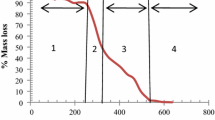
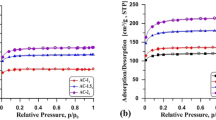
Abstract
The magnetization process of activated carbons prepared from coffee residue was performed according to two protocols: The first one involved a chemical impregnation with a mixture of two salts: FeSO4·7H2O and FeCl3, followed by a treatment with NaOH (2 M). The second procedure required an impregnation with both salts mentioned above, taken alone or in mixture, followed by a pyrolysis at 600 °C during an hour. A commercial activated carbon treated in the same way was used as reference. Physicochemical analysis of all materials revealed that activated carbon treated with iron sulfate (FeSO4·7H2O) alone contains magnetite; activated carbon prepared with iron chloride (FeCl3) alone contains magnetite, while activated carbon prepared with a combination of both salts contains magnetite and maghemite. Adsorption tests for the sulfamethazine removal from the aqueous phase were carried out at pH = 6 and pH = 4. Magnetized carbon prepared with an impregnation ratio of 75% iron sulfate followed by pyrolysis gave the most effective performance with an elimination efficiency of 64.12% of sulfamethazine.










Similar content being viewed by others

References
Ai L, Zhang C, Chen Z (2011) Removal of methylene blue from aqueous solution by a solvothermal-synthesized grapheme/magnetite composite. J Hazard Mater 192:1515–1524. https://doi.org/10.1016/j.jhazmat.2011.06.068
Alcamo J, Henrichs T, Rösch T (2000) Water in 2025: global modeling and scenario analysis for the World Commission for the 21st century. Report A0002. Center for Environmental Systems Research, University of Kassel, Germany 48. http://www.env-edu.gr/Documents/World%20Water%20in%202025.pdf
Altintig E, Altundag H, Tuzen M, Sarı A. (2017) Effective removal of methylene blue from aqueous solutions using magnetic loaded activated carbonas novel adsorbent. Chem Eng Res and Des 122:151-163
Boudrahem F, Soualah A, Aissani-Benissad F (2011) Pb (II) and Cd(II) removal from aqueous solutions using activated carbon developed from coffee residue activated with phosphoric acid and zinc chloride. J Chem Eng Data 56:1946–1955. https://doi.org/10.1021/je1009569
Cazetta AL, Pezoti O, Bedin KC, Silva TL, Paesano Junior A, Asefa T, Almeida VC (2016) Magnetic activated carbon derived from biomass waste by concurrent synthesis: efficient adsorbent for toxic dyes. ACS Sustain Chem Eng 4:1058–1068. https://doi.org/10.1021/acssuschemeng.5b01141
Chung WJ, Chun SY, Kim SS, Chang SW (2016) Photocatalytic removal of tetracycline using TiO2/Ge composite optimized by response surface methodology (RSM). J Ind Eng Chem 36:320–325. https://doi.org/10.1016/j.jiec.2016.02.022
Danalıoğlu ST, Bayazit SS, Kerkez Kuyumcu Ö, Salam MA (2017) Efficient removal of antibiotics by a novel magnetic adsorbent: magnetic activated carbon/chitosan (MACC) nanocomposite. J Mol Liq 240:589–596. https://doi.org/10.1016/j.molliq.2017.05.131
Davoud B, Fatemeh B, Zaccheus S, Nehad JA (2020) Adsorption properties of thermally treated rice husk for removal of sulfamethazine antibiotic from pharmaceutical wastewater. J Pharm Res Int 32:84–92
Demiral İ, Şamdan CA (2016) Preparation and characterisation of activated carbon from pumpkin seed shell using H3PO4. Anadolu Univ J Sci Technology A Appl Sci Eng 17:125–138
Ijaz H, Li Y, Qi J, Li J, Sun X, Shen J, Han W, Wang L (2018) Synthesis of magnetic yolk-shell mesoporous carbon architecture for the effective adsorption of sulfamethazine drug. Microporous Mesoporous Mater 255:110–118. https://doi.org/10.1016/j.micromeso.2017.07.027
Jagtoyen M, Thwaites M, Stencel J, Mc Enaney B, Derbyshire F (1993) Activated carbons from bituminous coals by reaction with H3PO4: the influence of coal cleaning. Fuel Process Technol 34:85–96. https://doi.org/10.1016/0378-3820(93)90093-J
Jia MY, Wang F, Bian YR, Stedtfeld RD, Liu GX, Yu JP, Jiang X (2018) Sorption of sulfamethazine to biochars as affected by dissolved organic matters of different origin. Bioresour Technol 248:36–43. https://doi.org/10.1016/j.biortech.2017.08.082
Juang RS, Yei YC, Liao CS, Lin KS, Lu HC, Wang SF, Sun AC (2018) Synthesis of magnetic Fe3O4/activated carbon nanocomposites with high surface area as recoverable adsorbents. J Taiwan Inst Chem Eng 90:51–60. https://doi.org/10.1016/j.jtice.2017.12.005
Kanari N, Nour-Eddine M, Etleva O, Seit S, Frederic D, Eric A, Jacques Y (2018) Thermal behavior of hydrated iron sulfate in various atmospheres. Metals 8:1084–1093. https://doi.org/10.3390/met8121084
Khenniche L, Aissani F (2008) Adsorption de l’acide salicylique sur un charbon actif préparé à partir d’un déchet solide et un charbon commercial. Algerian J Adv Mater 5:419–422
Khenniche L, Aissani F (2009a) Preparation and characterization of carbons from coffee residue: adsorption of salicylic acid on the prepared carbons. J Chem Eng Data 55:728–734. https://doi.org/10.1021/je900426a
Khenniche L, Aissani F (2009b) Characterization and utilization of activated carbons prepared from coffee residue for adsorptive removal of salicylic acid and phenol: kinetic and isotherm study. Desalin Water Treat 11:1–12. https://doi.org/10.5004/dwt.2009.801
Khenniche L, Aissani F (2010) Adsorptive removal of phenol by coffee residue activated carbon and commercial activated carbon: equilibrium, kinetics, and thermodynamics. J Chem Eng Data 55:4677–4686. https://doi.org/10.1021/je100302e
Kurade MB (2019) Toxicity of sulfamethazine and sulfamethoxazole and their removal by a green microalga, Scenedesmus obliquus. Chemosphere 218; 551-558.
