Abstract
Background
Circulating metabolites (CM) play a pivotal role in our overall health, yet the current evidence concerning the involvement of diverse CM in benign prostatic hyperplasia (BPH) remains limited. Mendelian randomization (MR) offers a promising avenue to explore the potential impact of CM on BPH.
Methods
In a forward MR analysis, a cohort of 249 circulating metabolites was employed as exposures to investigate their potential associations with BPH risk. Conversely, in a reverse MR analysis, BPH was employed as an exposure to assess its effects on CM.
Results
The forward MR analysis discerned a linkage between six metabolites and BPH, with careful consideration to excluding heterogeneity and pleiotropy. Subsequently, the reverse MR analysis unveiled that nine metabolic compounds, mainly comprising phospholipids and triglycerides, potentially exhibit elevated levels in BPH patients.
Conclusion
Bidirectional MR analysis furnishes genetic insight into the interplay between CM and BPH. The prominence of lipids and triglycerides emerges as significant factors intricately linked to BPH risk.
Similar content being viewed by others
Introduction
Benign prostatic hyperplasia (BPH): Benign prostatic hyperplasia (BPH) represents the foremost benign ailment catalyzing voiding abnormalities in middle-aged and elderly males. The incidence of BPH surges substantially, reaching up to 50%, in men aged over 60 years [1]. BPH emerges as a quintessential ailment of the aging process. Notably, with the advancing male age, the incidence and prevalence of BPH escalate concomitantly. Despite a plethora of scholarly investigations, the exact etiological basis remains a subject of controversy. Various theories, spanning genetics, androgens, hormones, cytokines, chemokines, and the pivotal role of stem cells, have been posited as potential determinants driving disease progression. These factors are widely acknowledged as pivotal contributors fostering the advancement of the condition. Nevertheless, thus far, a consensus concerning the etiology of BPH remains elusive [2]. The escalating number of patients seeking treatment for BPH-associated lower urinary tract symptoms (LUTS), coupled with the exponential rise in healthcare expenditures, underscores the pressing need for efficacious therapeutic interventions [3]. Hence, a proactive exploration into the etiological underpinnings and influential factors governing BPH assumes paramount significance in the realm of its management. Metabolomics analysis revealed that the specific metabolites were related to the disease progression of BPH. Prostate diseases can cause metabolic abnormalities, thus clarifying the relationship between various metabolites and prostate diseases is helpful to identify benign and malignant prostate diseases, so as to reduce the excessive treatments and economic burden in society and improve the living quality of patients [4]. Notably, some studies have demonstrated that serum metabolites become potential biomarkers of BPH owing to their superior sensitivity and specificity in identifying BPH [5]. Therefore, exploring the relationship between metabolites and BPH is helpful for the diagnosis and treatment of BPH. Mendelian randomization provides a pathway to explore the bidirectional relationship between circulating metabolites (CM) and BPH from the perspective of genetic exposure.
Mendelian randomization (MR): MR constitutes an epidemiological approach that capitalizes on genetic variants as instrumental variables to serve as surrogates for the variables of interest, thereby facilitating the evaluation of the causal repercussions of exposures on specific outcomes[6]. The inherent random assignment of single nucleotide polymorphisms (SNPs) confers resistance against confounding influences. Importantly, genetic variations remain immune to the development of subsequent outcome traits, mitigating the specter of reverse causality bias [7]. Augmenting its utility, genetic instrumental variables encapsulate lifelong exposures, rendering MR an adept tool for scrutinizing age-associated phenomena. In the contemporary landscape, bolstered by high-throughput metabolomics, the concurrent measurement of numerous circulating metabolite levels has become feasible. Recent epochs have witnessed a plethora of genome-wide association studies (GWASs) delving into the nexus between circulating metabolic biomarkers and SNPs [8,9,10]. In the present study, leveraging the largest human genomic dataset available to date, we employ the bidirectional MR methodology to discern the causal nexus between circulating metabolites and the susceptibility to the prevalent BPH characteristic among the elderly demographic.
