Abstract
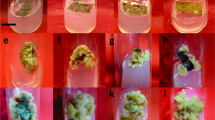
Five genotypes of ginger cultured in vitro produced calli and microrhizomes, and they were evaluated for pungency principles at different induction periods using HPLC. Differentiation and growth of microrhizomes and calli and yield of gingerols were found genotype dependent. Accumulation of oleoresin, gingerols and shogaol increased with time duration, reaching the highest after 3 months of induction. The microrhizomes and calli of genotype cv. Aswathy yielded the highest gingerols throughout the three-month induction period. However, microrhizomes accumulated 50 times (average 26.2 times) higher total gingerols compared to calli, irrespective of the genotypes. This study proved the potential of microrhizomes for in vitro gingerol synthesis. It is suggested that large-scale production of microrhizomes can partially substitute the conventional field growing of ginger for harvesting of high-quality clean gingerols and shogaol with no residue of the pesticides used during the field growing.





Similar content being viewed by others
References
Srinivasan S (2017) Ginger rhizomes (Zingiber officinale): a spice with multiple health beneficial potentials. Pharma Nutr 5(1):18–28. https://doi.org/10.1016/j.phanu.2017.01.001
Ahumada MCR, Timmermann BN, Gang DR (2006) Biosynthesis of curcuminoids and gingerols in turmeric (Curcuma longa) and ginger (Zingiber officinale): identification of curcuminoid synthase and hydroxycinnamoyl-CoA thioesterases. Phytochemistry 67:2017–2029. https://doi.org/10.1016/j.phytochem.2006.06.028
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15(3):473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Sharma TR, Singh BM (1995) In vitro microrhizome production in Zingiber officianale (Rosc). Plant Cell Rep 15:274–277. https://doi.org/10.1007/BF00193735
Cousins MM, Adelberg JW, Chen F, Rieck J (2007) Antioxidant capacity of fresh and dried rhizomes from four clones of turmeric (Curcuma longa L.) grown in vitro. Ind Crops Prod 25:129–135. https://doi.org/10.1016/j.indcrop.2006.08.004
Swarnathilaka DBR, Kottearachchi NS, Weerakkody WJSK (2016) Factors affecting on induction of microrhizomes in ginger (Zingiber officinale Rosc.), cultivar local from Sri Lanka. Biotechnol J Int 12(2): 23903 doi:https://doi.org/10.9734/BBJ/2016/23903
An NH, Chien TTM, Nhi HTH, Nga NTM, Phuc TT, Thuy LTN, Thanh TVB, Nguyen PTT, Phuong TTB (2020) The effects of sucrose, silver nitrate, plant growth regulators, and ammonium nitrate on microrhizome induction in perennially-cultivated ginger (Zingiber officinale Roscoe) from Hue, Vietnam. Acta Agrobot 73(2):7329. https://doi.org/10.5586/aa.7329
Zahid NA, Jaafar HZE, Hakiman M (2021) Alterations in microrhizome induction, shoot multiplication and rooting of ginger (Zingiber officinale Roscoe) var. Bentong with regards to sucrose and plant growth regulators application. Agronomy 11: 320. doi:https://doi.org/10.3390/agronomy11020320
Paul R, Shylaja MR (2010) Indirect organogenesis in ginger. Indian J Hortic 67:513–517
Shylaja MR, Parvathy KI, Lazer L (2016) Microrhizome technology for large scale production of disease free seed rhizomes of ginger (Zingiber officinale Rosc.). Indian J Arecanut Spices Med Plants 19:6–9
Shylaja MR, Sabitha KR, Kumar SP, Narayanankutty C, Narayanankutty MC, Mathew D (2018) Production technology for in vitro induced microrhizomes of ginger in high-tech polyhouse. Indian J Arecanut Spices Med Plants 20:3–7. https://doi.org/10.2137/ssrn.3577809
El-Nabarawy MA, El-Kafafi SH, Hamza MA, Omar MA (2015) The effect of some factors on stimulating the growth and production of active substances in Zingiber officinale callus cultures. Ann Agric Sci 60:1–9. https://doi.org/10.1016/j.aoas.2014.11.020
Aly UI, Abbas MS, Taha HS, Gaber EI (2013) Characterization of 6-gingerol for in vivo and in vitro ginger (Zingiber officinale) using high performance liquid chromatography. Global J Bot Sci 1:1–8. https://doi.org/10.12974/2311-858X.2013.01.01.2
Marfori E, Jane CM (2018) Influence of sucrose on growth and [6]-gingerol production of in vitro-grown ginger (Zingiber officinale Rosc). Int J Pharmacogn Phytochem Res 10:17–20. https://doi.org/10.25258/phyto.v10i01.11926
