Abstract
Introduction
This study evaluated the efficacy of combined interferon α-2b (IFNα2b) and 5-fluorouracil (5-FU) as primary treatment for ocular surface squamous neoplasia (OSSN).
Methods
In this retrospective study, 27 eyes with OSSN followed by topical application of combined IFNα2b and 5-FU were examined. Reported outcome measures were tumor response, visual acuity, time to complete resolution, recurrence and treatment complications.
Results
Twenty-six patients (17 male, 9 female) had a mean age of 63.9 (median, 67; range 22–83) years. Complete tumor response was observed in 24 eyes (88.9%). Three eyes (11.1%) showed partial response to the chemotherapy agents and later underwent surgical tumor removal. The median time to complete resolution was 6 (mean, 6.1; range, 3–11) weeks. Of these, the patients received between one to three cycles of 5-FU therapy (median, 2; mean, 1.8). Complications noted were transient irritation at 5-FU cycle (11 eyes, 40.7%). There was no tumor recurrence at mean follow-up of 16.1 (median, 12; range 6–38) months.
Conclusions
Combination therapy of IFNα2b and 5-FU was a safe and effective treatment, inducing a short duration of administration and low recurrence rate for OSSN.
Trial Registration
Retrospectively registered, UHCT22048.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Ocular surface squamous neoplasia (OSSN) is the most common malignancy of the ocular surface. Surgical removal for OSSN has a high recurrence and complication rate, including limbal stem cell deficiency and conjunctival scarring. |
There has been a shift in the treatment of OSSN from surgical treatment alone to the use of local chemotherapeutic agents alone or surgery in combination with chemotherapeutic agents, but chemotherapeutic agents like IFNα2b, MMC and 5-FU also have side effects. |
We hypothesized that IFNα2b combined with 5-FU could be more effective and rapid in treating OSSN than monotherapy. |
Combination therapy of IFNα2b and 5-FU was a safe and effective treatment for OSSN. This regimen reduced the duration of IFNα2b use and the frequency of 5-FU cycle with a high frequency of tumor resolution and low recurrence rates. It provides a new method for the treatment of OSSN. |
Introduction
Ocular surface squamous neoplasia (OSSN) is the most common malignancy of the ocular surface, with a worldwide incidence 0.13 to 1.9 per 100,000 individuals [1, 2]. Many factors contribute to the development of OSSN such as ultraviolet radiation, human immunodeficiency virus infection, human papillomavirus infection, vitamin A deficiency, xeroderma pigmentosum, allergic conjunctivitis, smoking, chemical exposure and immunosuppression caused by medications after organ transplantation [3,4,5,6,7]. Although surgical removal has been considered the standard care for OSSN [8,9,10], local chemotherapy has increased in popularity over the past 20 years [9, 11,12,13]. Topical chemotherapeutic agents used today are mainly mitomycin-C (MMC), 5-fluorouracil (5-FU) and interferon α-2b (IFNα2b) [14,15,16].
Mitomycin C is a potent antimetabolite resulting in DNA alkylation in all phases of the cell cycle [17]. It has frequent side effects that include pain in most patients and epitheliopathy [12, 18], which has limited its usage.
5-Fluorouracil is structurally similar to thymidine and uracil and inhibits DNA formation [19]. It has been used to treat many epithelial cancers because of its actions on rapidly multiplying tumor cells [20]. Treatment with 5-FU is generally well tolerated especially when administered four times daily for 1 week, followed by a drug holiday for 3 weeks [15].
IFNα2b is a recombinant glycoprotein that activates effector proteins by binding to the cell surface of their targets [21]. Its role as antineoplastic agent is due to a combination of antiproliferative, antiangiogenic and cytotoxic effects, as well as through a possible enhancement of the host antitumor surveillance mechanism [22]. IFNα2b maintains comparable rates of resolution and recurrence to the aforementioned drugs [12]. The fact that the topical form is extremely well tolerated makes it most popular nowadays [12].
Nevertheless, the resolution time is as long as 11 weeks to 6.6 months for 5-FU and 2.3–6 months for IFNα2b in the literature from 2003 to 2022 including > 20 cases (Table 1). We speculated the combined use of IFNα2b and 5-FU enhances the effectiveness by shortening the resolution time and reducing the 5-FU cycle. Herein, we share our experiences and insights from the combined application of IFNα2b and 5-FU in the treatment of OSSN.