Materials and methods
Research design description
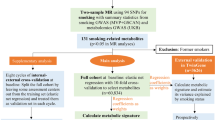
Figure 1 outlines the key steps of the bidirectional Mendelian randomization (MR) study investigating the interplay between CM and BPH. This study involves two MR analyses, using summary statistical data from genome-wide association studies (GWAS), to unveil potential associations between CM and BPH. In the forward MR analysis, CM is treated as the exposure variable and BPH as the outcome. In the reverse MR analysis, BPH is considered the exposure variable and CM the outcome. The fundamental MR assumptions underpinning the study are illustrated in Fig. 1. Given that this study relies on publicly available data, ethical approval is not required.
MR tool variable selection
The instrumental variables (IVs) for MR analysis were extracted from two distinct GWAS summary results. Initially, a genome-wide significance threshold (P < 5 × 10−8) was employed [11]. Subsequently, SNP independence was evaluated based on pairwise linkage disequilibrium. SNPs demonstrating an r2 > 0.001 within a 10,000 kb window were excluded, effectively addressing associations with multiple SNPs and SNPs exhibiting higher P values [12]. Linkage disequilibrium signifies the non-random association between alleles at distinct loci. In essence, if two genes are not inherited independently, some level of linkage would be evident. The parameter r2 ranges from 0 to 1, where r2 = 1 signifies complete linkage disequilibrium, and r2 = 0 denotes complete linkage equilibrium, indicating random allocation of the two SNPs. The linkage disequilibrium region's length is denoted by ‘kb’. The threshold r2 = 0.001 and a 10,000 kb window are used to exclude SNPs with r2 exceeding 0.001 within 10,000 kb. Additionally, F-statistics were computed to gauge the strength of individual SNPs. SNPs with F-statistic values exceeding 10 were deemed sufficiently robust to mitigate potential bias.
BPH data source and tool variable selection
The BPH data originates from the MRC IEU UK Biobank GWAS pipeline version 2 (available at: https://data.bris.ac.uk/data/dataset/pnoat8cxo0u52p6ynfaekeigi). This dataset encompasses a population primarily diagnosed with BPH. The GWAS data is leveraged to identify SNPs associated with BPH, which are subsequently selected as IVs (refer to Supplementary Table 1).
CM data source and tool variable selection
A summary-level GWAS dataset encompassing 249 CM was acquired from the UK Biobank (unpublished data, accessible via the MRC IEU OpenGWAS database) (see Supplementary Table 1 for details).
Table 1 Detailed information of included data sources
MR statistical analysis
SNPs pertaining to both CM and BPH were employed in subsequent forward and reverse MR analyses. The random-effects inverse variance weighting (IVW) method, encompassing the core MR assumptions, constituted the primary statistical approach for estimating potential bidirectional causal associations between CM and BPH [13]. When multiple IVs are available, the IVW method stands as the most robust, as it accounts for variant specificity and causal estimation heterogeneity. The IVW method further encompasses sensitivity analyses, including simple mode, weighted mode, weighted median, and MR Egger regression, to assess research findings' robustness [14]. Should IVs influence outcomes through alternate pathways, indicative of potential pleiotropy, the causal estimation via IVW might incur bias. To assess pleiotropy, MR Egger was utilized. A P-value exceeding 0.05 in MR Egger signifies the absence of level pleiotropy. Heterogeneity testing using MR heterogeneity was conducted to identify SNP-induced heterogeneity. Should heterogeneity be detected, a random-effects model was employed; otherwise, the fixed-effects model was assumed by default. Single SNPs were sequentially eliminated from MR analyses to assess their collective impact [15]. The Twosamplemr (v.0.5.6) within the R package (v.4.3.0) facilitated major statistical analysis and graphical representation [16]. Odds ratio (OR) and the accompanying 95% confidence interval (CI) gauged the extent of risk alteration for each additional standard deviation of exposure factors. Statistical significance was set at P < 0.05 [17].
