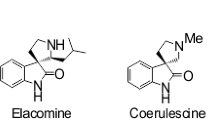
Abstract
Among isatin-based heterocyclic, spirooxindoles are interesting synthetic molecules because of their variety of pharmacological activities. There is a wide range of multi-component reactions that cover this topic. This review covers the different synthesis methods of spirooxindoles reported by Iranian researchers in the last 15 years.
Graphic abstract




























































































Similar content being viewed by others
Abbreviations
- [Bmim]Br:
-
1-Butyl-3-methylimidazolium Bromide
- [BMIM]Cl:
-
1-Butyl-3-methylimidazolium chloride
- [BmIm]PF6:
-
1-Butyl-3-methylimidazolium hexafluorophosphate
- [Bmim]OH:
-
1-Butyl-3-methylimidazolium hydroxide
- TMGTf:
-
N,N,N,N-Tetramethyl guanidinium triflate
- NPs:
-
Nanoparticles
- IFB:
-
Interrupted Feist–Bénary
- p-TSA:
-
p-Toluenesulfonic acid
- Alum:
-
KAl(SO4)2·12H2O
- KF/CP:
-
KF/clinoptilolite
- CSA:
-
Cellulose sulfuric acid
- DMA:
-
3,4-Dimethylaniline
- DIEA:
-
Diisopropylethylamine
- THAM:
-
Tris-hydroxymethylaminomethane
- DMAP:
-
4-Dimethylaminopyridine
- PEG-400:
-
Polyethylene glycol 400
- DES:
-
Deep-Eutectic solvent
- DCDBTSD:
-
N,2-Dibromo-6-chloro-3,4-dihydro-2H-benzo[e][1, 2, 4] thiadiazine-7-sulfonamide 1,1-dioxide
- COS@β-CD:
-
Calcined oyster shell functionalized with taurine immobilized on β-cyclodextrin
- PVI:
-
Poly (vinyl imidazole
References
S.N. Pandeya, R. Kumar, A.K. Pathak, G. Nath, Pharm. Chem. 2, 257 (2010)
A. Singh, K. Raghuwanshi, V.K. Patel, D.K. Jain, R. Veerasamy, A. Dixit, H. Rajak, Pharm. Chem. J. 51, 366 (2017)
R. Meleddu, S. Distinto, A. Corona, E. Tramontano, G. Bianco, C. Melis, F. Cottiglia, E. Maccioni, J. Enzyme Inhib. Med. Chem. 32, 130 (2017)
C. Xie, L.-M. Tang, F.-N. Li, L.-P. Guan, C.-Y. Pan, S.-H. Wang, Med. Chem. Res. 23, 2161 (2014)
P.K. Sharma, S. Balwani, D. Mathur, S. Malhotra, B.K. Singh, A.K. Prasad, C. Len, E.V. Van der Eycken, B. Ghosh, N.G. Richards, J. Enzyme Inhib. Med. Chem. 31, 1520 (2016)
B.K. Paul, D. Ray, N. Guchhait, Phys. Chem. Chem. Phys. 15, 1275 (2013)
G.M. Ziarani, R. Moradi, N. Lashgari, Ark. Online J. Organ. Chem. i, 1 (2016)
R. Kakkar, MedChemComm 10, 351 (2019)
B. Bhrigu, D. Pathak, N. Siddiqui, M.S. Alam, W. Ahsan, Int. J. Pharm. Sci. Drug Res. 2, 229 (2010)
N. Lashgari, G.M. Ziarani, Ark. Online J. Organ. Chem. i, 277 (2012)
H. Tinh Tran, N. White, T.N. Thanh Nguyen, H. Toa Nhu, D.T. Phung, J. Tarning, F. Nosten, B. Magnusson, J. Prakash Jain, and K. Hamed, Antimicrob. Agents Chemother. 61, e01940 (2016)
