Abstract
Purpose
To examine the factors that affect inter/intra-observer variability for breast cancer grading on histopathology images, to compare the level of agreement between histopathologists for assessment of virtual slides, and to introduce a new application, CACTUS, for the quality assurance of the cancer grading programme.
Methods
A new web-based tool for image archiving, annotation, distribution and evaluation (CACTUS) was developed. Four pathologists of varying practice experience in surgical pathology took part in three web-based image circulations using CACTUS. All pathologists evaluated the same 50 images, of which ten images were repeated to assess consistency. Reproducibility was evaluated by intra-observer (intraclass correlation coefficient) and inter-observer (kappa statistics) concordance rates and additional analyses.
Results
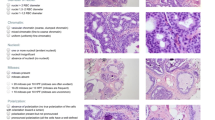
All pathologists found the interface of the tool and presentations of the images quick to learn and easy to use. The pathologists were asked to evaluate all of the images using a menu of questions meant to determine their assessment of the Nottingham grade of a series of \(\times\)40 images of breast carcinoma cases. For each image, they were asked to give their overall grade impression based on the \(\times\)40 single image. They were then directed to separate menus of questions to assess the principal components of Nottingham grading: mitotic rate, nuclear morphology and glandular morphology. Their consistency in re-evaluating the set of ten images was excellent agreement (> 0.90). While the overall inter-agreement for three sets is moderate (0.50–0.75), the overall intra-agreement between initial grades and mean value of mitosis, tubular and nuclear scores is excellent, even though opinions were based on single, \(\times\)40 images. The correlation between initial grade and four criteria/features (mitotic rate, nuclear pleomorphism, tubule formation and the mean value of these three criteria) has been calculated to show which feature is important among others or contributes to the actual score more. Tubular and nuclear scores are highly correlated with initial grades. Outliers among observers are identified by finding their distances from each other and computing the clusters.
Conclusion
This easy to use, easily modifiable, web-based image archiving, distribution and assessment tool can be useful for evaluating agreement amongst pathologists and disseminating anatomical pathology images for teaching. Moreover, it can be used against a gold standard for the purpose of diagnosis or quality assurance.








Similar content being viewed by others
References
Sinard, J. H. (2006). Pathology Informatics and The Future of Medicine. Practical Pathology Informatics: Demstifying Informatics for the Practicing Anatomic Pathologist (pp. 355–380). New York: Springer.
Leong, F. J., Leong, A. S., et al. (2004). Digital photography in anatomical pathology. Journal of Postgraduate Medicine, 50(1), 62.
Farahani, N., & Pantanowitz, L. (2015). Overview of telepathology. Surgical Pathology Clinics, 8(2), 223–231.
Griffin, J., & Treanor, D. (2017). Digital pathology in clinical use: where are we now and what is holding us back? Histopathology, 70(1), 134–145.
Furness, P. N. (1997). The use of digital images in pathology. The Journal of Pathology: A Journal of the Pathological Society of Great Britain and Ireland, 183(3), 253–263.
Al-Janabi, S., Huisman, A., & Van Diest, P. J. (2012). Digital pathology: current status and future perspectives. Histopathology, 61(1), 1–9.
Irshad, H., Montaser-Kouhsari, L., Waltz, G., Bucur, O., Nowak, J. A., Dong, F., Knoblauch, N. W., & Beck, A. H. (2014). Crowdsourcing image annotation for nucleus detection and segmentation in computational pathology: Evaluating experts, automated methods, and the crowd. In Pacific symposium on biocomputing Co-chairs, pp. 294–305. World Scientific.
Park, S. L., Pantanowitz, L., Sharma, G., & Parwani, A. V. (2012). Anatomic pathology laboratory information systems: A review. Advances in Anatomic Pathology, 19(2), 81–96.
Dee, F. R. (2009). Virtual microscopy in pathology education. Human Pathology, 40(8), 1112–1121.
Wright, A. I., Clarke, E. L., Dunn, C. M., Williams, B. J., Treanor, D. E., & Brettle, D. S. (2020). A point-of-use quality assurance tool for digital pathology remote working. Journal of Pathology Informatics, 11, 17.
Aksac, A., Ozyer, T., Demetrick, D. J., & Alhajj, R. (2020). Cactus: cancer image annotating, calibrating, testing, understanding and sharing in breast cancer histopathology. BMC Research Notes, 13(1), 1–7.