Results
Forward MR
The IVW analysis reveals a significant genetic correlation (P < 0.05) between six CM and BPH (Table 2, Figs. 2, 3). No substantial evidence of horizontal pleiotropy among SNPs is observed (Table 3, P > 0.05). Combining IVW and MR Egger outcomes, no substantial heterogeneity is detected in relation to the association (Table 4, all Cochran’s Q P values > 0.05).
Heatmap illustrating significant correlations in forward MR. The figure showcases varying P values in distinct blocks, color-coded from red to blue denoting ascending P values. The X-axis delineates three separate outcomes: MR Egger, weighted median, and inverse variance weighted. The Y-axis signifies different circulating metabolites
Forest plot depicting significant correlations in forward MR. The Y-axis represents diverse circulating metabolites, while the X-axis indicates OR values and corresponding 95% CIs. Various shapes in the graph symbolize distinct metabolite ORs, with the horizontal line denoting the range of the 95% CI
Reverse MR
The reverse MR analysis demonstrates disparities in 92 metabolites within BPH patients, accounting for heterogeneity and pleiotropy. As exposure escalates, the risk associated with certain metabolites increases, including the Phospholipids to total lipids ratio in IDL, Phospholipids to total lipids ratio in very small VLDL, Phospholipids to total lipids ratio in small HDL, Triglycerides to total lipids ratio in medium LDL, Phospholipids to total lipids ratio in very large HDL, Triglycerides to total lipids ratio in large LDL, Phenylalanine, Average diameter for HDL particles, and Triglycerides to total lipids ratio in IDL. Conversely, the risk of 83 other metabolites diminishes as exposure intensifies (refer to Figs. 4, 5 and Supplementary Table 2).
Heatmap displaying significant correlations in reverse MR. The illustration employs distinct colors to signify varying P values within individual blocks, transitioning from red to blue to denote ascending P values. The X-axis encompasses three distinct outcomes: MR Egger, weighted median, and inverse variance weighted. The Y-axis delineates diverse circulating metabolites
Forest plot illustrating significant correlations in reverse MR. The Y-axis represents distinct circulating metabolites, whereas the X-axis denotes OR values along with corresponding 95% CIs. Distinct shapes within the graph symbolize distinct metabolite ORs, with the horizontal line marking the range of the 95% CI
Discussion
Benign prostatic hyperplasia (BPH) stands as a prevalent health concern among males, influenced by a complex interplay of biological and environmental factors. The central objective of this study was to investigate the interrelation between circulating metabolites (CM) and BPH via a bidirectional Mendelian randomization (MR) analysis, thereby unveiling potential pathogenic mechanisms. This bidirectional MR investigation has unearthed a discernible association between BPH and CM.
The forward MR analysis divulges six metabolites within the CM realm that manifest associations with BPH, while meticulously addressing issues of heterogeneity and pleiotropy. This lends credence to the genetic impact of these metabolites upon BPH. Notably, elevated exposure to albumin, isoleucine, esters in medium HDL, cholesterol to total lipids ratio in medium LDL, phospholipids to total lipids ratio in small VLDL, and total concentration of branched-chain amino acids (leucine + isoleucine + valine) escalates the risk of BPH, thereby underscoring their potential as pathogenic factors for BPH. The prominence of lipids, amino acids, and albumin in providing requisite nutrients for BPH cell proliferation carries implications for BPH prevention, such as dietary lipid reduction. A study involving 307 men indicated that dyslipidemia and obesity act as BPH risk factors [18]. Obesity, a facet of the metabolic syndrome, is correlated with prostate volume, with the larger prostate size and heightened symptoms often observed among obese individuals [19].
Conversely, the reverse MR analysis highlights disparities in 92 metabolites among BPH patients, possibly signifying biological alterations within the bodies of these patients and reinforcing the nexus between metabolic dysregulation and BPH. These insights hold potential to identify novel avenues and strategies for diagnosing and treating metabolic ailments in the future. Of note, this reverse MR scrutiny underscores that nine key metabolic entities, chiefly phospholipids and triglycerides, heighten the risk in BPH patients, a discovery that could potentially evolve into a supplementary diagnostic criterion for BPH. An animal study has previously suggested a correlation between BPH and elevated triglyceride levels [20]. Additionally, a retrospective clinical study found a significant rise in prostate volume and prostate-specific antigen levels when triglyceride levels exceeded 150 mg/dl [21].
