Abstract
Introduction
Next-generation sequencing (NGS) enables simultaneous detection of actionable somatic variants and estimation of genomic signatures such as tumor mutational burden (TMB) or microsatellite instability (MSI) status, which empowers therapeutic decisions in clinical oncology.
Objective
Our retrospective study investigated the clinical performance of somatic variant detection in paired tissue and blood samples using a large targeted gene panel, the OncoScreen Plus, which interrogates 520 cancer-related genes.
Methods
We analyzed sequencing data derived from paired tissue and blood samples of 3005 patients spanning 20 solid tumor types, including lung (n = 1971), gastrointestinal (n = 625), breast (n = 120) and gynecological (n = 110), genitourinary (n = 38), and other cancers (n = 141).
Results
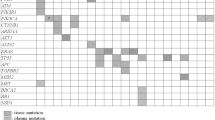
Across tumor types, the OncoScreen Plus panel achieved a high tissue detection rate, with an average of 97.9%. The average plasma detection rate was 72.2%, with an average tissue concordance rate of 36.6%. Considering all variant types, the plasma assay yielded an average sensitivity/true positive rate of 45.7%, with a positive predictive value of 64.7% relative to tissue assay. Pearson correlation analysis revealed a strong correlation in TMB estimated from blood and tissue samples (correlation coefficient 0.845, R2 = 0.756). MSI-high status was identified in five tumor types, including endometrial cancer (28.6%), colorectal cancer (2.5%), ovarian cancer (2.0%), gastric cancer (1.5%), and lung adenocarcinoma (0.2%).
Conclusion
Paired tumor and blood samples from a large cohort of patients spanning 20 tumor types demonstrated that the OncoScreen Plus is a reliable pan-cancer panel for the accurate detection of somatic variants and genomic signatures that could guide individualized treatment strategies to improve the care of patients with advanced cancer.




Similar content being viewed by others
References
Malone ER, Oliva M, Sabatini PJB, Stockley TL, Siu LL. Molecular profiling for precision cancer therapies. Genome Med. 2020;12(1):8.
Misyura M, Zhang T, Sukhai MA, Thomas M, Garg S, Kamel-Reid S, et al. Comparison of next-generation sequencing panels and platforms for detection and verification of somatic tumor variants for clinical diagnostics. J Mol Diagn. 2016;18(6):842–50.
Li W, Zhang J, Guo L, Chuai S, Shan L, Ying J. Combinational analysis of FISH and immunohistochemistry reveals rare genomic events in ALK fusion patterns in NSCLC that responds to crizotinib treatment. J Thorac Oncol. 2017;12(1):94–101.
Chalmers ZR, Connelly CF, Fabrizio D, Gay L, Ali SM, Ennis R, et al. Analysis of 100,000 human cancer genomes reveals the landscape of tumor mutational burden. Genome Med. 2017;9(1):34.
Dudley JC, Lin MT, Le DT, Eshleman JR. Microsatellite instability as a biomarker for PD-1 blockade. Clin Cancer Res. 2016;22(4):813–20.
Goodman AM, Kato S, Bazhenova L, Patel SP, Frampton GM, Miller V, et al. Tumor mutational burden as an independent predictor of response to immunotherapy in diverse cancers. Mol Cancer Ther. 2017;16(11):2598–608.
Schrock AB, Ouyang C, Sandhu J, Sokol E, Jin D, Ross JS, et al. Tumor mutational burden is predictive of response to immune checkpoint inhibitors in MSI-high metastatic colorectal cancer. Ann Oncol. 2019;30(7):1096–103.
Arora S, Velichinskii R, Lesh RW, Ali U, Kubiak M, Bansal P, et al. Existing and emerging biomarkers for immune checkpoint immunotherapy in solid tumors. Adv Ther. 2019;36(10):2638–78.
Gong B, Li D, Kusko R, Novoradovskaya N, Zhang Y, Wang S, et al. Cross-oncopanel study reveals high sensitivity and accuracy with overall analytical performance depending on genomic regions. Genome Biol. 2021;22(1):109.