Jahn, S. W., Plass, M., & Moinfar, F. (2020). Digital pathology: Advantages, limitations and emerging perspectives. Journal of Clinical Medicine, 9(11), 3697.
Hamilton, P. W., Wang, Y., & McCullough, S. J. (2012). Virtual microscopy and digital pathology in training and education. Apmis, 120(4), 305–315.
Elston, C. W., & Ellis, I. O. (1991). Pathological prognostic factors in breast cancer. i. the value of histological grade in breast cancer: experience from a large study with long-term follow-up. Histopathology, 19(5), 403–410.
Bloom, H. J. G., & Richardson, W. W. (1957). Histological grading and prognosis in breast cancer: a study of 1409 cases of which 359 have been followed for 15 years. British Journal of Cancer, 11(3), 359.
Robbins, P., Pinder, S., De Klerk, N., Dawkins, H., Harvey, J., Sterrett, G., Ellis, I., & Elston, C. (1995). Histological grading of breast carcinomas: A study of interobserver agreement. Human Pathology, 26(8), 873–879.
Frierson, H. F., Wolber, R. A., Berean, K. W., Franquemont, D. W., Gaffey, M. J., Boyd, J. C., & Wilbur, D. C. (1995). Interobserver reproducibility of the nottingham modification of the bloom and richardson histologic grading scheme for infiltrating ductal carcinoma. American Journal of Clinical Pathology, 103(2), 195–198.
Longacre, T. A., Ennis, M., Quenneville, L. A., Bane, A. L., Bleiweiss, Ira J., Carter, B. A., Catelano, E., Hendrickson, M. R., Hibshoosh, H., Layfield, L. J., et al. (2006). Interobserver agreement and reproducibility in classification of invasive breast carcinoma: An nci breast cancer family registry study. Modern Pathology, 19(2), 195.
Boiesen, P., Bendahl, P.-O., Anagnostaki, L., Domanski, H., Holm, E., Idvall, I., Johansson, S., Ljungberg, O., & Ringberg, A. (2000). Histologic grading in breast cancer: Reproducibility between seven pathologic departments. Acta Oncologica, 39(1), 41–45.
Gomes, D. S., Porto, S. S., Balabram, D., & Gobbi, H. (2014). Inter-observer variability between general pathologists and a specialist in breast pathology in the diagnosis of lobular neoplasia, columnar cell lesions, atypical ductal hyperplasia and ductal carcinoma in situ of the breast. Diagnostic Pathology, 9(1), 121.
van Riel, S. J., Sánchez, C. I., Bankier, A. A., Naidich, D. P., Verschakelen, J., Scholten, E. T., de Jong, P. A., Jacobs, C., van Rikxoort, E., Peters-Bax, L., et al. (2015). Observer variability for classification of pulmonary nodules on low-dose ct images and its effect on nodule management. Radiology, 277(3), 863–871.
Bueno-de Mesquita, J. M., Nuyten, D. S. A., Wesseling, J., van Tinteren, H., Linn, S. C., & van De Vijver, M. J. (2010). The impact of inter-observer variation in pathological assessment of node-negative breast cancer on clinical risk assessment and patient selection for adjuvant systemic treatment. Annals of Oncology, 21(1), 40–47.
Gilchrist, K. W., Kalish, L., Gould, V. E., Hirschl, S., Imbriglia, J. E., Levy, W. M., Patchefsky, A. S., Penner, D. W., Pickren, J., Roth, J. A., et al. (1985). Interobserver reproducibility of histopathological features in stage ii breast cancer. Breast Cancer Research and Treatment, 5(1), 3–10.
Aksac, A., Demetrick, D. J., Ozyer, T., & Alhajj, R. (2019). Brecahad: A dataset for breast cancer histopathological annotation and diagnosis. BMC Research Notes, 12(1), 82.
Kayser, K., Kayser, G., Radziszowski, D., & Oehmann, A. (1999). From telepathology to virtual pathology institution: the new world of digital pathology. Romanian Journal of Morphology and Embryology, 45, 3–9.
Szymas, J. (2000). Teleeducation and telepathology for open and distance education. Analytical Cellular Pathology, 21(3–4), 183–191.
Preston, K., Jr. (1986). High-resolution image analysis. Journal of Histochemistry & Cytochemistry, 34(1), 67–74.