The findings of this study underscore that dyslipidemia constitutes a risk factor for BPH, potentially mediated through genetic influences. Lipids and triglycerides, among other factors, could be genetically intertwined with BPH progression. While the study's findings provide initial insights into the interplay between CM and BPH, a comprehensive understanding of the underlying mechanisms necessitates further investigation. CM might impact BPH development through various pathways including inflammation and lipid metabolism. In the realm of clinical practice, these findings could offer novel perspectives for BPH prevention and treatment, with the potential utilization of CM as biomarkers to assess patient risk and decipher associated biological mechanisms.
Distinct from conventional observational studies, the chief strength of this inquiry lies in its MR-derived causal estimation, effectively circumventing reverse causality and confounding bias. Furthermore, the utilization of extensive and robust GWAS data enhances the precision of outcomes. However, certain limitations intrinsic to this research cannot be disregarded. Notably, the generalizability of outcomes may be constrained due to the predominantly European population used in MR analysis. Subsequent research endeavors could be enriched through larger sample sizes and comprehensive metabolomic analyses, enabling a more nuanced assessment of CM's role in BPH development. As our comprehension matures, the elucidation of the specific mechanisms underpinning CM's action on BPH remains a pertinent avenue for exploration.
Availability of data and materials
Upon request, data can be obtained from supplementary documents or corresponding authors.
References
Ding K, Tang R, Yu J (2022) Recommendations for the management of patients with benign prostatic hyperplasia in the context of the COVID-19 pandemic: a retrospective study of 314 cases. Biomed Res Int 19:5739574. https://doi.org/10.1155/2022/5739574
Devlin CM, Simms MS, Maitland NJ (2021) Benign prostatic hyperplasia—what do we know? BJU Int 127:389–399. https://doi.org/10.1111/bju.15229
Robert G, De La Taille A, Descazeaud A (2018) Données épidémiologiques en rapport avec la prise en charge de l’HBP [Epidemiology of benign prostatic hyperplasia]. Prog Urol 28:803–812. https://doi.org/10.1016/j.purol.2018.08.005 (French)
Bansal N, Kumar M, Sankhwar SN et al (2023) Evaluation of prostate cancer tissue metabolomics: would clinics utilise it for diagnosis? Expert Rev Mol Med 7:e26. https://doi.org/10.1017/erm.2023.22
He J, Han Z, Luo W et al (2023) Serum organic acid metabolites can be used as potential biomarkers to identify prostatitis, benign prostatic hyperplasia, and prostate cancer. Front Immunol 4:998447. https://doi.org/10.3389/fimmu.2022.998447
Davey Smith G, Hemani G (2014) Mendelian randomization: genetic anchors for causal inference in epidemiological studies. Hum Mol Genet 23:89–98. https://doi.org/10.1093/hmg/ddu328
Sanderson E, Smith GD, Windmeijer F et al (2020) Corrigendum to: An examination of multivariable Mendelian randomization in the single-sample and two-sample summary data settings. Int J Epidemiol 49:1057. https://doi.org/10.1093/ije/dyaa101.Erratumfor:IntJEpidemiol.2019Jun1;48(3):713-727.PMID:32529219;PMCID:PMC7394940
Shin SY, Fauman EB, Petersen AK et al (2014) An atlas of genetic influences on human blood metabolites. Nat Genet 46:543–550. https://doi.org/10.1038/ng.2982
Roederer M, Quaye L, Mangino M et al (2015) The genetic architecture of the human immune system: a bioresource for autoimmunity and disease pathogenesis. Cell 161:387–403. https://doi.org/10.1016/j.cell.2015.02.046
Kettunen J, Demirkan A, Wurtz P et al (2016) Genome-wide study for circulating metabolites identifies 62 loci and reveals novel systemic effects of LPA. Nat Commun 7:11122. https://doi.org/10.1038/ncomms11122