Mao X, Zhang Z, Zheng X, Xie F, Duan F, Jiang L, et al. Capture-based targeted ultradeep sequencing in paired tissue and plasma samples demonstrates differential subclonal ctDNA-releasing capability in advanced lung cancer. J Thorac Oncol. 2017;12(4):663–72.
Li YS, Jiang BY, Yang JJ, Zhang XC, Zhang Z, Ye JY, et al. Unique genetic profiles from cerebrospinal fluid cell-free DNA in leptomeningeal metastases of EGFR-mutant non-small-cell lung cancer: a new medium of liquid biopsy. Ann Oncol. 2018;29(4):945–52.
Li H, Durbin R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics. 2009;25(14):1754–60.
McKenna A, Hanna M, Banks E, Sivachenko A, Cibulskis K, Kernytsky A, et al. The Genome Analysis Toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 2010;20(9):1297–303.
Koboldt DC, Zhang Q, Larson DE, Shen D, McLellan MD, Lin L, et al. VarScan 2: somatic mutation and copy number alteration discovery in cancer by exome sequencing. Genome Res. 2012;22(3):568–76.
Wang K, Li M, Hakonarson H. ANNOVAR: functional annotation of genetic variants from high-throughput sequencing data. Nucleic Acids Res. 2010;38(16):e164.
Cingolani P, Platts A, le Wang L, Coon M, Nguyen T, Wang L, et al. A program for annotating and predicting the effects of single nucleotide polymorphisms, SnpEff: SNPs in the genome of Drosophila melanogaster strain w1118; iso-2; iso-3. Fly (Austin). 2012;6(2):80–92.
Newman AM, Bratman SV, Stehr H, Lee LJ, Liu CL, Diehn M, et al. FACTERA: a practical method for the discovery of genomic rearrangements at breakpoint resolution. Bioinformatics. 2014;30(23):3390–3.
Li MM, Datto M, Duncavage EJ, Kulkarni S, Lindeman NI, Roy S, et al. Standards and Guidelines for the Interpretation and Reporting of Sequence Variants in Cancer: a Joint Consensus Recommendation of the Association for Molecular Pathology, American Society of Clinical Oncology, and College of American Pathologists. J Mol Diagn. 2017;19(1):4–23.
Tang Y, Li Y, Wang W, Lizaso A, Hou T, Jiang L, et al. Tumor mutation burden derived from small next generation sequencing targeted gene panel as an initial screening method. Transl Lung Cancer Res. 2020;9(1):71–81.
Zhu L, Huang Y, Fang X, Liu C, Deng W, Zhong C, et al. A novel and reliable method to detect microsatellite instability in colorectal cancer by next-generation sequencing. J Mol Diagn. 2018;20(2):225–31.
Cai Z, Wang Z, Liu C, Shi D, Li D, Zheng M, et al. Detection of Microsatellite Instability from Circulating Tumor DNA by Targeted Deep Sequencing. J Mol Diagn. 2020;22(7):860–70.
Willis J, Lefterova MI, Artyomenko A, Kasi PM, Nakamura Y, Mody K, et al. Validation of microsatellite instability detection using a comprehensive plasma-based genotyping panel. Clin Cancer Res. 2019;25(23):7035–45.
Mosele F, Remon J, Mateo J, Westphalen CB, Barlesi F, Lolkema MP, et al. Recommendations for the use of next-generation sequencing (NGS) for patients with metastatic cancers: a report from the ESMO Precision Medicine Working Group. Ann Oncol. 2020;31(11):1491–505.
Cheng DT, Mitchell TN, Zehir A, Shah RH, Benayed R, Syed A, et al. Memorial sloan kettering-integrated mutation profiling of actionable cancer targets (MSK-IMPACT): a hybridization capture-based next-generation sequencing clinical assay for solid tumor molecular oncology. J Mol Diagn. 2015;17(3):251–64.
Samstein RM, Lee CH, Shoushtari AN, Hellmann MD, Shen R, Janjigian YY, et al. Tumor mutational load predicts survival after immunotherapy across multiple cancer types. Nat Genet. 2019;51(2):202–6.