Methods
Study Population
This retrospective study was approved (UHCT22048) by the Ethics Committee of Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, according to the tenets of the Declaration of Helsinki. Twenty-six consecutive patients (27 eyes) with a clinical diagnosis of OSSN by two cornea specialists using slit-lamp biomicroscopy between July 1, 2016, and July 1, 2021, were recorded.
Data Extracted
Patient records were reviewed for demographic information (age, sex), best corrected visual acuity (BCVA), OSSN risk factors (human immunodeficiency virus, smoking, history of OSSN, chronic systemic immunosuppression) and other diagnoses. Specific characteristics of each tumor were documented including the involved eye, unifocal versus multifocal status, number of involved limbal clock hours and morphological findings (leukoplakic, gelatinous, papillomatous, or flat) based on descriptions and photographs.
Topical IFNα2b Chemotherapy Combined with 5-FU
Topical IFNα2b (1 million IU per milliliter, MIU/ml) was compounded by adding 4 ml of distilled sterile water to 1 ml 5 MIU/ml IFNα2b (Anfulong, Tianjing Sinobioway Biomedicine, China) and preserved in a refrigerator at 4 °C. The drops were administered as one drop four times daily and continued until clinical resolution. To prevent tumor recurrence, an additional 4-week treatment was used after the tumor had completely resolved. Topical 5-FU was used at a concentration of 1%, administered as one drop four times daily for 1 week, followed by a drug holiday for 3 weeks. This monthly cycle was continued until complete clinical resolution, after which the drops were discontinued. For some patients who were sensitive to side effects, preservative-free artificial tears were added four times daily. All patients were followed up weekly until complete resolution and then at different intervals.
Outcome Measures
The main outcome measures were tumor response, time to tumor complete resolution, total duration of IFNα2b, 5-FU cycle, tumor recurrence, new tumor appearance, visual acuity and treatment-related complications.
Tumor response was recorded as the time to complete resolution (clinically defined on slit-lamp examination). Partial response to chemotherapy was defined as initial effectiveness followed by 2 consecutive weeks of no significant change in tumor. No response was defined as no significant change for 4 consecutive weeks from the start of treatment. Recurrence was defined as reappearance at a similar location as the original tumor (after complete resolution of original tumor). New tumors were defined as those appearing at a different location from that of original tumor (after complete resolution of original tumor). Follow-up duration was defined from the time of resolution of lesion until the last visit. Complication surveys included irritation (pain, redness, tearing and photophobia), limbal stem cell deficiency and infection.
Statistical Analyses
Statistical analyses were performed using Microsoft Excel (Microsoft, Redmont, WA). Frequencies of demographic and clinical variables were calculated. The data were expressed as means ± SD, median and range for quantitative parameters.
Results
Pretreatment Characteristics
Demographics of the study population are presented in Table 2. Seventeen males (65.4%) and nine females (34.6%) were included in this study. The average age was 63.9 (range, 22 to 83) years. Of the risk factors associated with OSSN, 1 (3.8%) patient had a history of HIV, 16 (61.5.%) were current smokers, 4 (15.4%) had a prior history of OSSN, and 1 (3.8%) patient with thrombocytopenia, a chronic systemic immune disorder, was being treated with oral steroids. Other ocular diagnoses of patients included 14 (53.8%) cataracts, 1 (3.8%) chronic dacryocystitis, 1 (3.8%) intraocular lens eye and 1 (3.8%) myopia.
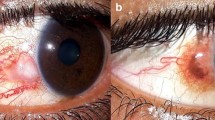
The 27 eyes included 16 (59.3%) right eyes and 11 (40.7%) left eyes (one patient with bilateral onset). The mean number of limbal clock hours involved was 4.3 (median, 4; range, 0–9). The tumor appearances were 19 (70.1%) papillomatous (Fig. 1A), 2 (7.4%) gelatinous (Fig. 1B, white arrows), 2 (7.4%) leukoplakic (Fig. 1C) and 4 (14.8%) flat (Fig. 1D, E white arrows). Four (14.8%) papillomatous eyes developed multifocal tumors (Fig. 1F).
