Abstract
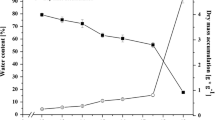
Seeds may desiccate after independence from parent plant and remain in a dry inactive state until the conditions for germination are conducive. Dehydrin proteins pattern and expression in mature pistachio seeds were surveyed at fresh and 48-h desiccated states by western blot method, using an antibody against conserved K-segment. Mature seeds lost 92.4% moisture after desiccation without any loss in complete germinability. Five dehydrin bands were detected in both cotyledon and embryonic axis. At fresh state, 23, 25, and 48 kDa bands had a lower content in cotyledon than in axis, but increased to an equal level by desiccation. The 32 and 39 kDa bands were detected only in fresh stage, preferentially in cotyledons, and disappeared after desiccation; therefore, they have other roles rather than dehydration protection. Some bands of 17, 19, 20, 28, 67, 77 and 98 kDa were detected only in axes at both states. Among them, the 28 kDa band was only used for densitometry and was not stimulated by dehydration. Further studies are required to establish the exact role of dehydrins which do not have a role in dehydration tolerance.



Similar content being viewed by others
Abbreviations
- DC:
-
Desiccated cotyledon
- DE:
-
Desiccated embryonic axis
- FC:
-
Fresh cotyledon
- FE:
-
Fresh embryonic axis
- kDa:
-
Kilo Dalton
- MC:
-
Moisture content
References
Angelovici R, Galili G, Fernie AR, Fait A (2010) Seed desiccation: a bridge between maturation and germination. Trends Plant Sci 15:211–218
Ali-Benali MA, Alary R, Joudrier P, Gautier MF (2005) Comparative expression of five Lea genes during wheat seed development and in response to abiotic stresses by real-time quantitative RT-PCR. BBA-Gene Struct Expr 1730:56–65
Banerjee A, Roychoudhury A (2016) Group II late embryogenesis abundant (LEA) proteins: structural and functional aspects in plant abiotic stress. Plant Growth Regul 79:1–17
Barreca D, Laganà G, Leuzzi U, Smeriglio A, Trombetta D, Bellocco E (2016) Evaluation of the nutraceutical, antioxidant and cytoprotective properties of ripe pistachio (Pistachia vera L. variety Bronte) hulls. Food Chem 196:493–502
Boudet J, Buitink J, Hoekstra FA, Rogniaux H, Larré C, Satour P, Leprince O (2006) Comparative analysis of the heat stable proteome of radicles of Medicago truncatula seeds during germination identifies late embryogenesis abundant proteins associated with desiccation tolerance. Plant Physiol 140(4):1418–1436
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Burrieza HP, López-Fernández MP, Chiquieri TB, Silveira V, Maldonado S (2012) Accumulation pattern of dehydrins during sugarcane (var. SP80. 3280) somatic embryogenesis. Plant Cell Rep 31:2139–2149
Chatelain E, Hundertmark M, Leprince O, Le Gall S, Satour P, Deligny-Penninck S, Rogniaux H, Buitink J (2012) Temporal profiling of the heat-stable proteome during late maturation of Medicago truncatula seeds identifies a restricted subset of late embryogenesis abundant proteins associated with longevity. Plant Cell Environ 35:1440–1455
Chen K, Fessehaie A, Arora R (2012) Dehydrin metabolism is altered during seed osmopriming and subsequent germination under chilling and desiccation in Spinacia oleracea L. cv. Bloomsdale: possible role in stress tolerance. Plant Sci 183:27–36
Clarke MW, Boddington KF, Warnica JM, Atkinson J, McKenna S, Madge J, Barker CH, Graether SP (2015) Structural and functional insights into the cryoprotection of membranes by the intrinsically disordered dehydrins. J Biol Chem 290:26900–26913