Woodhouse R, Li M, Hughes J, Delfosse D, Skoletsky J, Ma P, et al. Clinical and analytical validation of FoundationOne Liquid CDx, a novel 324-Gene cfDNA-based comprehensive genomic profiling assay for cancers of solid tumor origin. PLoS ONE. 2020;15(9):e0237802.
Odegaard JI, Vincent JJ, Mortimer S, Vowles JV, Ulrich BC, Banks KC, et al. Validation of a plasma-based comprehensive cancer genotyping assay utilizing orthogonal tissue- and plasma-based methodologies. Clin Cancer Res. 2018;24(15):3539–49.
Zill OA, Banks KC, Fairclough SR, Mortimer SA, Vowles JV, Mokhtari R, et al. The landscape of actionable genomic alterations in cell-free circulating tumor DNA from 21,807 advanced cancer patients. Clin Cancer Res. 2018;24(15):3528–38.
Acknowledgments
The authors thank all the patients and their families for their cooperation and support. We also thank the investigators, study coordinators, operation staff, and the whole project team who worked on this project. We also appreciate the active support of the staff of Burning Rock Biotech, particularly Analyn Lizaso, Jian Wang, Jianxing Xiang, Zhange Chen, Jiaqi Chu, Chanhe Li, Min Li, Yuan Zhou, Jinying Liu, Jing Liu, Wenjie Sun, and Zhou Zhang.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Funding
This study was supported by grants from the National Natural Science Foundation of China (grant number: 81871886 to MW), the Science and Technology Program of Guangzhou, China (grant number: 202103000063 to MW), the Special fund of Foshan Summit Plan General Project (grant number: 2020B004 to FL), the Ningbo Health Branding Subject Fund (grant number: PPXK2018-05 to GZ), The Natural Science Foundation of Ningbo (grant number: 2019A610225 to GZ), the Zhejiang Medicine and Health Science and Technology Project (grant numbers: 2022KY1138 and 2021KY1009 to GZ), The Key Discipline of Hwamei Hospital, University of Chinese Academy of Science (grant number: 2020ZDXK03 to GZ), The Hwamei Fund (grant number: 2019HM to GZ), and the Wu Jieping Medical Foundation (grant number: 320.6750.18105 to TX). The funders had no role in the conceptualization, design, data collection or analysis, decision to publish, or preparation of the manuscript.
Ethical approval and consent to participate
All procedures performed in studies involving human participants were performed in accordance with the Declaration of Helsinki. This study protocol was approved by the ethics committee of The Second Affiliated Hospital of Harbin Medical University (approval number: KY2021-189). Written informed consent was obtained from all participants included in the study.
Consent for publication
Not applicable
Competing interests
Minghui Wang, Xianshan Chen, Yongmei Dai, Duoguang Wu, Fang Liu, Zheng Yang, Baozhi Song, Li Xie, Liangwei Yang, Weidi Zhao, Chenxu Zhang, Weixi Shen, Chengjuan Fan, Chong Teng, Xue Zhao, Naisheng Gao, Di Shang, Guofang Zhao, and Tao Xin have no conflicts of interest that are directly relevant to the content of this article.
Availability of data and material
Not applicable.
Availability of data and material
All authors confirm adherence to the policy. The data that support the findings of this study are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
Author contributions
MW, YD, GZ, and TX conceived of the study and drafted the manuscript. MW, FL, GZ, and TX acquired funding. All the authors collected and analyzed the clinical data, participated in the data interpretation, data analysis, and manuscript writing and editing. XC contributed significantly to the data analysis and interpretation during the manuscript revision. All authors contributed to the revision and approved the final manuscript.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, M., Chen, X., Dai, Y. et al. Concordance Study of a 520-Gene Next-Generation Sequencing-Based Genomic Profiling Assay of Tissue and Plasma Samples. Mol Diagn Ther 26, 309–322 (2022). https://doi.org/10.1007/s40291-022-00579-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40291-022-00579-1