Abstract
Background
Renal tubular injury, accompanied by damaging inflammation, has been identified to drive diabetic kidney disease (DKD) toward end-stage renal disease. However, it is unclear how damage-associated molecular patterns (DAMPs) activate innate immunity to mediate tubular epithelial cell (TEC) injury, which in turn causes with subsequent sterile inflammation in diabetic kidneys. High mobility group nucleosome-binding protein 1 (HMGN1) is a novel DAMP that contributes to generating the innate immune response. In this study, we focused on determining whether HMGN1 is involved in DKD progression.
Methods
Streptozotocin (STZ)-induced diabetic mice model was established. Then we downrergulated HMGN1 expression in kidney with or without HMGN1 administration. The renal dysfunction and morphological lesions in the kidneys were evaluated. The expressions of KIM-1, MCP-1, F4/80, CD68, and HMGN1/TLR4 signaling were examined in the renal tissue. In vitro, HK2 cells were exposed in the high glucose with or without HMGN1, and further pre-incubated with TAK242 was applied to elucidate the underlying mechanism.
Results
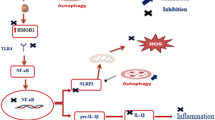
We demonstrated that HMGN1 was upregulated in the tubular epithelial cells of streptozotocin (STZ)-induced type 1 and type 2 diabetic mouse kidneys compared to controls, while being positively correlated with increased TLR4, KIM-1, and MCP-1. Down-regulation of renal HMGN1 attenuated diabetic kidney injury, decreased the TLR4, KIM-1, and MCP-1 expression levels, and reduced interstitial infiltrating macrophages. However, these phenotypes were reversed after administration of HMGN1. In HK-2 cells, HMGN1 promoted the expression of KIM-1 and MCP-1 via regulating MyD88/NF-κB pathway; inhibition of TLR4 effectively diminished the in vitro response to HMGN1.
Conclusions
Our study provides novel insight into HMGN1 signaling mechanisms that contribute to tubular sterile injury and low-grade inflammation in DKD. The study findings may help to develop new HMGN1-targeted approaches as therapy for immune-mediated kidney damage rather than as an anti-infection treatments.






Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
References
Sabanayagam C, Chee ML, Banu R, Cheng C-Y, Lim SC, Shyong Tai E, Coffman T, Wong TY (2019) Association of diabetic retinopathy and diabetic kidney disease with all-cause and cardiovascular mortality in a multiethnic Asian population. JAMA Netw Open 2:e191540
Saeedi P, Petersohn I, Salpea P, Malanda B, Karuranga S, Unwin N, Colagiuri S, Guariguata L, Motala AA, Ogurtsova K, Shaw JE, Bright D, Williams R (2019) Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res Clin Pract 157:107843
Samsu N (2021) Diabetic nephropathy: challenges in pathogenesis, diagnosis, and treatment. Biomed Res Int 8:1497449
Niewczas MA, Pavkov ME, Skupien J, Smiles A, Dom ZI, Wilson JM, Park J, Nair V, Schlafly A, Saulnier P-J, Satake E, Simeone CA, Shah H, Qiu C, Looker HC, Fiorina P, Ware CF, Sun JK, Doria A, Kretzler M, Susztak K, Duffin KL, Nelson RG, Krolewski AS (2019) A signature of circulating inflammatory proteins and development of end-stage renal disease in diabetes. Nat Med 25:805–813
Hanouneh M, Echouffo TJ, Jaar BG (2021) Recent advances in diabetic kidney disease. BMC Med 19:180
Wang J, Xiang H, Lu Y, Tao W, Guang J (2021) New progress in drugs treatment of diabetic kidney disease. Biomed Pharmacother 141:111918