Kyzas GZ, Deiyanni EA, Lazaridis NK (2014) Magnetic modification of microporous carbon for dye adsorption. J Colloid Interface Sci 430:166–173. https://doi.org/10.1016/j.jcis.2014.05.049
Lamine MS, Ridha C, Mahfoud HM, Mouad C, Lotfi B, Al-Dujaili HA (2014) Chemical activation of an activated carbon prepared from coffee residue. Energy Procedia 50:393–400. https://doi.org/10.1016/j.egypro.2014.06.047
Liu Y, Liu X, Dong W, Zhang L, Kong Q, Wang W (2017) Efficient adsorption of sulfamethazine onto modified activated carbon: a plausible adsorption mechanism. Sci Rep 7:12437. https://doi.org/10.1038/s41598-017-12805-6
Livani MJ, GhorbaniM MH (2018) Preparation of an activated carbon from hazelnut shells and its hybrids with magnetic NiFe2O4 nanoparticles. New Carbon Mater 33:578–586. https://doi.org/10.1016/s1872-5805(18)60358-0
Lu S, Liu Y, Feng L, Sun Z, Zhang L (2018) Characterization of ferromagnetic sludge-based activated carbon and its application in catalytic ozonation of p-chlorobenzoic acid. Environ Sci Pollut Res Int 25:5086–5094. https://doi.org/10.1007/s11356-017-8680-7
Lua AC, Yang T (2004) Effect of activation temperature on the textural and chemical properties of potassium hydroxide activated carbon prepared from pistachio-nut shell. J Colloid Interface Sci 274:594–601. https://doi.org/10.1016/j.jcis.2003.10.001
Mohan D, Sarswat A, Singh VK, Alexandre FM, Pittman CU Jr (2011) Development of magnetic activated carbon from almond shells for trinitrophenol removal from water. Chem Eng J 172:1111–1125. https://doi.org/10.1016/j.cej.2011.06.054
Olorundare OF, Msagati TAM, Krause MRW, Okonkwo JO, Mamba BB (2014) Activated carbon from lignocellulosic waste residues: effect of activating agent on porosity characteristics and use as adsorbents for organic species. Water Air Soil Pollut 225:1876–1890. https://doi.org/10.1007/s11270-014-1876-2
Philippova O, Barabanova A, Molchanov V, Khokhlov A (2011) Magnetic polymerbeads: recent trends and developments in synthetic design and applications. Eur Polym J 47:542–559. https://doi.org/10.1016/j.2010.11.006
Ratola N, Alessandra C, Arminda A, Athanasios K (2012) Occurrence of organic microcontaminants in the wastewater treatment process. A mini review. J Hazard Mater 239:1–18. https://doi.org/10.1016/j.jhazmat.2012.05.040
Reinoso R, Molina-Sabio M (2004) Role of chemical activation in the development of carbon porosity. Colloid Surf A-Physicochem Eng Asp 241:15–25. https://doi.org/10.1016/j.colsurfa.2004.04.007
Rivera-Utrilla J, Bautista-Toledo I, Ferro-Garcia MA, Moreno-Castilla C (2001) Activated carbon surface modifications by adsorption of bacteria and their effect on aqueous lead adsorption. J Chem Technol Biotechnol 76:1209–1215. https://doi.org/10.1002/jctb.506
Saleh TA, Naeemullah TM, Sari A (2017) Polyethylenimine modified activated carbon as novel magnetic adsorbent for the removal of uranium from aqueous solution. Chem Eng Res Des 117:218–227. https://doi.org/10.1016/J.CHERD.2016.10.030
Shah I, Adnan R, Ngah WSW, Mohomed N (2015) Iron impregnated carbon materials with improved physicochemical characteristics. Mater Sci Eng B 201:1–12. https://doi.org/10.1016/j.mseb.2015.07.004
Singh AP, Mishra M, Sambyal P, Gupta BK, Singh BP, Chandra A, Dhawan SK (2014) Encapsulation of γ-Fe2O3 decorated reduced graphene oxide in polyaniline core–shell tubes as an exceptional tracker for electromagnetic environmental pollution. J Mater Chem A 2:3581–3593. https://doi.org/10.1039/C3TA14212D
Stacia RWU, Divina AGN, Lisa Z, Diana SA (2014) Assessing antibiotic sorption in soil: a literature review and new case studies on sulfonamides and macrolides. Chem Cent 8:1–12. https://doi.org/10.1186/1752-153X-8-5