Close TJ (1996) Dehydrins: emergence of a biochemical role of a family of plant dehydration proteins. Physiol Plant 97:795–803
Cuevas-Velazquez CL, Rendon-Luna DF, Covarrubias AA (2014) Dissecting the cryoprotection mechanisms for dehydrins. Front Plant Sci 5:583
Debeaujon I, Léon-Kloosterziel KM, Koornneef M (2000) Influence of the testa on seed dormancy, germination, and longevity in Arabidopsis. Plant Physiol 122:403–414
Eriksson SK, Kutzer M, Procek J, Gröbner G, Harryson P (2011) Tunable membrane binding of the intrinsically disordered dehydrin Lti30, a cold-induced plant stress protein. Plant Cell 23:2391–2404
Eriksson S, Eremina N, Barth A, Danielsson J, Harryson P (2016) Membrane-induced folding of the plant stress dehydrin Lti30. Plant Physiol 171:932–943
Farrant JM, Pammenter NW, Berjak P, Farnsworth EJ, Vertucci CW (1996) Presence of dehydrin-like proteins and levels of abscisic acid in recalcitrant (desiccation sensitive) seeds may be related to habitat. Seed Sci Res 6:175–182
Franchi GG, Piotto B, Nepi M, Baskin CC, Baskin JM, Pacini E (2011) Pollen and seed desiccation tolerance in relation to degree of developmental arrest, dispersal, and survival. J Exp Bot 62(15):5267–5281
Gallardo K, Firnhaber C, Zuber H, Héricher D, Belghazi M, Henry C, Küster H, Thompson R (2007) A combined proteome and transcriptome analysis of developing Medicago truncatula seeds: evidence for metabolic specialization of maternal and filial tissues. Mol Cell Proteomics 6:2165–2179
Garnczarska M, Zalewski T, Wojtyla Ł (2008) A comparative study of water distribution and dehydrin protein localization in maturing pea seeds. J Plant Physiol 165:1940–1946
Golan-Goldhirsh A (1998a) Developmental proteins of Pistacia vera L. bark and bud and their biotechnological properties: a review. J Food Biochem 22:375–382
Golan-Goldhirsh A (1998b) Seasonal changes in a dehydrin-like protein and proteases of Pistacia vera inflorescence buds. Acta Hortic (ISHS) 470:359–364
Gupta A, Marzinek JK, Jefferies D, Bond PJ, Harryson P, Wohland T (2019) The disordered plant dehydrin Lti30 protects the membrane during water-related stress by cross-linking lipids. J Biol Chem 294:6468–6482
Hanana M, Daldoul S, Fouquet R, Deluc L, Leon C, Hoefer M, Barrieu F, Ghorbel A (2014) Identification and characterization of a seed-specific grapevine dehydrin involved in abiotic stress response within tolerant varieties. Turk J Bot 38:1157–1168
Hara M, Shinoda Y, Tanaka Y, Kuboi T (2009) DNA binding of citrus dehydrin promoted by zinc ion. Plant Cell Environ 32(5):532–541
Hernandez-Sanchez IE, Martynowicz DM, Rodriguez-Hernandez AA, Perez-Morales MB, Graether SP, Jimenez-Bremont JF (2014) A dehydrin-dehydrin interaction: the case of SK3 from Opuntia streptacantha. Front Plant Sci 5:520
Hoekstra FA, Golovina EA, Buitink J (2001) Mechanisms of plant desiccation tolerance. Trends Plant Sci 6:431–438
Kalemba EM, Pukacka S (2008) Changes in late embryogenesis abundant proteins and a small heat shock protein during storage of beech (Fagus sylvatica L.) seeds. Environ Exp Bot 63:274–280
Kalemba EM, Pukacka S (2012) Association of protective proteins with dehydration and desiccation of orthodox and recalcitrant category seeds of three Acer genus species. J Plant Growth Regul 31:351–362
Kalemba EM, Bagniewska-Zadworna A, Ratajczak E (2015) Multiple subcellular localizations of dehydrin-like proteins in the embryonic axes of common beech (Fagus sylvatica L.) seeds during maturation and dry storage. J Plant Growth Regul 34:137–149