Selby NM, Taal MW (2020) An updated overview of diabetic nephropathy: diagnosis, prognosis, treatment goals and latest guidelines. Diabetes Obes Metab 22(Suppl 1):3–15
La RE, Fiorina P, Astorri E, Rossetti C, Lucignani G, Fazio F, Giudici D, Castoldi R, Bianchi G, Di CV, Pozza G, Secchi A (2000) Patient survival and cardiovascular events after kidney-pancreas transplantation: comparison with kidney transplantation alone in uremic IDDM patients. Cell Transpl 9(6):929–932
Lazzaroni E, Ben NM, Loretelli C, Pastore I, Plebani L, Lunati ME, Vallone L, Bolla AM, Rossi A, Montefusco L, Ippolito E, Berra C, D’Addio F, Zuccotti GV, Fiorina P (2021) Anti-diabetic drugs and weight loss in patients with type 2 diabetes. Pharmacol Res 171:105782
Tang S, Yiu WH (2020) Innate immunity in diabetic kidney disease. Nat Rev Nephrol 16:206–222
Ichimura T, Asseldonk EJ, Humphreys BD, Gunaratnam L, Duffield JS, Bonventre JV (2008) Kidney injury molecule-1 is a phosphatidylserine receptor that confers a phagocytic phenotype on epithelial cells. J Clin Investig 118:1657–1668
Liu BC, Tang TT, Lv LL, Lan H-Y (2018) Renal tubule injury: a driving force toward chronic kidney disease. Kidney Int 93(3):568–579
Panduru NM, Sandholm N, Forsblom C, Saraheimo M, Dahlström EH, Thorn LM, Gordin D, Tolonen N, Wadén J, Harjutsalo V, Bierhaus A, Humpert PM (2015) Kidney injury molecule-1 and the loss of kidney function in diabetic nephropathy: a likely causal link in patients with type 1 diabetes. Diabetes Care 38(6):1130–1137
Tuttle KR, Alicic RZ (2021) Glycemic variability and KIM-1-induced inflammation in the diabetic kidney. Diabetes 70:1617–1619
Zhao X, Chen X, Zhang Y, George J, Cobbs A, Wang G, Li L, Emmett N (2019) Kidney injury molecule-1 is upregulated in renal lipotoxicity and mediates palmitate-induced tubular cell injury and inflammatory response. Int J Mol Sci 20:3406
Banba N, Nakamura T, Matsumura M, Kuroda H, Hattori Y, Kasai K (2000) Possible relationship of monocyte chemoattractant protein-1 with diabetic nephropathy. Kidney Int 58:684–690
Brennan E, Kantharidis P, Cooper ME, Godson C (2021) Pro-resolving lipid mediators: regulators of inflammation, metabolism and kidney function. Nat Rev Nephrol 17:725–739
Skyler JS, Bakris GL, Bonifacio E, Darsow T, Eckel RH, Groop L, Groop P-H, Handelsman Y, Insel RA, Mathieu C, McElvaine AT, Palmer JP, Pugliese A, Schatz DA, Sosenko JM, Wilding JPH, Ratner RE (2017) Differentiation of diabetes by pathophysiology, natural history, and prognosis. Diabetes 66:241–255
Chen GY, Nunez G (2010) Sterile inflammation: sensing and reacting to damage. Nat Rev Immunol 10:826–837
Perez-Morales RE, Del PM, Valdivielso JM, Pérez-Morales RE, Del Pino MD, Valdivielso JM, Ortiz A, Mora-Fernández C, Navarro-González JF (2019) Inflammation in diabetic kidney disease. Nephron 143:12–16
Tang SCW, Yiu WH (2020) Innate immunity in diabetic kidney disease. Nat Rev Nephrol 16(4):206–222
Murao A, Aziz M, Wang H, Brenner M, Wang P (2021) Release mechanisms of major DAMPs. Apoptosis 26(3–4):152–162
Zhou ZF, Jiang L, Zhao Q, Yu W, Zhou J, Chen Q-K, Lv J-L (2020) Roles of pattern recognition receptors in diabetic nephropathy. J Zhejiang Univ Sci B 21:192–203
Wada J, Makino H (2016) Innate immunity in diabetes and diabetic nephropathy. Nat Rev Nephrol 12(1):13–26. https://doi.org/10.1038/nrneph.2015.175. (Epub 2015 Nov 16 PMID: 26568190)
Gong T, Liu L, Jiang W, Zhou R (2020) DAMP-sensing receptors in sterile inflammation and inflammatory diseases. Nat Rev Immunol 20(2):95–112