Tumor Response
Tumors of all patients responded to IFNα2b combined with 5-FU therapy (Table 3). Twenty-four (88.9%) lesions resolved completely. Among the 24 eyes, the median time to complete resolution was 6 (mean, 6.1; range, 3–11) weeks. Patients received one to three cycles (median, 2; mean, 1.8) of 5-FU therapy. The total duration of IFNα2b ranged from 8–15 (median, 10; mean, 10.25) weeks.
Two patients (3 eyes) showed partial response to the chemotherapy. The number and size of the masses decreased during the first 4 to 5 weeks. However, over the next 2 weeks, the tumor changes stalled. Eventually, one patient underwent surgery at week 7 and the other at week 8. Postoperatively, both were then treated with adjuvant IFNα2b for 4 weeks and one cycle of 5-FU.
Visual Acuity
The causes of low visual acuity at presentation included preexisting cataract (n = 14), myopia (n = 1) and tumor-induced occlusion of the pupillary area (n = 9). No loss of vision was associated with the treatment. At the last follow-up, 9 (37.5%) patients showed improvement of visual acuity; 15 (62.5%) patients still had their pretreatment visual acuity (Table 3).
Side Effects and Complications
Eleven (40.7%) patients experienced medication-related side effects which were tolerated. All of them experienced redness, pain, tearing, photophobia and swelling during 5-FU use, which disappeared within 1 to 2 weeks after 5-FU was discontinued. All patients completed the course of 5-FU therapy. No signs of partial or total limbal stem cell deficiency or persistent epithelial defect were noted (Table 3).
Follow-up and Tumor Recurrence
With longer follow-up (median, 12 months; mean 16.1 months; range, 6–38 months), there was no evidence of recurrence in all patients (Table 3).
Representative Case Reports
Case 8: Papillomatous Tumor
An 80-year-old man complained of redness and loss of vision in his right eye for 10 months. He had undergone excisional biopsy 1 year ago in another hospital because of a history of OSSN. The pathology confirmed atypical hyperplasia without specific description of the margin. The papillomatous tumor involved 8 clock hours of limbus (Fig. 2A1). His visual acuity had deteriorated to finger counting/50 cm because of the occlusion of the pupillary area (Fig. 2A1, A2). After topical chemotherapy, the tumor displayed complete regression at the 3-week visit (Fig. 2B1) although an uneven corneal surface was noted (Fig. 2B2). He received 8 weeks of chemotherapy including two cycles of 5-FU. No tumor recurrence was observed (Fig. 2C1) and the ocular surface was stable (Fig. 2C2) at 38-month follow-up.
Case 8: Slit-lamp photographs of an 80-year-old man with OSSN. A1 Papillomatous tumor in the right eye involving 8 clock hours of limbus, cornea and conjunctiva. B1 After topical chemotherapy, the tumor displayed complete regression at 3-week visit (B2) although an uneven corneal surface was noted. C1 No tumor recurrence was observed and (C2) the ocular surface was stable at 38-month follow-up
Case 21: Gelatinous Tumor
A 43-year-old female complained of redness, foreign body sensation and the presence of a reddish mass in her left eye for 6 months. She had a medical history of chronic dacryocystitis without any treatment. The gelatinous tumor involved 2.5 clock hours of limbus (Fig. 3A). It displayed complete tumor regression after 4 weeks of topical IFNα2b and one cycle of 5-FU without corneal epithelial defect (Fig. 3C). She received another 4 weeks of topical IFNα2b after complete resolution to prevent tumor recurrence. The ocular surface was stable without any inflammation or recurrence for 18 months of follow-up (Fig. 3D).
Case 21: Slit-lamp photographs of a 43-year-old female with diagnosis of OSSN and chronic dacryocystitis. A The gelatinous tumor in the right eye involving 2.5 clock hours of limbus. B It displayed complete tumor regression with 2 weeks of topical IFNα2b and one cycle of topical 5-FU and showed typical symptoms of redness and irritation during the 5-FU treatment, (C) without corneal epithelial defect. D No tumor recurrence was observed at 18-month follow-up
Case 26: Flat Tumor
A 71-year-old female was referred to our clinic with foreign body sensation and blurred vision for 9 months. Slit-lamp biomicroscopy showed an elevated diffuse epitheliopathy (Fig. 4A1, white arrows) with a central ulcer (Fig. 4A2, black arrow). Under the clinical diagnosis of OSSN, topical IFNα2b and 5-FU chemotherapy was administrated. The tumor had shrunk (Fig. 4B1, B2, white arrows) and ulcer healed at the 4-week visit (Fig. 4B2). Complete regression was observed after 5-week treatment including two cycles of 5-FU (Fig. 4C1, C2). She was followed up for 6 months without recurrence.
