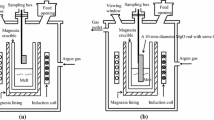
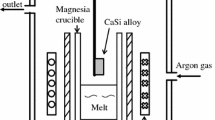
Abstract
The effect of Ca treatment on the modification of inclusions in Al-killed GCr18Mo bearing steel was studied and a thermodynamic and kinetic model was established. The experimental results showed that oxygen content in the steel could be reduced from 120 μg/g to the range 5.5–21.6 μg/g by Al-Ca complex deoxidization at 1 873 K. An appropriate increase in Ca/Al mass ratio is beneficial to reduce the total oxygen content in steel. When the content of dissolved aluminum was in the range of 0.03%–0.3%, with the increase of Ca/Al mass ratio from 0.8–1.6 to 2.4–3.2, the number of inclusions per unit area significantly reduced. In addition, the main types of inclusions were modified from large-sized Al2O3 clusters to plastic or semi-plastic liquid phase calcium aluminates. The experimental results matched well with the thermodynamic analysis.













Similar content being viewed by others
References
Holappa L (2010) On physico-chemical and technical limits in clean steel production. Steel Res Int 81(10):869–874
Yang W, Zhang L, Wang X et al (2013) Characteristics of inclusions in low carbon Al-killed steel during ladle furnace refining and calcium treatment. ISIJ Int 53(8):1401–1410
Verma N (2010) Modification of spinel inclusions by calcium in liquid steel. Iron Steel Technol 7(1):189–197
Carl B, Gilles L, Hoang L (1997) Development of an integrated method for fully characterizing multiphase inclusions and its application to calcium-treated steels. Mater Charact 38(1):25–37
Yang SF (2012) Formation and modification of MgO·Al2O3-based inclusions in alloy steels. Metall Mater Trans B 43(4):731–750
Ma W, Bao Y, Wang M et al (2014) Effect of Mg and Ca treatment on behavior and particle size of inclusions in bearing steels. ISIJ Int 54(3):536–542
Huang XH (1981) Principles of iron and steel metallurgy. Metallurgical Industry Press, Beijing
Song B, Han Q (1998) Equilibrium of calcium vapor with liquid iron and the interaction of third elements. Metall Mater Trans B 29(2):415–420
Itoh H, Hino M, Banya S (1997) Deoxidation equilibrium of calcium in liquid iron. Tetsu to-Hagane 83(11):695–700
Suito H, Inoue R (1996) Thermodynamics on control of inclusions composition in ultra-clean steels. Trans Iron Steel Inst Jpn 36(5):528–536
Chen JX (2010) Commonly used chart data handbook for steelmaking. Metallurgical Industry Press, Beijing
Taguchi K, Hideki ON, Usui T et al (2005) Complex deoxidation equilibria of molten iron by aluminum and calcium. ISIJ Int 45(11):1572–1576
Park JH, Todoroki H (2010) Control of MgO·Al2O3 spinel inclusions in stainless steels. ISIJ Int 50(10):1333–1346
Hallstedl B (1990) Assessment of the CaO-Al2O3 system. J Am Ceram Soc 73(1):15–23
Won-Gap S, Han WH, Jeong-Sik K et al (2007) Deoxidation equilibria among Mg, Al and O in liquid iron in the presence of MgO·Al2O3 spinel. ISIJ Int 43(2):201–208
Bielefeldt WV, Vilela ACF (2015) Study of inclusions in high sulfur, Al-killed Ca-treated steel via experiments and thermodynamic calculations. Steel Res Int 86(4):375–385
Murray JD, Johnson RF (1963) Effect of inclusions on the properties of 1%C-Cr bearing steels. J Iron Steel Inst London Spec Rep No 77: 104–109
Ning M, Fu JC, Zheng JH (2005) Study on inclusions modification in steel by weeding Ca-Si wire (Ca-treatment). In: CSM 2005 annual meeting proceedings, pp 246–251
Ye G, Jönsson P, Lund T (1996) Thermodynamics and kinetics of the modification of Al2O3 inclusions. ISIJ Int 36(S):S105–S108
Parra R, Allibert M (1999) Metal-slag reaction through a solid interfacial layer. Can Metall Q 38(1):11–21
Park JH, Lee SB, Kim DS (2005) Inclusion control of ferritic stainless steel by aluminum deoxidation and calcium treatment. Metall Mater Trans B 36(1):67–73
Acknowledgements
This study was financially supported by the Shanghai University and the Joint Funds of the National Natural Science Foundation of China (Grant No. U1560202).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zheng, HY., Guo, SQ., Qiao, MR. et al. Study on the modification of inclusions by Ca treatment in GCr18Mo bearing steel. Adv. Manuf. 7, 438–447 (2019). https://doi.org/10.1007/s40436-019-00266-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40436-019-00266-1