Jondet, M., Agoli-Agbo, R., & Dehennin, L. (2010). Automatic measurement of epithelium differentiation and classification of cervical intraneoplasia by computerized image analysis. Diagnostic Pathology, 5(1), 7.
Kamenetsky, I., Rangayyan, R. M., & Benediktsson, H. (2010). Analysis of the glomerular basement membrane in images of renal biopsies using the split-and-merge method: a pilot study. Journal of Digital Imaging, 23(4), 463–474.
Loukas, C., Kostopoulos, S., Tanoglidi, A., Glotsos, D., Sfikas, C., & Cavouras, D. (2013). Breast cancer characterization based on image classification of tissue sections visualized under low magnification. Computational and Mathematical Methods in Medicine. https://doi.org/10.1155/2013/829461
Yoshida, H., Shimazu, T., Kiyuna, T., Marugame, A., Yamashita, Y., Cosatto, E., Taniguchi, H., Sekine, S., & Ochiai, A. (2018). Automated histological classification of whole-slide images of gastric biopsy specimens. Gastric Cancer, 21(2), 249–257.
Fung, K.-M., & Tihan, T. (2009). Internet and world wide web-based tools for neuropathology practice and education. Brain Pathology, 19(2), 323–331.
Jones, K. N., Kreisle, R., Geiss, R. W., Holliman, J. H., Lill, P. H., & Anderson, P. G. (2002). Group for research in pathology education online resources to facilitate pathology instruction. Archives of Pathology & Laboratory Medicine, 126(3), 346–350.
Chen, Y.-K., Hsue, S-S., Lin, D.-C., Wang, W-C., Chen, J.-Y., Lin, C.-C., & Lin, L.-M. (2008). An application of virtual microscopy in the teaching of an oral and maxillofacial pathology laboratory course. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontology, 105(3), 342–347.
Johnston, D. J., Costello, S. P., Dervan, P. A., & O’Shea, D. G. (2005). Development and preliminary evaluation of the vps replaysuite: a virtual double-headed microscope for pathology. BMC Medical Informatics and Decision Making, 5(1), 10.
Bruch, L. A., De Young, B. R., Kreiter, C. D., Haugen, T. H., Leaven, T. C., & Dee, F. R. (2009). Competency assessment of residents in surgical pathology using virtual microscopy. Human Pathology, 40(8), 1122–1128.
Dessauvagie, B. F., Lee, A. H. S., Meehan, K., Nijhawan, A., Tan, P. H, Thomas, J., Tie, B., Treanor, D., Umar, S., Hanby, A. M., et al. (2018). Interobserver variation in the diagnosis of fibroepithelial lesions of the breast: a multicentre audit by digital pathology. Journal of Clinical Pathology, 71(8), 672–679.
Linder, E., Lundin, M., Thors, C., Lebbad, M., Winiecka-Krusnell, J., Helin, H., Leiva, B., Isola, J., & Lundin, J. (2008). Web-based virtual microscopy for parasitology: a novel tool for education and quality assurance. NLoS neglected Tropical Diseases, 2(10), 10371
Kldiashvili, E, & Schrader, T. (2011). Reproducibility of telecytology diagnosis of cervical smears in a quality assurance program: the georgian experience. Telemedicine and e-Health, 17(7), 565–568.
Lee, E. S., Kim, I. S., Choi, J. S., Yeom, B. W., Kim, H. K., Han, J. H., Lee, M. S., & Leong, A. S Y. (2003). Accuracy and reproducibility of telecytology diagnosis of cervical smears: a tool for quality assurance programs. American journal of Clinical Pathology, 119(3), 356–360.
Cristiani, P., Costa, S., Schincaglia, P., Garutti, P., Bianchi, P. S. D., Naldoni, C., Sideri, M., & Bucchi, L. (2014). An online quality assurance program for colposcopy in a population-based cervical screening setting in italy: Results on colposcopic impression. Journal of Lower Genital Tract Disease, 18(4), 309–313.
Theissig, F., Kunze, K. D., Haroske, G., & Meyer, W. (1990). Histological grading of breast cancer: Interobserver, reproducibility and prognostic significance. Pathology-Research and Practice, 186(6), 732–736.
Malon, C., Brachtel, E., Cosatto, E., Graf, H. P., Kurata, A., Kuroda, M., Meyer, J. S., Saito, A., Shulin, W., & Yagi, Y. (2012). Mitotic figure recognition: Agreement among pathologists and computerized detector. Analytical Cellular Pathology, 35(2), 97–100.