Case 26: Slit-lamp photographs of a 71-year-old female with flat OSSN. A1 Slit-lamp biomicroscopy showed an elevated diffuse epitheliopathy (white arrows) with a central ulcer (A2, black arrow). B1, B2 The tumor had shrunk (white arrows) and ulcer healed at the 4- week visit after topical IFNα2b and 5-FU chemotherapy. C1, C2 Complete regression was observed after 5 weeks of treatment
Case 22: Mutifocal Tumor Partial Response to Chemotherapy
A 34-year-old male presented with a recurrent OSSN in both eyes for 12 months. He had undergone surgical resection 18 months before in another hospital (pathological diagnosis was atypical hyperplasia). The multiple papillomatous tumors in the right eye involved upper and lower palpebral and fornix conjunctiva (Fig. 5A). The tumors shrank after 8 weeks of IFNα2b and two cycles of 5-FU (Fig. 5B). Partial response was judged when tumors had not shrunk further according to comparison of the slit-lamp photos from week 6 and week 8. The patient received bilateral surgery to remove the masses at week 8. Postoperatively, adjuvant IFNα2b for an additional 4 weeks combined with one cycle of 5-FU was administered. No tumor recurrence was observed at 11-month follow-up (Fig. 5C).
Case 22: Slit-lamp photographs of a 34-year-old male with multiple papillomatous tumors in both eyes. A Papillomatous multiple tumors in the right eye involving upper and lower fornix palpebral conjunctiva. B The tumors shrank after 8 weeks of IFNα2b and two cycles of 5-FU. The patient received bilateral surgery to remove the masses at week 8. C No tumor recurrence was observed at 11-month follow-up
Discussion
Surgical removal of OSSN has the advantage of serving as both a diagnostic and therapeutic procedure, providing both an accurate histological diagnosis and rapid tumor resolution. Studies have shown that no difference in the recurrence rate of OSSN was found between surgical versus IFNα2b therapy [23]. However, surgery often leads to serious complications including limbal stem cell deficiency and conjunctival scarring [24]. Over the past 20 years, there has been a shift in the treatment of OSSN from surgical treatment alone to the use of local chemotherapeutic agents or surgery in combination with chemotherapeutic agents. Topical chemotherapy minimizes complications, and the medication acts on the entire ocular surface, offering significant advantages in the treatment of diffuse lesions, microscopic lesions and surgical residual tumors [14, 23, 25]. However, local chemotherapeutic agents like IFNα2b and 5-FU have long duration of use and cause irritation symptoms and pain, which often lead to poor patient compliance. Therefore, we strove to achieve the goal of rapid regression of OSSN by combining IFNα2b and 5-FU.
Since use of IFN and 5-FU for ocular surface lesions was reported in the nineteenth century, several studies have confirmed their effectiveness in the treatment of OSSN [26,27,28,29]. For IFN treatment, the overall response frequency ranged from 75 to 100% [16, 30, 31]; for 5-FU, it ranged from 82–100% [15, 32, 33]. In our current study, all the tumors were in remission, although 12.5% showed partial response. There was no one case of primary tumor recurrence, new tumor or metastasis. In the two cases of partial response, one patient was HIV infected and the other had a history of OSSN and had undergone excisional surgery as the primary treatment in both eyes 18 months ago; both were identified as recurrent patients. Both patients had multiple masses. These factors may reduce the sensitivity of tumors to chemotherapeutic agents. OSSN often presents a variety of appearances [34]. A patient with OSSN masquerading as a recalcitrant epithelial keratitis recovered after four cycles of topical chemotherapy with 5-FU [35]. Flat tumor with epithelial defect (case 26) tends to be misdiagnosed as keratitis, and doctors should pay more attention to it.
