Abstract
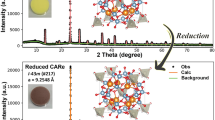
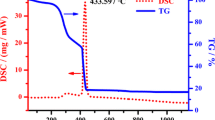
A new silicate fluoride, NaBa3Si2O7F, has been successfully synthesized by a high-temperature solution method. It crystallizes in the orthorhombic space group Cmcm (No. 63). NaBa3Si2O7F is the first barium-containing alkali metal silicate fluoride with the [NaO6] polyhedra, the [BaO8F] polyhedra and isolated [Si2O7] units. The optical characterizations indicate that NaBa3Si2O7F possesses wide transparent window and available luminescence properties. To confirm the coordination surroundings of anionic groups and its thermostability, infrared spectroscopy and thermal behaviors were also analyzed, which proved the existence of tetrahedronly coordinated silicium atoms and the good stability of NaBa3Si2O7F at high temperature. First-principles calculation was also implemented for better understanding the relationship between the structure of NaBa3Si2O7F and its property. Additionally, to further explore the structural novelty of NaBa3Si2O7F, the comparison of the anionic structures was carried out in mixed alkali and alkaline-earth metal silicate fluorides. Interestingly, the result indicates the isolated [Si2O7] dimer is rare among the above systems, which enriches the structural chemistry of silicate fluorides.
摘要
本文采用高温熔液法成功合成了新型硅酸盐氟化物NaBa3 Si2O7F. 它结晶于正交晶系, 空间群为Cmcm (No. 63). NaBa3Si2O7F 是已知的第一例含钡碱金属硅酸盐氟化物, 它包含了[NaO6]多面 体、[BaO8F]多面体和孤立的[Si2O7]二聚体. 光学表征表明该化合物具有宽的透过窗口和良好的荧光性质. 为了确定该化合物的阴离子基团的配位环境及热稳定性, 还对其进行了红外光谱测试和热学行为分析, 结果证明其结构中存在硅氧四面体且该化合物具有良好的热稳定性. 为了更好地理解结构和性能之间的关系, 对该化合物进行了第一性原理计算. 与此同时, 为了进一步探索NaBa3Si2O7F的结构新颖性, 我们还对混合碱金属和/或碱土金属硅酸盐氟化物的阴离子结构进行了比较, 结果表明孤立的[Si2O7]二聚体在上述体系中较为少见, 这一研究结果丰富了硅酸盐氟化物的结构化学.
Article PDF
Similar content being viewed by others
References
Liebau F. Structural Chemistry of Silicates: Structure, Bonding, and Classification. Berlin: Springer-Verlag, 1985
Geng Y, Zhang Z, Tegus O, et al. Microstructure, magnetic and magnetocaloric properties of Fe2−xMnxP04Si0.6 alloys. Sci China Mater, 2016, 59: 1062–1068
Wentzcovitch RM, da Silva C, Chelikowsky JR, et al. A new phase and pressure induced amorphization in silica. Phys Rev Lett, 1998, 80: 2149–2152
Xia Z, Ma C, Molokeev MS, et al. Chemical unit cosubstitution and tuning of photoluminescence in the Ca2(Al1−xMgx)(Al1−xSi1+x)O7: Eu2+ phosphor. J Am Chem Soc, 2015, 137: 12494–12497
Zhou J, Xia Z, Yang M, et al. High efficiency blue-emitting phosphor: Ce3+-doped Ca5.45Li3.55(SiO4)3O0.45F1.55 for near UV-pumped light-emitting diodes. J Mater Chem, 2012, 22: 21935
Liu KK, Zhou R, Liang YC, et al. Towards efficient and stable multi-color carbon nanoparticle phosphors: Synergy between inner polar groups and outer silica matrix. Sci China Mater, 2018, 61: 1191–1200
Wang M, Yang M, Zhao X, et al. Spinel LiMn2−xSixO4 (x<1) through Si4+ substitution as a potential cathode material for lithium-ion batteries. Sci China Mater, 2016, 59: 558–566
Li SK, Mao LB, Gao HL, et al. Bio-inspired clay nanosheets/polymer matrix/mineral nanofibers ternary composite films with optimal balance of strength and toughness. Sci China Mater, 2017, 60: 909–917
Xu Y, Shen X, Peng C, et al. Synthesis of ultra-small mordenite zeolite nanoparticles. Sci China Mater, 2018, 61: 1185–1190
Wang Z, Wang X, Tong Y, et al. Impact of structure and flow-path on in situ synthesis of AlN: Dynamic microstructural evolution of Al-AlN-Si materials. Sci China Mater, 2018, 61: 948–960