Yang D, Tewary P, de la Rosa G, Wei F, Oppenheim JJ (2010) The alarmin functions of high-mobility group proteins. Biochim Biophys Acta (BBA) Gene Regul Mech 1799:157–163
Alam MM, Yang D, Trivett A, Meyer TJ, Oppenheim JJ (2018) HMGN1 and R848 synergistically activate dendritic cells using multiple signaling pathways. Front Immunol 9:2982
Yang D, Postnikov YV, Li Y, Tewary P, de la Rosa G, Wei F, Klinman D, Gioannini T, Weiss JP, Furusawa T, Bustin M, Oppenheim JJ (2012) High-mobility group nucleosome-binding protein 1 acts as an alarmin and is critical for lipopolysaccharide-induced immune responses. J Exp Med 209:157–171
Yu J, Da J, Dong R, Yi S, Yingjie N, Fuxun Y, Li Z, Yan Z (2018) IGF-1R inhibitor ameliorates diabetic nephropathy with suppressed HMGN1/TLR4 pathway. Endocr Metab Immune Disord Drug Targets 18:241–250
Yu J, Dong R, Da J, Li J, Yu F, Zha Y (2019) High-mobility group nucleosome-binding protein 1 mediates renal fibrosis correlating with macrophages accumulation and epithelial-to-mesenchymal transition in diabetic nephropathy mice model. Kidney Blood Press Res 44(3):331–343
Mao Y, Yu J, Da J, Yu F, Zha Y (2023) Acteoside alleviates UUO-induced inflammation and fibrosis by regulating the HMGN1/TLR4/TREM1 signaling pathway. PeerJ 11:e14765
Yang Z, Han Z, Alam MM, Oppenheim JJ (2018) High-mobility group nucleosome binding domain 1 (HMGN1) functions as a Th1-polarizing alarmin. Semin Immunol 38:49–53
Arts RJW, Yang D, Joosten LAB, van der Meer JWM, Oppenheim JJ, Netea MG, Cheng S-C (2018) High-mobility group nucleosome-binding protein 1 as endogenous ligand induces innate immune tolerance in a TLR4-sirtuin-1 dependent manner in human blood peripheral mononuclear cells. Front Immunol 9:526
Leemans JC, Kors L, Anders HJ, Florquin S (2014) Pattern recognition receptors and the inflammasome in kidney disease. Nat Rev Nephrol 10(7):398–414
Anders HJ, Banas B, Schlöndorff D (2004) Signaling danger: toll-like receptors and their potential roles in kidney disease. J Am Soc Nephrol 15(4):854–867
Arumugam TV, Okun E, Tang SC, Thundyil JS, Taylor SM, Woodruff TM (2009) Toll-like receptors in ischemia-reperfusion injury. Shock 32(1):4–16
Lin M, Yiu WH, Wu HJ, Chan LYY, Leung JCK, Au WS, Chan KW, Lai KN, Tang SCW (2012) Toll-like receptor 4 promotes tubular inflammation in diabetic nephropathy. J Am Soc Nephrol 23(1):86–102
Lin M, Yiu WH, Li RX, Wu HJ, Wong DWL, Chan LYY, Leung JCK, Lai KN, Tang SCW (2013) The TLR4 antagonist CRX-526 protects against advanced diabetic nephropathy. Kidney Int 83(5):887–900
Mudaliar H, Pollock C, Komala MG, Chadban S, Wu H, Panchapakesan U (2013) The role of Toll-like receptor proteins (TLR) 2 and 4 in mediating inflammation in proximal tubules. Am J Physiol Renal Physiol 305(2):F143–F154
Campbell MT, Hile KL, Zhang H, Asanuma H, Vanderbrink BA, Rink RR, Meldrum KK (2011) Toll-like receptor 4: a novel signaling pathway during renal fibrogenesis. J Surg Res 168(1):e61–e69
Wolfs TG, Buurman WA, van Schadewijk A, de Vries B, Daemen MARC, Hiemstra PS, van’t Veer C (2002) In vivo expression of Toll-like receptor 2 and 4 by renal epithelial cells: IFN-gamma and TNF-alpha mediated up-regulation during inflammation. J Immunol 168(3):1286–1293
Mori Y, Ajay AK, Chang J-H, Mou S, Zhao H, Kishi S, Li J, Brooks CR, Xiao S, Woo H-M, Sabbisetti VS, Palmer SC, Galichon P, Li L, Henderson JM, Kuchroo VK, Hawkins J, Ichimura T, Bonventre JV (2021) KIM-1 mediates fatty acid uptake by renal tubular cells to promote progressive diabetic kidney disease. Cell Metab 33:1042–1061
