Abstract
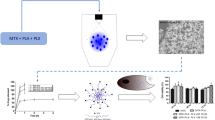
Poly(lactic-co-glycolic acid)(PLGA) microparticles represent an important class of materials used for drug delivery. Current synthesis frequently uses conventional emulsion, where dichloromethane(DCM) is used as the organic phase solvent. Due to the health and environmental toxicity of DCM and its slow degradation, this work replaces DCM with a greener solvent, dimethyl carbonate(DMC). To attain narrow distribution of PLGA particle size, microfluidic flow focusing was chosen over conventional emulsion. This new approach successfully produced PLGA microparticles encapsulated with flavopiridol, a kinase inhibitor. These particles exhibit sustained release profile more desirable than the conventional counterparts. The cytotoxicity and activity tests have demonstrated high biocompatibility and efficacy of these PLGA particles. The high sustainability is also evaluated using simple E-Factor(sEF) and complete E-Factor(cEF). The lower health and environmental toxicities of DMC than DCM are evidenced by approximately one order of magnitude higher in lethal dose, i. e., 50%(LD50) values in rat, 5-fold faster degradation rate, and 30% higher GlaxoSmithKline(GSK) combined greenness value. The approach reported in this work shall provide a new and green means for drug delivery in general. The products enable local sustained delivery of flavopiridol for prevention of post-traumatic osteoarthritis, and anti-cancer therapy.
Similar content being viewed by others
References
Panyam J., Labhasetwar V., Adv. Drug Deliv. Rev., 2003, 55, 329
Kumari A., Yadav S. K., Yadav S. C., Colloids Surf. B. Biointerfaces, 2010, 75, 1
Makadia H. K., Siegel S. J., Polymers(Basel), 2011, 3, 1377
Liu G., Li Y., Yang S., Zhao Y. A., Lu T., Jia W., Ji X., Luo Y., Chem. Res. Chinese Universities, 2019, 35(3), 514
Cai Q., Qiao C., Ning J., Ding X., Wang H., Zhou Y., Chem. Res. Chinese Universities, 2019, 35(5), 908
Kim T., See C. W., Li X., Zhu D., Eng. Regen., 2020, 1, 6
Bolandparvaz A., Yik J. H., Lewis J., Haudenschild D. R., Osteoarthr. Cartil., 2018, 26, S303
Ren H., Han M., Zhou J., Zheng Z.-F., Lu P., Wang J.-J., Wang J.-Q., Mao Q.-J., Gao J.-Q., Ouyang H. W., Biomaterials, 2014, 35, 6585
Xu Q., Hashimoto M., Dang T. T., Hoare T., Kohane D. S., Whitesides G. M., Langer R., Anderson D. G., Small, 2009, 5, 1575
Vasiliauskas R., Liu D., Cito S., Zhang H., Shahbazi M.-A., Sikanen T., Mazutis L., Santos H. A., ACS Appl. Mater. Interfaces, 2015, 7, 14822
Hung L.-H., Teh S.-Y., Jester J., Lee A. P., Lab. Chip., 2010, 10, 1820
Zhao Y., Shum H. C., Chen H., Adams L. L. A., Gu Z., Weitz D. A., J. Am. Chem. Soc., 2011, 133, 8790
Wang H., Liu Y., Chen Z., Sun L., Zhao Y., Sci. Adv., 2020, 6, eaay1438
Chen H., Bian F., Sun L., Zhang D., Shang L., Zhao Y., Adv. Mater., 2020, 32, 2005394
Zhang H., Chen G., Yu Y., Guo J., Tan Q., Zhao Y., Adv. Sci., 2020, 7, 2000789
Liu D., Zhang H., Fontana F., Hirvonen J. T., Santos H. A., Lab Chip, 2017, 17, 1856
Xu J., Zhang S., Machado A., Lecommandoux S., Sandre O., Gu F., Colin A., Sci. Rep., 2017, 7, 4794
Kim H.-G., Kim K.-M., Kim Y. H., Lee S. H., Kim G. M., J. Biobased Mater. Bioenergy, 2013, 7, 108
Schlosser P. M., Bale A. S., Gibbons C. F., Wilkins A., Cooper G. S., Environ Health Perspect, 2015, 123, 114
Kim H., Kim S., Sah H., J. Biomater. Sci. Polym. Ed., 2018, 29, 35
Environmental Protection Agency, Federal Register, 2019, 84, 11420
Grassian V. H., Environmental Science & Technology, 2007, 41, 4840
Yang X., Zhao X., Phelps M. A., Piao L., Rozewski D. M., Liu Q., Lee L. J., Marcucci G., Grever M. R., Byrd J. C., Dalton J. T., Lee R. J., Int. J. Pharm., 2009, 365, 170