Zhang M, Wang J, Ding W, et al. Luminescence properties of M2MgSi2O7:Eu2+ (M=Ca, Sr) phosphors and their effects on yellow and blue LEDs for solid-state lighting. Opt Mater, 2007, 30: 571–578
Kong F, Jiang HL, Mao JG. La4(Si5.2Ge2.8O18)(TeO3)4 and La2(Si6O13)(TeO3)2: Intergrowth of the lanthanum(III) tellurite layer with the XO4 (X=Si/Ge) tetrahedral layer. J Solid State Chem, 2008, 181: 263–268
Du J, Corrales LR. Characterization of the structural and electronic properties of crystalline lithium silicates. J Phys Chem B, 2006, 110: 22346–22352
Shen C, Zhong C, Hou Q, et al. White LED based on CaAl2Si2O8: Eu2+ Mn2+ phosphor and CdS/ZnS quantum dots. J Phys-Conf Ser, 2011, 276: 012184
Mochizuki D, Shimojima A, Imagawa T, et al. Molecular manipulation of two- and three-dimensional silica nanostructures by alkoxysilylation of a layered silicate octosilicate and subsequent hydrolysis of alkoxy groups. J Am Chem Soc, 2005, 127: 7183–7191
Xia Z, Liu RS. Tunable blue-green color emission and energy transfer of Ca2Al3O6F:Ce3+, Tb3+ phosphors for near-UV white leds. J Phys Chem C, 2012, 116: 15604–15609
Xia Z, Zhang Y, Molokeev MS, et al. Structural and luminescence properties of yellow-emitting NaScSi2O6:Eu2+ phosphors: Eu2+ site preference analysis and generation of red emission by codoping Mn2+ for white-light-emitting diode applications. J Phys Chem C, 2013, 117: 20847–20854
Han G, Lei BH, Yang Z, et al. A fluorooxosilicophosphate with an unprecedented SiO2F4 species. Angew Chem Int Ed, 2018, 57: 9828–9832
Chen X, Zhang F, Liu L, et al. Li3 AlSiO5: the first aluminosilicate as a potential deep-ultraviolet nonlinear optical crystal with the quaternary diamond-like structure. Phys Chem Chem Phys, 2016, 18: 4362–4369
Wu H, Yu H, Pan S, et al. Cs2B4SiO9: A deep-ultraviolet nonlinear optical crystal. Angew Chem Int Ed, 2013, 52: 3406–3410
Morrison G, Latshaw AM, Spagnuolo NR, et al. Observation of intense X-ray scintillation in a family of mixed anion silicates, Cs3RESi4O10F2 (RE = Y, Eu-Lu), obtained via an enhanced flux crystal growth technique. J Am Chem Soc, 2017, 139: 14743–14748
Tang RL, Hu CL, Mao FF, et al. Ba4Bi2(Si8−xB4+xO29) (x = 0.09): a new acentric metal borosilicate as a promising nonlinear optical material. Chem Sci, 2019, 10: 837–842
Zhou Z, Qiu Y, Liang F, et al. CsSiB3O7: A beryllium-free deep-ultraviolet nonlinear optical material discovered by the combination of electron diffraction and first-principles calculations. Chem Mater, 2018, 30: 2203–2207
Zheng L, Xu J, Zhao G, et al. Bulk crystal growth and efficient diode-pumped laser performance of Yb3+:Sc2SiO5. Appl Phys B, 2008, 91: 443–445
Melcher CL, Schweitzer JS. Cerium-doped lutetium oxyorthosilicate: A fast, efficient new scintillator. IEEE Trans Nucl Sci, 1992, 39: 502–505
Ding H, Lu SC, Du GX. Surface modification of wollastonite by the mechano-activated method and its properties. Int J Miner Metall Mater, 2011, 18: 83–88
Hou X, Ding H, Liang Y, et al. Mechanism of surface hydro-phobicity modification of wollastonite powder. Mater Res Innovations, 2014, 17: 260–266
Lei BH, Yang Z, Pan S. Enhancing optical anisotropy of crystals by optimizing bonding electron distribution in anionic groups. Chem Commun, 2017, 53: 2818–2821
Viallis-Terrisse H, Nonat A, Petit JC. Zeta-potential study of calcium silicate hydrates interacting with alkaline cations. J Colloid Interface Sci, 2001, 244: 58–65