Wadler et al. [36] first reported that IFNα2b combined with 5-FU has achieved certain effects on the treatment of colon cancer. This combination therapy was also applied to esophageal cancer [37] and gastric cancer [38] and achieved satisfactory results. The combination of IFNα2b and 5-FU reduced the dosage of chemotherapy agents and the toxic and side effects on normal tissue cells. Several investigations suggest that alpha-IFN could play a positive role in immuno-chemotherapy of cancer through multiple mechanisms not entirely related to direct antitumor effects of the agent [39, 40]. Alpha-IFN might elevate the levels of the active 5-FU metabolite 5-fluoro-2’-deoxyuridine-5’- monophosphate in the cell, possibly leading to increased inhibition of the target enzyme thymidylate synthase, which might enhance DNA damage [41]. Compared with monotherapy in the most recently published analysis (Table 1), our study confirmed that combination therapy could reduce the frequency of 5-FU use (median, 2; mean, 1.8; range, 1–3) and shorten the duration of IFNα2b treatment (median, 10 weeks; mean, 10.25; range, 8–15;) for OSSN. Joag et al. [15] reported that among the 44 eyes, 36 lesions (82%) resolved completely with 5-FU therapy; the median number of cycles was 4 (range, 2–9; mean, 3.8). In Venkateswaran’s study [42], the number of 5-FU cycles was 4 (range, 2–12; mean, 4.2), and she also reported that in the 48 eyes treated with IFNα2b therapy, 81.3% of lesions (n = 39) completely resolved (median number of months treated 4, range 2–8, mean 4.2). In most studies, the duration of IFNα2b was a few (range 2–6) months [30, 43, 44]. However, the mechanism of the combination treatment in OSSN needs to be further studied.
In this study, pathological biopsies were not performed on all patients. Instead, two experienced cornea specialists independently made the diagnosis of OSSN. Biopsy, on the other hand, increases the risk of seed transfer and additional invasive procedures for patients. It may not make sense to remove part of the tumor to confirm the pathological diagnosis while treating the rest with chemotherapy. Hence, we chose to avoid biopsy as this is consistent with how most clinicians currently approach clinically evident OSSN, especially when starting topical therapy. This view was recently supported by Shields [45]. Nowadays, as a non-contact, non-invasive examination, ultra-high-resolution anterior segment optical coherence tomography (HR-OCT) has emerged as a useful methods to aid in the diagnosis and management of OSSN [46]. It is particularly useful at detecting epithelial thickening and differentiating epithelial lesions from subepithelial lesions of the conjunctiva and cornea, assisting in the differentiation of OSSN, pterygia, pinguecula, lymphoma and melanocytic lesions [47, 48]. Some researchers also believed that the tumor response defined by a detailed slit-lamp examination would result in a premature termination and reduction time of treatment in some patients with subclinical disease that can be detected with HR-OCT [15, 49]. Although definitive imaging and pathology were lacking to exclude subclinical lesions, no tumor recurrence was found at mean follow-up time of 16.1 months; there was no premature discontinuation of chemotherapy in our study.
The costs of treatment are also an important factor influencing the treatment options. Of the therapeutic agents, interferon has fewer side effects but is much more expensive (approximately $600 for a month's supply in the USA) and requires refrigeration for storage [34, 50]. 5-Fluorouracil is relatively inexpensive and stable at room temperature. A 1-month treatment of 5-FU costs approximately $38 in the US [34, 42]. Fortunately, in China, both IFNα2b and 5-FU are cheap and readily available. For patients in our study, the cost of IFNα2b drops was $16 for a 4-week supply and $8 for one cycle of 5-FU, which is much lower than that of surgical treatment in our hospital. Combination chemotherapy has been proposed as a promising therapeutic strategy to overcome drug resistance and improve efficacy of monotherapy regimens in cancer. Since we proposed this combination of IFNα2b and 5-FU based on clinical observations, more cellular and molecular experimenta will be performed in our next study to demonstrate the advantages of this therapy.
In our current study, few patients experienced discomfort during IFNα2b treatment. Side effects were more likely observed during 5-FU use. One limitation of our study was a non-standardized method for evaluating side effects and treatment. In this retrospective, we classified redness, pain, tearing, photophobia, swelling and itching as 5-FU irritation, which resulted in a much higher side effect index (40.7%) than previously reported [15, 42]. Encouragingly, all side effects abated when the chemotherapeutic agents were discontinued. A confocal microscopy study showed no long-term differences between the treated and control (fellow) eyes after treatment with 5-FU [33]. Other limitations were due to the nature of retrospective studies. Ultimately, a prospective study with a larger sample size on IFNα2b combined with 5-FU will be needed to further validate our results.