Chao S.-H., Price D. H., J. Biol. Chem., 2001, 276, 31793
Bugai A., Quaresma A. J. C., Friedel C. C., Lenasi T., Düster R., Sibley C. R., Fujinaga K., Kukanja P., Hennig T., Blasius M., Geyer M., Ule J., Dölken L., Barborič M., Mol. Cell, 2019, 74, 254
Morales F., Giordano A., Cell Cycle, 2016, 15, 519
Yik J. H. N., Hu Z. A., Kumari R., Christiansen B. A., Haudenschild D. R., Arthritis Rheumatol, 2014, 66, 1537
Hu Z., Yik J. H. N., Cissell D. D., Michelier P. V., Athanasiou K. A., Haudenschild D. R., Eur. Cell Mater., 2016, 30, 200
Fukui T., Yik J. H. N., Doyran B., Davis J., Haudenschild A. K., Adamopoulos I. E., Han L., Haudenschild D. R., Osteoarthr. Cartil., 2021, 29, 68
Malachowski T., Hassel A., Eng. Regen., 2020, 1, 35
Tundo P., Selva M., Acc. Chem. Res., 2002, 35, 706
McDonald J. C., Duffy D. C., Anderson J. R., Chiu D. T., Wu H., Schueller O. J. A., Whitesides G. M., Electrophoresis, 2000, 21, 27
Trantidou T., Elani Y., Parsons E., Ces O., Microsyst. Nanoeng., 2017, 3, 16091
Hines D. J., Kaplan D. L., Crit. Rev. Ther. Drug Carrier Syst., 2013, 30, 257
Sedlacek H. H., Crit. Rev. Oncol. Hematol., 2001, 38, 139
Sedlacek H. H., Czech J., Naik R., Kaur G., Worland P., Losiewicz M., Parker B., Carlson B., Smith A., Senderowicz A., Sausville E., Int. J. Oncol., 1996, 9, 1143
Xiong S., George S., Yu H., Damoiseaux R., France B., Ng K. W., Loo J. S.-C., Arch. Toxicol., 2013, 87, 1075
Porceddu M., Buron N., Roussel C., Labbe G., Fromenty B., Borgne-Sanchez A., Toxicological Sciences, 2012, 129, 332
Sanabria-Ríos D. J., Rivera-Torres Y., Rosario J., Ríos C., Gutierrez R., Carballeira N. M., Vélez C., Zayas B., Álvarez-Colón F., Ortiz-Soto G., Serrano V., Altieri-Rivera J., Ríos-Olivares E., Rodríguez J. W., Bioorg. Med. Chem. Lett., 2015, 25, 2174
Albisa A., Piacentini E., Arruebo M., Sebastian V., Giorno L., ACS Sustain. Chem. Eng., 2018, 6, 6663
Pinheiro C. T., Quina M. J., Gando-Ferreira L. M., ACS Sustain. Chem. Eng., 2018, 6, 6820
Alder C. M., Hayler J. D., Henderson R. K., Redman A. M., Shukla L., Shuster L. E., Sneddon H. F., Green Chemistry, 2016, 18, 3879
Han F. Y., Thurecht K. J., Whittaker A. K., Smith M. T., Front Pharmacol., 2016, 7, 185
Zhai S., Senderowicz A. M., Sausville E. A., Figg W. D., Ann. Pharmacother., 2002, 36, 905
Center for Drug Evaluation and Research, U. S. Food and Drug Administration, 1997, BP2, 20
Blewis M. E., Lao B. J., Jadin K. D., McCarty W. J., Bugbee W. D., Firestein G. S., Sah R. L., Biotechnol. Bioeng., 2010, 106, 149
Sheikh Z., Brooks P. J., Barzilay O., Fine N., Glogauer M., Materials(Basel), 2015, 8, 5671
Gustafson H. H., Holt-Casper D., Grainger D. W., Ghandehari H., Nano Today, 2015, 10, 487
Conaghan P. G., Cohen S. B., Berenbaum F., Lufkin J., Johnson J. R., Bodick N., Arthritis Rheumatol, 2018, 70, 204
Kumar A., Bendele A. M., Blanks R. C., Bodick N., Osteoarthr. Cartil., 2015, 23, 151
Office of Chemical Safety, Environmental Protection Agency, 2018, EPA-740-R1-7016, 33
Acknowledgements
This work was supported by the DOD(CDMRP Award PR171305) and UC Davis. We thank Dr. Arpad KARSAI, Professor Jamal LEWIS, Ms. Audrey SULKANEN, and Ms. Minyuan WANG for helpful discussions. We also thank Professor Xi CHEN and Dr. Hai YU for their technical assistance in post synthesis treatments.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare no conflicts of interest.
Supporting Information for
Rights and permissions
About this article
Cite this article
Owen, M.J., Yik, J.H.N., Ye, C. et al. A Green Approach to Producing Polymer Microparticles for Local Sustained Release of Flavopiridol. Chem. Res. Chin. Univ. 37, 1116–1124 (2021). https://doi.org/10.1007/s40242-021-1262-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40242-021-1262-8