Bujdák J, Iyi N, Kaneko Y, et al. Molecular arrangement of rhodamine 6G cations in the films of layered silicates: The effect of the layer charge. Phys Chem Chem Phys, 2003, 5: 4680–4685
Ruiz-Hitzky E, Aranda P. Polymer-salt intercalation complexes in layer silicates. Adv Mater, 1990, 2: 545–547
Oh SJ, Lee DW, Ok KM. Influence of the cation size on the framework structures and space group centricities in AMo2O5(SeO3)2 (A = Sr, Pb, and Ba). Inorg Chem, 2012, 51: 5393–5399
Halasyamani PS. Asymmetric cation coordination in oxide materials: influence of lone-pair cations on the intra-octahedral distortion in d0 transition metals. Chem Mater, 2004, 16: 3586–3592
Pei HL, Wei Q, Sun L, et al. [M(1,2-dap)3][B10O13(OH)6] (M=Co, Ni): Two new borates containing [B10O13(OH)6]2-cluster units. J Clust Sci, 2018, 29: 49–55
Yang G, Wu K. Two-dimensional deep-ultraviolet beryllium-free KBe2BO3F2 family nonlinear-optical monolayer. Inorg Chem, 2018, 57: 7503–7506
Yang GS, Gong PF, Lin ZS, et al. AZn2BO3X2 (A = K, Rb, NH4; X = Cl, Br): New members of KBBF family exhibiting large SHG response and the enhancement of layer interaction by modified structures. Chem Mater, 2016, 28: 9122–9131
Yu H, Zhang W, Young J, et al. Design and synthesis of the beryllium-free deep-ultraviolet nonlinear optical material Ba3(ZnB5O10)PO4. Adv Mater, 2015, 27: 7380–7385
Xie Z, Wang Y, Cheng S, et al. Synthesis, characterization, and theoretical analysis of three new nonlinear optical materials K7MRE2B15O30 (M= Ca and Ba, RE= La and Bi). Sci China Mater, 2019, 62: 1151–1161
Yang Y, Dong X, Pan S, et al. The rubidium barium borate resulting from B7O15 fundamental building block exhibits DUV cutoff edge. Inorg Chem, 2018, 57: 13380–13385
Lee DW, Ok KM. New alkali-metal gallium selenites, AGa(SeO3)2 (A = Li, Na, K, and Cs): Effect of cation size on the framework structures and macroscopic centricities. Inorg Chem, 2013, 52: 5176–5184
Song SY, Ok KM. Modulation offramework and centricity: Cation size effect in new quaternary selenites, ASc(SeO3)2 (A = Na, K, Rb, and Cs). Inorg Chem, 2015, 54: 5032–5038
Zhang W, Yu H, Cantwell J, et al. LiNa5Mo9O30: Crystal growth, linear, and nonlinear optical properties. Chem Mater, 2016, 28: 4483–4491
Zhang JH, Hu CL, Xu X, et al. New second-order NLO materials based on polymeric borate clusters and GeO4 tetrahedra: A combined experimental and theoretical study. Inorg Chem, 2011, 50: 1973–1982
Qiao J, Xia Z, Zhang Z, et al. Near UV-pumped yellow-emitting Sr9MgLi(PO4)7:Eu2+ phosphor for white-light LEDs. Sci China Mater, 2018, 61: 985–992
Zhang X, Wu H, Yu H, et al. Ba4M(CO3)2(BO3)2 (M=Ba, Sr): two borate-carbonates synthesized by open high temperature solution method. Sci China Mater, 2019, 62: 1023–1032
Dong X, Jing Q, Shi Y, et al. Pb2 Ba3(BO3)3Cl: A material with large SHG enhancement activated by Pb-chelated BO3 groups. J Am Chem Soc, 2015, 137: 9417–9422
Xu X, Hu CL, Kong F, et al. Ca10Ge16B6O51 and Cd12Ge17B8O58: Two types of new 3D frameworks based on BO4 tetrahedra and 1D [Ge4O12]n chains. Inorg Chem, 2011, 50: 8861–8868
Wang Y, Zhang B, Yang Z, et al. Cation-tuned synthesis of fluorooxoborates: Towards optimal deep-ultraviolet nonlinear optical materials. Angew Chem Int Ed, 2018, 57: 2150–2154
Wang X, Wang Y, Zhang B, et al. CsB4O6F: A congruent-melting deep-ultraviolet nonlinear optical material by combining superior functional units. Angew Chem Int Ed, 2017, 56: 14119–14123