Kermode AR (1997) Approaches to elucidate the basis of desiccation-tolerance in seeds. Seed Sci Res 7:75–95
Kumar P, Sharma SK, Chandel RS, Singh J, Kumar A (2016) Nutrient dynamics in pistachios (Pistacia vera L.): the effect of mode of nutrient supply on agronomic performance and alternate-bearing in dry temperate ecosystem. Sci Hortic 210:108–121
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Nachbar M, Mozafari M, Krull F, Maul KJ, Preu L, Hara M, Watzig H (2017) Metal ion—Dehydrin interactions investigated by affinity capillary electrophoresis and computer models. J Plant Physiol 216:219–228
Ouellet F, Houde M, Sarhan F (1993) Purification, characterization and cDNA cloning of the 200 kDa protein induced by cold acclimation in wheat. Plant Cell Physiol 34:59–65
Panza V, Distéfano AJ, Carjuzaa P, Láinez V, Del Vas M, Maldonado S (2007) Detection of dehydrin-like proteins in embryos and endosperm of mature Euterpe edulis seeds. Protoplasma 231:1–5
Prieto-Dapena P, Castaño R, Almoguera C, Jordano J (2006) Improved resistance to controlled deterioration in transgenic seeds. Plant Physiol 142:1102–1112
Rao NK, Hanson J, Dulloo ME, Ghosh K, Nowel D, Larinde M (2006) Manual of seed handling in genebanks. In: Handbooks for Genebanks No. 8. Bioversity International, Rome, Italy. Available in English, Spanish and French, pp 50–51
Rorat T (2006) Plant dehydrins—tissue location, structure and function. Cell Mol Biol Lett 11:536–556
Shen-Miller J, Mudgett MB, Schopf JW, Clarke S, Berger R (1995) Exceptional seed longevity and robust growth: ancient scared lotus from China. Am J Bot 82:1367–1380
Vaseva II, Anders I, Feller U (2014) Identification and expression of different dehydrin subclasses involved in the drought response of Trifolium repens. J Plant Physiol 171:213–224
Verdier J, Lalanne D, Pelletier S, Torres-Jerez I, Righetti K, Bandyopadhyay K, Leprince O, Chatelain E, Vu BL, Gouzy J, Gamas P, Udvardi MK, Buitink J (2013) A regulatory network-based approach dissects late maturation processes related to the acquisition of desiccation tolerance and longevity of Medicago truncatula seeds. Plant Physiol 163(2):757–774
Vertucci CW, Farrant JM (1995) Acquisition and loss of desiccation tolerance. In: Galili G, Kigel J (eds) Seed development and germination. Marcel Dekker, New York, pp 701–746
Waterworth WM, Masnavi G, Bhardwaj RM, Jiang Q, Bray CM, West CE (2010) A plant DNA ligase is an important determinant of seed longevity. Plant J 63:848–860
Yakubov B, Barazani O, Shachack A, Rowland LJ, Shoseyov O, Golan-Goldhirsh A (2005) Cloning and expression of a dehydrin-like protein from Pistacia vera L. Trees 19:224–230
ZamaniBahramabadi E, Jonoubi P, Rezanejad F (2018) Some cytological and physiochemical features relating to non-storability of pistachio (Pistacia vera L.) pollen. Grana. https://doi.org/10.1080/00173134.2018.1476584
Acknowledgements
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors agree on the content of the paper and have no conflict of interest.
Rights and permissions
About this article
Cite this article
ZamaniBahramabadi, E., Jonoubi, P. & Rezanejad, F. Dehydrin Content in Fresh and Desiccated Pistachio (Pistacia vera L.) Seeds. Iran J Sci Technol Trans Sci 43, 2099–2105 (2019). https://doi.org/10.1007/s40995-019-00712-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40995-019-00712-6