Conclusion
Our study with a cohort of 26 patients showed the efficacy of IFNα2b combined with 5-FU in the treatment of OSSN. This regimen reduced the duration of IFNα2b use and the frequency of 5-FU cycles with an increased tumor resolution and low recurrence rate.
References
Hollhumer R, Williams S, Michelow P. Ocular surface squamous neoplasia: population demographics, pathogenesis and risk factors. Afr Vis Eye Health J. 2020;79:1.
Basti S, Macsai MS. Ocular surface squamous neoplasia: a review. Cornea. 2003;22(7):687–704.
Maudgal PC, Van Damme B, Missotten L. Corneal epithelial dysplasia after trifluridine use. Graefes Arch Clin Exp Ophthalmol. 1983;220(1):6–12.
Shields CL, Ramasubramanian A, Mellen PL, Shields JA. Conjunctival squamous cell carcinoma arising in immunosuppressed patients (organ transplant, human immunodeficiency virus infection). Ophthalmology. 2011;118(11):2133–7.
McClellan AJ, McClellan AL, Pezon CF, et al. Epidemiology of ocular surface squamous neoplasia in a veterans affairs population. Cornea. 2013;32(10):1354–8.
Mittal R, Rath S, Vemuganti GK. Ocular surface squamous neoplasia—review of etio-pathogenesis and an update on clinico-pathological diagnosis. Saudi J Ophthalmol. 2013;27(3):177–86.
Afrogheh AH, Jakobiec FA, Hammon R, et al. Evaluation for high-risk HPV in squamous cell carcinomas and precursor lesions arising in the conjunctiva and lacrimal sac. Am J Surg Pathol. 2016;40(4):519–28.
Stone DU, Butt AL, Chodosh J. Ocular surface squamous neoplasia: a standard of care survey. Cornea. 2005;24(3):297–300.
Adler E, Turner JR, Stone DU. Ocular surface squamous neoplasia: a survey of changes in the standard of care from 2003 to 2012. Cornea. 2013;32(12):1558–61.
Xie HT, Zhang YY, Jiang DL, et al. Amniotic membrane transplantation with topical interferon alfa-2b after excision of ocular surface squamous neoplasia. Int J Ophthalmol. 2018;11(1):160–2.
Nanji AA, Sayyad FE, Karp CL. Topical chemotherapy for ocular surface squamous neoplasia. Curr Opin Ophthalmol. 2013;24(4):336–42.
Al Bayyat G, Arreaza-Kaufman D, Venkateswaran N, Galor A, Karp CL. Update on pharmacotherapy for ocular surface squamous neoplasia. Eye Vis (Lond). 2019;6:24.
Chaugule SS, Park J, Finger PT. Topical chemotherapy for giant ocular surface squamous neoplasia of the conjunctiva and cornea: Is surgery necessary? Indian J Ophthalmol. 2018;66(1):55–60.
Gupta A, Muecke J. Treatment of ocular surface squamous neoplasia with Mitomycin C. Br J Ophthalmol. 2010;94(5):555–8.
Joag MG, Sise A, Murillo JC, et al. Topical 5-fluorouracil 1% as primary treatment for ocular surface squamous neoplasia. Ophthalmology. 2016;123(7):1442–8.
Kusumesh R, Ambastha A, Sinha B, Kumar R. Topical interferon alpha-2b as a single therapy for primary ocular surface squamous neoplasia. Asia Pac J Ophthalmol (Phila). 2015;4(5):279–82.
Crooke ST, Bradner WT. Mitomycin C: a review. Cancer Treat Rev. 1976;3(3):121–39.
Khong JJ, Muecke J. Complications of mitomycin C therapy in 100 eyes with ocular surface neoplasia. Br J Ophthalmol. 2006;90(7):819–22.
Longley DB, Harkin DP, Johnston PG. 5-fluorouracil: mechanisms of action and clinical strategies. Nat Rev Cancer. 2003;3(5):330–8.
Armstrong RD, Lewis M, Stern SG, Cadman EC. Acute effect of 5-fluorouracil on cytoplasmic and nuclear dihydrofolate reductase messenger RNA metabolism. J Biol Chem. 1986;261(16):7366–71.