Paul R, Shylaja MR (2012) Field evaluation of tissue culture raised somaclones of ginger (Zingiber officinale) for productivity-linked traits. J Med Aromatic Plant Sci 34:20–27
Snedecor GW, Cochran WG (1956) Statistical methods applied to experiments in agriculture and biology, 5th edn. Iowa State University Press, Ames
Kambaska KB, Santilata S (2009) Effect of plant growth regulator on micropropagation of ginger (Zingiber officinale Rosc.) cv-Suprava and Suruchi. J Agric Technol 5:271–280
Archana CP, Pillai GS, Balachandran I (2013) In vitro microrhizome induction in three high yielding cultivars of Zingiber officinale Rosc. and their phytopathological analysis. Int J Adv Biotechnol Res 4:296–300
Gahan PB, George EF (2008) Adventitious regeneration. Plant propagation by tissue culture. In: George MA, Klerk GD (eds) Plant propagation by tissue culture, 3rd edn. Springer, New York, pp 377–383
Looney NE, Taylor JS, Pharis RP (1988) Relationship of endogenous gibberellin and cytokinin levels in shoot tip to apical form in four strains of ‘McIntosh’ apple. J Am Soc Hortic Sci 113:395–398
Marino G (1988) In vitro (14C) labelled 6-benzyladenine uptake and 14CO2 evolution in two Japanese plum cultivars. Plant Cell Tiss Org Cult 13:49–59. https://doi.org/10.1007/BF00043046
Jean M, Cappadocia M (1991) In vitro tuberization in Dioscorea alata L. ‘Brazo fuerte’ and ‘Florida’ and D. Abbssinica Hoch. Plant Cell Tiss Org Cult 26:147–152. https://doi.org/10.1007/BF00039936
George EF (1993) Plant propagation by tissue culture Part 1. In: The Technology, 2nd edn. Exegetics Ltd, England, pp 3468
Purseglove JW, Brown EG, Green CL, Robins SRJ (1981) Spices. Longman, London and New York, pp 447–813
Pawar N, Pai S, Nimbalkar M, Dixit G (2015) RP-HPLC analysis of phenolic antioxidant compound 6-gingerol from in vitro cultures of Zingiber officinale Rosc. Plant Sci Today 2:24–28. https://doi.org/10.14719/pst.2015.2.1.103
Ghasemzadeh A, Jaafar HZ, Ashkani S, Rahmat A, Juraimi AS, Puteh A, Mohamed MTM (2016) Variation in secondary metabolite production as well as antioxidant and antibacterial activities of Zingiber zerumbet (L.) at different stages of growth. BMC Compl Med Ther 16:104 doi:https://doi.org/10.1186/s12906-016-1072-6
Zarate R, Yeoman M (1994) Studies of the cellular localization of the phenolic pungent principle of ginger, Zingiber officinale Roscoe. New Phytol 126:295–300. https://doi.org/10.1111/j.1469-8137.1994tb03948.x
Arijanti S, Suryaningsih DR (2019) Biosynthesis of secondary metabolites (gingerol, shogaol, and zingerone) from callus of three ginger varieties. Drug Invent Today 11:496–500
Janarthanam M, Gopalakrishnan B, Sekar T (2010) Secondary metabolite production in callus cultures of Stevia rebaudiana Bertoni. Bangladesh J Sci Ind Res 45:243–248. https://doi.org/10.3329/bjsir.v45i3.6532
Dehghani I, Mostajeran A, Asghari G (2011) In vitro and in vivo production of gingerols and zingiberene in ginger plant (Zingiber officinale Roscoe). Iran J Pharmaceut Res 7(2):117–121
Funding
Moved to title page for double-blind peer review.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Availability of Data and Material
Moved to title page for double-blind peer review.
Code availability
Not applicable.
Consent to Participate
All authors have read the content of the manuscript consent to participate in the publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Significance statement: This paper details viable and alternate strategy for the clean synthesis of industrially important gingerols and shogaol. The protocols and varietal, explant and maturity factors, for the maximum in vitro recovery of these molecules, are discussed.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Rani, M., Shylaja, M.R., Mathew, D. et al. Potential of Microrhizomes for In Vitro Gingerol and Shogaol Synthesis in Ginger (Zingiber officinale Rosc.). Proc. Natl. Acad. Sci., India, Sect. B Biol. Sci. 92, 121–129 (2022). https://doi.org/10.1007/s40011-021-01314-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40011-021-01314-2