Wang YB, Yang L, Deng YQ et al (2022) Causal relationship between obesity, lifestyle factors and risk of benign prostatic hyperplasia: a univariable and multivariable Mendelian randomization study. J Transl Med 20:495. https://doi.org/10.1186/s12967-022-03722-y
Lee YH (2020) Overview of Mendelian randomization analysis. J Rheum Dis 27:241–246. https://doi.org/10.4078/jrd.2020.27.4.241
Huang D, Lin S, He J et al (2022) Association between COVID-19 and telomere length: a bidirectional Mendelian randomization study. J Med Virol 11:5345–5353. https://doi.org/10.1002/jmv.28008
Burgess S, Davey Smith G, Davies NM et al (2020) Guidelines for performing Mendelian randomization investigations. Wellcome Open Res 28:186. https://doi.org/10.12688/wellcomeopenres.15555.2
Yang M, Wan X, Zheng H et al (2023) No evidence of a genetic causal relationship between ankylosing spondylitis and gut microbiota: a two-sample Mendelian randomization study. Nutrients 15:1057. https://doi.org/10.3390/nu15041057
Hemani G, Zheng J, Elsworth B et al (2018) The MR-base platform supports systematic causal inference across the human phenome. Elife 7:1–29. https://doi.org/10.7554/eLife.34408
Lu H, Wu PF, Zhang W et al (2021) Circulating interleukins and risk of multiple sclerosis: a Mendelian randomization study. Front Immunol 15:647588. https://doi.org/10.3389/fimmu.2021.647588
Hammarsten J, Högstedt B (2001) Hyperinsulinaemia as a risk factor for developing benign prostatic hyperplasia. Eur Urol 39:151–158. https://doi.org/10.1159/000052430
Moudi E, Akbarzadeh-Pasha A (2017) Comparative analysis of resected prostate weight in diabetic and non-diabetic benign prostatic hyperplasia Patients. Caspian J Intern Med 8:99–103. https://doi.org/10.22088/cjim.8.2.99
Chen IH, Tong YC, Cheng JT (2012) Metabolic syndrome decreases tissue perfusion and induces glandular hyperplasia in the fructose-fed rat prostate. Neurourol Urodyn 31:600–604. https://doi.org/10.1002/nau.21192
Byun HK, Sung YH, Kim W et al (2012) Relationships between prostate-specific antigen, prostate volume, and components of metabolic syndrome in healthy Korean men. Korean J Urol 53:774–778. https://doi.org/10.4111/kju.2012.53.11.774
Funding
The authors received funding, staff, and equipment support for the following research projects: Fundamental Research Ability Improvement Project for Young and Middle-aged Teachers in Guangxi Universities (Natural Science), Agreement No. 2022KY0300; Innovation Project of Guangxi Graduate Education of GXUCM, Agreement No. YCBXJ2023040; Administration of Traditional Chinese Medicine of Guangxi Zhuang Autonomous Region Self-funded Scientific Research Project (Natural Science), Agreement No. GXZYZ20210346; Health Commission of Guangxi Zhuang Autonomous Region self-funded scientific research project (Youth Fund), Agreement No. Z20211659; Natural Science Research Project of Guangxi University of Traditional Chinese Medicine (Youth Fund), Agreement No. 2021QN029. Guangxi Science and Technology Base and Talent Project, Agreement No. AD22035869; The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
ZCZ and HJG conceived and designed the study. BTT, CJ. Shang, and MZ conducted data analysis. ZSL, CJS, and YWY carried out data collation. ZCZ, YC, and SP. Huang wrote the paper. PJ, YWY, and SPH reviewed and edited the manuscript. All authors approved the final version of the article. ZCZ, YC, YWY, SPH, and PJ contributed equally to this work and are co-first authors.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval and consent to participate
This study does not involve ethics approval.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, Zc., Huang, Sp., Lin, Zs. et al. The relationship between circulating metabolites and prostate hyperplasia: a Mendelian randomization study. Aging Clin Exp Res 36, 10 (2024). https://doi.org/10.1007/s40520-023-02669-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s40520-023-02669-4