Baron S, Tyring SK, Fleischmann WR, et al. The interferons. Mechanisms of action and clinical applications. JAMA. 1991;266(10):1375–83.
Gutterman JU. Cytokine therapeutics: lessons from interferon alpha. Proc Natl Acad Sci USA. 1994;91(4):1198–205.
Mercado CL, Pole C, Wong J, et al. Surgical versus medical treatment for ocular surface squamous neoplasia: a quality of life comparison. Ocul Surf. 2019;17(1):60–3.
Galor A, Karp CL, Oellers P, et al. Predictors of ocular surface squamous neoplasia recurrence after excisional surgery. Ophthalmology. 2012;119(10):1974–81.
Karp CL, Moore JK, Rosa RH Jr. Treatment of conjunctival and corneal intraepithelial neoplasia with topical interferon alpha-2b. Ophthalmology. 2001;108(6):1093–8.
de Keizer RJ, de Wolff-Rouendaal D, van Delft JL. Topical application of 5-fluorouracil in premalignant lesions of cornea, conjunctiva and eyelid. Doc Ophthalmol. 1986;64(1):31–42.
Hu FR, Wu MJ, Kuo SH. Interferon treatment for corneolimbal squamous dysplasia. Am J Ophthalmol. 1998;125(1):118–9.
Vann RR, Karp CL. Perilesional and topical interferon alfa-2b for conjunctival and corneal neoplasia. Ophthalmology. 1999;106(1):91–7.
Maskin SL. Regression of limbal epithelial dysplasia with topical interferon. Arch Ophthalmol. 1994;112(9):1145–6.
Zarei-Ghanavati S, Alizadeh R, Deng SX. Topical interferon alpha-2b for treatment of noninvasive ocular surface squamous neoplasia with 360 degrees limbal involvement. J Ophthalmic Vis Res. 2014;9(4):423–6.
Schechter BA, Koreishi AF, Karp CL, Feuer W. Long-term follow-up of conjunctival and corneal intraepithelial neoplasia treated with topical interferon alfa-2b. Ophthalmology. 2008;115(8):1291–6.
Parrozzani R, Frizziero L, Trainiti S, et al. Topical 1% 5-fluoruracil as a sole treatment of corneoconjunctival ocular surface squamous neoplasia: long-term study. Br J Ophthalmol. 2017;101(8):1094–9.
Parrozzani R, Lazzarini D, Alemany-Rubio E, Urban F, Midena E. Topical 1% 5-fluorouracil in ocular surface squamous neoplasia: a long-term safety study. Br J Ophthalmol. 2011;95(3):355–9.
Sayed-Ahmed IO, Palioura S, Galor A, Karp CL. Diagnosis and medical management of ocular surface squamous neoplasia. Expert Rev Ophthalmol. 2017;12(1):11–9.
Watane A, Hansen E, Vazquez LE, Karp CL. Ocular surface squamous neoplasia masquerading as recalcitrant epithelial keratitis. Cornea. 2021;2021:5.
Wadler S, Schwartz EL, Goldman M, et al. Fluorouracil and recombinant alfa-2a-interferon: an active regimen against advanced colorectal carcinoma. J Clin Oncol. 1989;7(12):1769–75.
Kelsen D, Lovett D, Wong J, et al. Interferon alfa-2a and fluorouracil in the treatment of patients with advanced esophageal cancer. J Clin Oncol. 1992;10(2):269–74.
Lee KH, Lee JS, Suh C, et al. Combination of 5-fluorouracil and recombinant interferon alpha-2B in advanced gastric cancer. A phase I study. Am J Clin Oncol. 1992;15(2):141–5.
De Filippi R, Cucchiara G, Prete SP, et al. Immuno-chemotherapy of advanced colorectal cancer with alpha-2a interferon and 5-fluorouracil. Immunopharmacol Stud Ann Oncol. 1991;2(10):759–64.
Schwartz EL, Baptiste N, Megati S, Wadler S, Otter BA. 5-Ethoxy-2’-deoxyuridine, a novel substrate for thymidine phosphorylase, potentiates the antitumor activity of 5-fluorouracil when used in combination with interferon, an inducer of thymidine phosphorylase expression. Cancer Res. 1995;55(16):3543–50.
van der Wilt CL, Smid K, Aherne GW, Noordhuis P, Peters GJ. Biochemical mechanisms of interferon modulation of 5-fluorouracil activity in colon cancer cells. Eur J Cancer. 1997;33(3):471–8.