Tzeng TW, Liu YT, Deng YY, Hsieh C, Tan CC, Wang SL, Huang ST, Tzou YM (2016) Removal of sulfamethazine antibiotics using cow manure-based carbon adsorbents. Int J Environ Sci Techno 13:973–984. https://doi.org/10.1007/s13762-015-0929-4
Wong KT, Eu NC, Ibrahim S, Kim H, Yoon Y, Jang M (2016) Recyclable magnetite-loaded palm shell-waste based AC for the effective removal of methyl blue from aqueous solution. J Clean Prod 115:337–342. https://doi.org/10.1016/j.jclepro.2015.12.063
Xiangdong Z, Feng Q, Yuchen L, Shicheng Z, Jianmin C (2015) Environmental performances of hydrochar-derived magnetic carbon composite affected by its carbonaceous precursor. RSC Adv 5:60713–60722. https://doi.org/10.1039/C5RA07339A
Yan Q, Zhang Z, Zhang Y, Umar A, Guo Z, O’Hare D, Wang Q (2015) Hierarchical Fe3O4 core-shell layered double hydroxide composites as magnetic adsorbents for an- ionic dye removal from wastewater. Eur J Inorg Chem 25:4182–4191. https://doi.org/10.1002/ejic.201500650
Yang W, Zheng F, Xue X (2011) Investigation into adsorption mechanisms of sulfonamides onto porous adsorbents. J Colloid Interface Sci 362:503–509. https://doi.org/10.1016/j.jcis.2011.06.071
Yin Y, Zhou S, Min C, Wu L (2011) Preparation of rattle-type magnetic mesoporous carbon spheres and their highly efficient adsorption and separation. J Colloid Interface Sci 361:527–533. https://doi.org/10.1016/j.jcis.2011.05.014
Ying L, Xiaohui L, Wenping D, Lingli Z, Qiang K, Weiliang W (2017) Efficient adsorption of sulfamethazine onto modified activated carbon: a plausible adsorption mechanism. Sci Rep 7:12437. https://doi.org/10.1038/s41598-017-12805-6
Yu J, Yang M, Lin TF, Guo Z, Zhang Y, Gu J, Zhang S (2007) Effects of surface characteristics of activated carbon on the adsorption of 2-methylisobornel (MIB) and geosmin from natural water. Sep Purif Technol 56:363–370. https://doi.org/10.1016/j.seppur.2007.01.039
Zhang J (2011) Preparation and characterization of magnetic coal-based activated carbon in the presence of Fe3O4. Adv Mater Res 393–395:1355–1358. https://doi.org/10.4028/www.scientific.net/AMR.393-395.1355
Zhang S, Liaoa F, Wanga Y, Li M, Meng L, Jiang J (2011) Removal of methylene blue from aqueous solution with magnetite loaded multi-wall carbon nanotube: kinetic, isotherm and mechanism analysis. J Hazard Mater 198:282–290. https://doi.org/10.1016/j.jhazmat.2011.10.041
Zhang M, Zhao Y, Wang J, Bai J, Li K (2021) Removal of sulfamethazine by corn biochars from aqueous solution: sorption mechanisms and efficiency. J Ocean Univ China 20:590–598. https://doi.org/10.1007/s11802-021-4412-1
Zhu X, Qian F, Liu Y, Zhang S, Chen J (2015) Environmental performances of hydrochar-derived magnetic carbon composite affected by its carbonaceous precursor. R Soc Chem Adv 5:60713–60722. https://doi.org/10.1039/C5RA07339A
Acknowledgements
We gratefully acknowledge the help received from Pr. F. BOUDRAHAM (Professor of Process Engineering, University of Bejaia) for the manuscript corrections and Dr. S. ZIANI (Professor of Process Engineering, University of BOUIRA, ALGERIA) for the spectral analysis of the adsorbents. The authors are thankful to Miss BRADAI and Miss BENAMOR for providing facilities to realize all the solutions analysis. The authors are also grateful to Mr BENIDER and Mr MAMAS to have performed the structural and thermal analyses for the different prepared adsorbents.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Human or animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Editorial responsibility: Xu Han.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
khenniche, l., Saidou Souleymane, M., Chemache, Z. et al. Single-step and two-step syntheses of magnetic carbons from coffee residue: elimination of sulfamethazine by adsorption. Int. J. Environ. Sci. Technol. 20, 755–768 (2023). https://doi.org/10.1007/s13762-022-04116-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-022-04116-9