Shi G, Wang Y, Zhang F, et al. Finding the next deep-ultraviolet nonlinear optical material: NH4B4O6F. J Am Chem Soc, 2017, 139: 10645–10648
Zhang Z, Wang Y, Zhang B, et al. Polar fluorooxoborate, NaB4O6F: A promising material for ionic conduction and nonlinear optics. Angew Chem Int Ed, 2018, 57: 6577–6581
Wu H, Pan S, Poeppelmeier KR, et al. K3B6O10Cl: A new structure analogous to perovskite with a large second harmonic generation response and deep UV absorption edge. J Am Chem Soc, 2011, 133: 7786–7790
Zhang B, Shi G, Yang Z, et al. Fluorooxoborates: Beryllium-free deep-ultraviolet nonlinear optical materials without layered growth. Angew Chem Int Ed, 2017, 56: 3916–3919
Lin Y, Nan CW, Zhou X, et al. Preparation and characterization of long afterglow M2MgSi2O7-based (M: Ca, Sr, Ba) photoluminescent phosphors. Mater Chem Phys, 2003, 82: 860–863
Inc Madison WI. Bruker, Analytical X-ray Instruments, SAINT, version 7.60A, 2008
Dolomanov OV, Bourhis LJ, Gildea RJ, et al. OLEX2: a complete structure solution, refinement and analysis program. J Appl Crystlogr, 2009, 42: 339–341
Sheldrick GM. SHELXT-Integrated space-group and crystal-structure determination. Acta Crystlogr A Found Adv, 2015, 71: 3–8
Sheldrick GM. Crystal structure refinement with SHELXL. Acta Crystlogr C Struct Chem, 2015, 71: 3–8
Spek AL. Single-crystal structure validation with the program PLATON. J Appl Crystlogr, 2003, 36: 7–13
Kubelka P, Munk FZ. A contribution to the optics of pigments. Tech Phys, 1931, 12: 593–603
Segall MD, Lindan PJD, Probert MJ, et al. First-principles simulation: Ideas, illustrations and the CASTEP code. J Phys-Condens Matter, 2002, 14: 2717–2744
Saburi S, Kawahara A, Henmi C, et al. The refinement of the crystal structure of cuspidine. Mineral J, 1977, 8: 286–298
Kaneva E, Lacalamita M, Mesto E, et al. Structure and modeling of disorder in miserite from the Murun (Russia) and Dara-i-Pioz (Tajikistan) massifs. Phys Chem Miner, 2013, 41: 49–63
Cannillo E, Giuseppetti G, Tazzoli V. The crystal structure of leucophanite. Acta Cryst, 1967, 23: 255–259
Grice JD, Hawthorne FC. Refinement of the crystal structure of leucophanite. Can Mineral, 1989, 27: 193–197
Olsen E, Huebner JS, Dougla JAV, et al. Meteoritic amphibole. Am Mineral, 1973, 58: 869–872
Cameron M, Sueno S, Papike JJ, et al. High temperature crystal chemistry of K and Na fluor-richterites. Am Mineral, 1983, 68: 924–943
Sueno S, Matsuura S, Gibbs GV, et al. A crystal chemical study of protoanthophyllite: Orthoamphiboles with the protoamphibole structure. Phys Chem Miner, 1998, 25: 366–377
Rozhdestvenskaya IV, Mugnaioli E, Czank M, et al. Essential features of the polytypic charoite-96 structure compared to charoite-90. Mineral mag, 2018, 75: 2833–2846
Sokolova GV, Kashayev AA, Drits VA, et al. The crystal-structure of fedorite. Kristallogr, 1983, 28: 170–172
Mitchell RH, Burns PC. The structure of fedorite: A re-appraisal. Canadian Miner, 2001, 39: 769–777
Lin X, Zhang F, Pan S, et al. Ba4(BO3)3(SiO4)Ba3X (X = Cl, Br): New salt-inclusion borosilicate halides as potential deep UV nonlinear optical materials. J Mater Chem C, 2014, 2: 4257–4264
Zhen N, Wu K, Li Q, et al. Synthesis, structures, and properties of two magnesium silicate fluorides Mg5(SiO4)2F2 and Mg3SiO4F2. New J Chem, 2015, 39: 8866–8873
Leng Z, Li L, Liu Y, et al. Tunable luminescence and energy transfer properties of KSr4(BO3)3:Dy3+, Eu3+ phosphors for near-UV warm-white LEDs. J Lumin, 2016, 173: 171–176