Gandomkar, Z., Brennan, P. C., & Mello-Thoms, C. (2019). Computer-assisted nuclear atypia scoring of breast cancer: A preliminary study. Journal of Digital Imaging, 32(5), 702–712.
Rakha, E. A., Aleskandarani, M., Toss, M. S., Green, A. R., Ball, G., Ellis, I. O., & Dalton, L. W. (2018). Breast cancer histologic grading using digital microscopy: Concordance and outcome association. Journal of Clinical Pathology, 71(8), 680–686.
Dalton, L. W., Page, D. L., & Dupont, W. D. (1994). Histologic grading of breast carcinoma. A reproducibility study. Cancer, 73(11), 2765–2770.
Aksac, A., Özyer, T., & Alhajj, R. (2019). Cutesc: cutting edge spatial clustering technique based on proximity graphs. Pattern Recognition, 96, 106948.
Aksac, A., Ozyer, T., & Alhajj, R. (2020). Data on cut-edge for spatial clustering based on proximity graphs. Data in Brief, 28, 104899.
Cohen, J. (1960). A coefficient of agreement for nominal scales. Educational and Psychological Measurement, 20(1), 37–46.
Fleiss, J. L. (1971). Measuring nominal scale agreement among many raters. Psychological Bulletin, 76(5), 378.
Shrout, P. E., & Fleiss, J. L. (1979). Intraclass correlations: Uses in assessing rater reliability. Psychological Bulletin, 86(2), 420.
Landis, J. R., & Koch, G. G. (1977). The measurement of observer agreement for categorical data. Biometrics, 33, 159–174.
Altman, D. G. (1990). Practical Statistics for Medical Research. London: CRC Press.
Koo, T. K., & Li, M. Y. (2016). A guideline of selecting and reporting intraclass correlation coefficients for reliability research. Journal of Chiropractic Medicine, 15(2), 155–163.
Oommen, B. J., & Loke Loke, R. K S. (1997). Pattern recognition of strings with substitutions, insertions, deletions and generalized transpositions. Pattern Recognition, 30(5), 789–800.
Comaniciu, D., & Meer, P. (2002). Mean shift: A robust approach toward feature space analysis. IEEE Transactions on Pattern Analysis and Machine Intelligence, 24(5), 603–619.
R Core Team. (2018). R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/.
Contesso, G., Mouriesse, H., Friedman, S., Genin, J., Sarrazin, D., & Rouesse, J. (1987). The importance of histologic grade in long-term prognosis of breast cancer: A study of 1,010 patients, uniformly treated at the institut gustave-roussy. Journal of Clinical Oncology, 5(9), 1378–1386.
Henson, D. E., Ries, L., Freedman, L. S., & Carriaga, M. (1991). Relationship among outcome, stage of disease, and histologic grade for 22,616 cases of breast cancer. the basis for a prognostic index. Cancer, 68(10), 2142–2149.
Elmore, J. G., Barnhill, R. L., Elder, D. E., Longton, G. M., Pepe, M. S., Reisch, L. M., et al. (2017). Pathologists’ diagnosis of invasive melanoma and melanocytic proliferations: Observer accuracy and reproducibility study. BMJ, 357, 2813.
Geller, B. M., Nelson, H. D., Carney, P. A., Weaver, D. L., Onega, T., Allison, K. H., Frederick, P. D., Tosteson, A. N A., & Elmore, J. G. (2014). Second opinion in breast pathology: Policy, practice and perception. Journal of Clinical Pathology, 67(11), 955–960.
Swapp, R. E., Aubry, M. C., Salomão, D. R., & Cheville, J. C. (2013). Outside case review of surgical pathology for referred patients: The impact on patient care. Archives of Pathology & Laboratory Medicine, 137(2), 233–240.
Wright, K. C., Harnden, P., Moss, S., Berney, D. M., & Melia, J. (2011). A practical application of analysing weighted kappa for panels of experts and eqa schemes in pathology. Journal of Clinical Pathology, 64, 257–260.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Aksac, A., Demetrick, D.J., Box, A. et al. CACTUS: A Digital Tool for Quality Assurance, Education and Evaluation in Surgical Pathology. J. Med. Biol. Eng. 41, 470–481 (2021). https://doi.org/10.1007/s40846-021-00643-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40846-021-00643-x