857E
Mohsen S, Seyedsina H, Shiva M, Ondrej T, Mokhtar M, Abdulmohsen A, Sohrab S, Radek Z, Patrik S (2019) Photocatalytic H2 evolution: dealloying as efficient tool for the fabrication of Rh-decorated TiO2 nanotubes. ChemCatChem 11:6258–6262. https://doi.org/10.1002/cctc.201901183
Mokhtar M, Ohlinger O, Schlander H, Turek T (2001) Hydrogenolysis of dimethyl maleate on Cu/ZnO/Al2O3 catalysts. Chem Eng Technol 24. https://doi.org/10.1002/1521-4125(200104)24:4<423::AID-CEAT423>3.0.CO;2-G
Muralikrishna S, ManjunathK SD, Reddy V, Ramakrishnappa T, Nagaraju DH (2015) Hydrothermal synthesis of 2D MoS2 nanosheets for electrocatalytic hydrogen evolution reaction. RSC Adv 5:89389. https://doi.org/10.1039/C5RA18855E
Nagi Radwan RE, Mokhtar M, El-Shobaky GA (2003) Surface and catalytic properties of CuO and Co3O4 solids as influenced by treatment with Co2+ and Cu2+ species. Appl Catal A 241:77–90. https://doi.org/10.1016/S0926-860X(02)00459-3
Norskov J, Bligaard K, Rossmeisl T, Christensen CH (2009) Towards the computational design of solid catalysts. Nat Chem 1:37–46
StephensIfan EL, Bondarenko Alexnader S, Gronbjerg U, Rossmeisl J, Chorkendorff I (2012) Understanding the electrocatalysis of oxygen reduction on platinum and its alloys. Energy Environ Sci 5:6744–6762. https://doi.org/10.1039/C2EE03590A
Subbaraman R, Dusan T, Dusan S, Kee-Chul C, Masanobu U, Arvydas P, Vojislav S, Nenad M (2011) Enhancing hydrogen evolution activity in water splitting by tailoring Liþ–Ni(OH)2–Pt interfaces. Science 334:1256–1260. https://doi.org/10.1126/science.1211934
Symes Mark D, Cronin L (2013) Decoupling hydrogen and oxygen evolution during electrolytic water splitting using an electron-coupled-proton buffer. Nat Chem 5:403–409. https://doi.org/10.1038/NCHEM.1621
Yang XF, Aiqin W, Botao Q, Jun L, Jingyue L, Tao Z (2013) Single-atom catalysts: a new frontier in heterogeneous catalysis. Acc Chem Res 46:1740–1748
Yin HJ, Shenlong Z, Kun Z, Abdul M, Hongjie T, Lin C, Huijun Z, Yan G, Zhiyong T (2015) Ultrathin platinum nanowires grown on single-layered nickel hydroxide with high hydrogen evolution activity. Nat Commun 6:6430. https://doi.org/10.1038/ncomms7430
Acknowledgments
With the approval of Project No. 37(2)/14/18/2018, dated 11/07/2018, instrumental and financial funding from the Department of Atomic Energy (DAE) Board of Research in Nuclear Sciences (BRNS), Mumbai, and Government of India. Srinivas University, College of Engineering & Technology, Srinivasnagar, Mangaluru, Karnataka, has provided laboratory support.
Funding
The work was funded by the Department of Atomic Energy (DAE), Board of Research in Nuclear Sciences (BRNS), Mumbai, Government of India, with the approval of Project No. 37(2)/14/18/2018-BRNS, 11/07/2018. The writers express their gratitude for providing laboratory support from Srinivas University, College of Engineering & Technology, Srinivasnagar, Mangaluru, Karnataka.
Author information
Authors and Affiliations
Contributions
BKD is involved in conceptualization, methodology, visualization, investigation, software, data curation, and writing-original drafts. BMP is involved in methodology, article writing, formal analysis, resources, project administration, data curation and investigation. VST involved in giving suggestions for correction of the manuscript, supervision and project administration. Participated in giving suggestions for correction of the document, enquiry, visual analytics and editing. DHN participated in giving suggestions for correction of the manuscript, investigation, visualization and editing. KON was involved in the review of the manuscript and editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and Animal Rights
No persons/animals were used in our study.
Consent to Participate
All authors accepted for the participation of the Work in ‘‘Iranian Journal of Science and Technology, Transaction A: Science.
Consent to Publish
The Authors hereby consents to publication of the Work in ‘‘Iranian Journal of Science and Technology, Transaction A: Science.
Rights and permissions
About this article
Cite this article
Devendra, B.K., Praveen, B.M., Tripathi, V.S. et al. Hydrogen Evolution Reaction by Platinum Coating. Iran J Sci Technol Trans Sci 45, 1993–2000 (2021). https://doi.org/10.1007/s40995-021-01220-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40995-021-01220-2