Zhang Y, Li X, Li K, et al. Crystal-site engineering control for the reduction of Eu3+ to Eu2+ in CaYAlO4: Structure refinement and tunable emission properties. ACS Appl Mater Interfaces, 2015, 7: 2715–2725
Akella A, Keszler DA. Sr2LiSiO4F: Synthesis, structure, and Eu2+ luminescence. Chem Mater, 1995, 7: 1299–1302
Godby RW, Schlüter M, Sham LJ. Trends in self-energy operators and their corresponding exchange-correlation potentials. Phys Rev B, 1987, 36: 6497–6500
Acknowledgements
This work was supported by the National Natural Science Foundation of China (U1703132, 51872325 and 61835014), Tianshan Innovation Team Program (2018D14001), Xinjiang International Science & Technology Cooperation Program (2017E01014), the National Key Research Project (2016YFB0402104), the Science and Technology Project of Urumqi (P161010002), Xinjiang Key Research and Development Program (2016B02021), Major Program of Xinjiang Uygur Autonomous Region of China during the 13th Five-Year Plan Period (2016A02003), and West Light Foundation of the Chinese Academy of Sciences (2016-YJRC-2).
Author information
Authors and Affiliations
Contributions
Author contributions Miao Z, Yang Y and Abudouwufu T performed the experiments, data analysis, and paper writing; Wei Z and Yang Z performed the theoretical data analysis; Pan S designed the concept and supervised the experiments. All authors contributed to the general discussion.
Corresponding author
Additional information
Conflict of interest The authors declare that they have no conflict of interest.
Zhaohong Miao received her BSc degree in Shandong Normal University in 2016. She then joined Professor Shilie Pan’s research group as a master student at the University of Chinese Academy of Sciences (UCAS). Her research is currently focusing on the optical materials.
Yun Yang completed her PhD in material physics and chemistry under the supervision of Professor Shilie Pan at UCAS in 2011. She started her career as an assistant professor at Xinjiang Technical Institute of Physics & Chemistry of CAS (XTIPC, CAS) in 2007. In 2013, she was promoted to associate professor at XTIPC. In 2018, she was promoted to programs fellow and worked as a full professor at XTIPC. Her current research interest focuses on optical materials.
Shilie Pan received his BSc degree in chemistry from Zhengzhou University in 1996. He completed his PhD under the supervision of Professor Yicheng Wu (Academician) at the University of Science & Technology of China in 2002. From 2002 to 2004, he was a post-doctoral fellow at the Technical Institute of Physics & Chemistry of CAS in the laboratory of Professor Chuangtian Chen (Academician). From 2004 to 2007, he was a post-doctoral fellow at the Northwestern University in the laboratory of Professor Kenneth R. Poeppelmeier in USA. Since 2007, he has worked as a full professor at XTIPC, CAS. His current research interests include the design, synthesis, crystal growth and evaluation of new optical-electronic functional materials.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Miao, Z., Yang, Y., Wei, Z. et al. A new barium-containing alkali metal silicate fluoride NaBa3Si2O7F with deep-UV optical property. Sci. China Mater. 62, 1454–1462 (2019). https://doi.org/10.1007/s40843-019-9448-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40843-019-9448-y