Venkateswaran N, Mercado C, Galor A, Karp CL. Comparison of topical 5-fluorouracil and interferon Alfa-2b as primary treatment modalities for ocular surface squamous neoplasia. Am J Ophthalmol. 2019;199:216–22.
Shah SU, Kaliki S, Kim HJ, et al. Topical interferon alfa-2b for management of ocular surface squamous neoplasia in 23 cases: outcomes based on American Joint Committee on Cancer classification. Arch Ophthalmol. 2012;130(2):159–64.
Shields CL, Kaliki S, Kim HJ, et al. Interferon for ocular surface squamous neoplasia in 81 cases: outcomes based on the American Joint Committee on Cancer classification. Cornea. 2013;32(3):248–56.
Shields CL, Constantinescu AB, Paulose SA, et al. Primary treatment of ocular surface squamous neoplasia with topical interferon alpha-2b: comparative analysis of outcomes based on original tumor configuration. Indian J Ophthalmol. 2021;69(3):563–7.
Thomas BJ, Galor A, Nanji AA, et al. Ultra high-resolution anterior segment optical coherence tomography in the diagnosis and management of ocular surface squamous neoplasia. Ocul Surf. 2014;12(1):46–58.
Shousha MA, Karp CL, Canto AP, et al. Diagnosis of ocular surface lesions using ultra-high-resolution optical coherence tomography. Ophthalmology. 2013;120(5):883–91.
Kieval JZ, Karp CL, Abou Shousha M, et al. Ultra-high resolution optical coherence tomography for differentiation of ocular surface squamous neoplasia and pterygia. Ophthalmology. 2012;119(3):481–6.
Tran AQ, Venkateswaran N, Galor A, Karp CL. Utility of high-resolution anterior segment optical coherence tomography in the diagnosis and management of sub-clinical ocular surface squamous neoplasia. Eye Vis (Lond). 2019;6:27.
Hllhumer R, Williams S, Michelow P. Ocular surface squamous neoplasia: management and outcomes. Eye (Lond). 2021;35(6):1562–73.
Nanji AA, Moon CS, Galor A, et al. Surgical versus medical treatment of ocular surface squamous neoplasia: a comparison of recurrences and complications. Ophthalmology. 2014;121(5):994–1000.
Kusumesh R, Ambastha A, Kumar S, Sinha BP, Imam N. Retrospective comparative study of topical interferon alpha2b versus mitomycin C for primary ocular surface squamous neoplasia. Cornea. 2017;36(3):327–31.
Kaliki S, Bejjanki KM, Desai A, Mohamed A. Interferon Alfa 2b for ocular surface squamous neoplasia: factors influencing the treatment response. Semin Ophthalmol. 2019;34(7–8):465–72.
Acknowledgements
Funding
This study was supported by the National Natural Science Foundation of China (grants: 82070934, 82171025). The journal’s Rapid Service Fee was funded by the authors.
Author Contributions
Conceptualization, W.G. and HT.X; methodology, JS.W. and BJ.S.; formal analysis, W.G.; investigation, HT.X and MC.Z; data curation, W.G.; writing-original draft preparation, W.G.; writing-review and editing, W.G, HT.X. and MC.Z.; supervision, MC.Z.; funding acquisition, MC.Z. All authors have read and agreed to the published version of the manuscript.
Disclosures
Wen Geng, Jia-Song Wang, Bing-Jie Shi, Hua-Tao Xie, and Ming-Chang Zhang have nothing to disclose.
Compliance with Ethics Guidelines
This retrospective study was approved (UHCT22048) by the Ethics Committee of Wuhan Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, and the study was conducted in accordance with the tenets of the Declaration of Helsinki. The institutional review board waived the requirement for informed consent owing to the retrospective nature of the study.
Data Availability
Data from this study are available upon reasonable request to the corresponding author.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Geng, W., Wang, JS., Shi, BJ. et al. Topical Chemotherapy for Treating Ocular Surface Squamous Neoplasia with a Combination of Interferon α-2b and 5-Fluorouracil. Ophthalmol Ther 11, 1563–1576 (2022). https://doi.org/10.1007/s40123-022-00535-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40123-022-00535-6