Scurt FG, Menne J, Brandt S, Bernhardt A, Mertens PR, Haller H, Chatzikyrkou C (2022) Monocyte chemoattractant protein-1 predicts the development of diabetic nephropathy. Diabetes Metab Res Rev 38:e3497
Yiu WH, Lin M, Tang SC (2014) Toll-like receptor activation: from renal inflammation to fibrosis. Kidney Int Suppl 4:20–25
Satoh T, Akira S (2016) Toll-like receptor signaling and its inducible proteins. Microbiol Spectr. https://doi.org/10.1128/microbiolspec.MCHD-0040-2016
Tam FW, Sanders JS, George A, Hammad T, Miller C, Dougan T, Cook HT, Kallenberg CG, Gaskin G, Levy JB, Pusey CD (2004) Urinary monocyte chemoattractant protein-1 (MCP-1) is a marker of active renal vasculitis. Nephrol Dial Transpl 19(11):2761–2768. https://doi.org/10.1093/ndt/gfh487. (Epub 2004 Sep 7. PMID: 15353578)
Alicic RZ, Rooney MT, Tuttle KR (2017) Diabetic kidney disease: challenges, progress, and possibilities. Clin J Am Soc Nephrol 12(12):2032–2045
Kawamoto T, Li M, Kitazaki T, Iizawa Y, Kimura H (2008) TAK-242 selectively suppresses Toll-like receptor 4-signaling mediated by the intracellular domain. Eur J Pharmacol 584:40–48
Acknowledgements
We thank the NHC Key Laboratory of Pulmonary Immunological Disease and Research Laboratory Center in Guizhou Provincial People’s Hospital for providing the experimental platform for this study. This work was supported by grants from Guizhou Provincial Science and Technology Projects (QKHJC-ZK[2022] General 265; QKHJC[2020]1Y304; QKHPTRC2018-5636-2; QKHCG2023-ZD010), Science and Technology Fund project of Guizhou Provincial Health Commission in 2021 (gzwkj2021-136), Youth Fund of Guizhou Provincial People’s Hospital in 2021 (GZSYQN[2021]12), Guizhou Provincial Administration of Traditional Chinese Medicine and ethnic Medicine Science and Technology Project (QZYY-2023-018), National Natural Science Foundation of China (82360148), the Special Fund for Basic Scientific Research Operating of Central Public Welfare Research Institutes, Chinese Academy of Medical Sciences (2019PT320003).
Author information
Authors and Affiliations
Contributions
JYu analyzed the data, edited the manuscript, and completed the submission. YZ and JD conceived and supervised the study. JYu performed most experiments. FY reviewed and edited the manuscript. YZ and JY provided funding acquisition and methodology, respectively. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflicts of interest in this work.
Ethics Statement
All animal experiments with mice were performed in compliance with the principles and procedures outlined in the National Institutes of Health Guide for the Humane Care and Use of Animals and were approved by the institutional committees of the Animal Research Committee and Animal Ethics Committee of the Guizhou Provincial People’s Hospital.
Informed consent
Informed consent is not required in our animal study.
Research involving human participants and/or animals
This study was approved by the Ethics Committee of Guizhou Provincial People’s Hospital (EC Review Animal 2022-012), and all experiments were conducted in accordance with the approved guidelines.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yu, J., Da, J., Yu, F. et al. HMGN1 down-regulation in the diabetic kidney attenuates tubular cells injury and protects against renal inflammation via suppressing MCP-1 and KIM-1 expression through TLR4. J Endocrinol Invest 47, 1015–1027 (2024). https://doi.org/10.1007/s40618-023-02292-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-023-02292-0