Y. Li, Q. Niu, T. Wei, T. Li, Anal. Chim. Acta 1049, 196 (2019)
Y.-C. Tsai, J.-P. Liou, R. Liao, C.-Y. Cheng, P.-L. Tao, Bioorgan. Med. Chem. Lett. 8, 1813 (1998)
R. Rojas-Duran, G. González-Aspajo, C. Ruiz-Martel, G. Bourdy, V.H. Doroteo-Ortega, J. Alban-Castillo, G. Robert, P. Auberger, E. Deharo, J. Ethnopharmacol. 143, 801 (2012)
T.-H. Kang, K. Matsumoto, M. Tohda, Y. Murakami, H. Takayama, M. Kitajima, N. Aimi, H. Watanabe, Eur. J. Pharmacol. 444, 39 (2002)
Y. Zhao, S. Yu, W. Sun, L. Liu, J. Lu, D. McEachern, S. Shargary, D. Bernard, X. Li, T. Zhao, P. Zou, D. Sun, S. Wang, J. Med. Chem. 56, 5553 (2013)
F. Wong, P. Gines, H. Watson, Y. Horsmans, P. Angeli, P. Gow, P. Minini, M. Bernardi, J. Hepatol. 53, 283 (2010)
R. Moradi, G.M. Ziarani, N. Lashgari, Organ. Chem. i, 148 (2017)
M. Flores, J. Pena, P. García García, N.M. Garrido, D. Diez, Curr. Organ. Chem. 17, 1957 (2013)
N.R. Ball-Jones, J.J. Badillo, A.K. Franz, Organ. Biomol. Chem. 10, 5165 (2012)
Y. Liu, H. Wang, J. Wan, Asian J. Organ. Chem. 2, 374 (2013)
G.M. Ziarani, R. Moradi, N. Lashgari, Tetrahedron 74, 1323 (2018)
M.A. Borad, M.N. Bhoi, N.P. Prajapati, H.D. Patel, Synth. Commun. 44, 897 (2014)
M.A. Borad, M.N. Bhoi, N.P. Prajapati, H.D. Patel, Synth. Commun. 44, 1043 (2014)
G.S. Singh, Z.Y. Desta, Chem. Rev. 112, 6104 (2012)
P. Pakravan, S. Kashanian, M.M. Khodaei, F.J. Harding, Pharmacol. Rep. 65, 313 (2013)
K. Rad-Moghadam, L. Youseftabar-Miri, Tetrahedron 67, 5693 (2011)
K. Rad-Moghadam, L. Youseftabar-Miri, J. Fluor. Chem. 135, 213 (2012)
H. Hosseinjani-Pirdehi, K. Rad-Moghadam, L. Youseftabar-Miri, Tetrahedron 70, 1780 (2014)
K. Rad-Moghadam, L. Youseftabar-Miri, Synlett 2010, 1969 (2010)
L. Youseftabar-Miri, H. Hosseinjani-Pirdehi, Asian J. Green Chem. 1, 56 (2017)
K. Rad-Moghadam, L. Youseftabar-Miri, ARKIVOC 11, 43 (2011)
J. Azizian, A.V. Morady, S. Soozangarzadeh, A. Asadi, Tetrahedron Lett. 43, 9721 (2002)
L. Faraji, H. Arvinnezhad, N. Alikami, K. Jadidi, Lett. Organ. Chem. 7, 472 (2010)
Y. Sarrafi, M. Sadatshahabi, K. Alimohammadi, Green Chem. 13, 114 (2011)
S.J.T. Rezaei, M.R. Nabid, A. Yari, S.W. Ng, Ultrason. Sonochem. 18, 49 (2011)
M.J. Taghizadeh, K. Jadidi, Q. Iran, Chem. Commun. 3, 114 (2015)
M.J. Taghizadeh, A. Javidan, S. Keshipour, Chem. Heterocycl. Compd. 51, 467 (2015)
M.J. Taghizadeh, K. Jadidi, Iran. Chem. Commun. 3, 57 (2015)
M.J. Taghizadeh, H. Arvinnezhad, S. Samadi, K. Jadidi, A. Javidan, B. Notash, Tetrahedron Lett. 53, 5148 (2012)
M. Kiamehr, M.R. Khodabakhshi, F.M. Moghaddam, A. Villinger, P. Langer, Organ. Chem. v, 20 (2017)
Y. Sarrafi, M. Hamzehloueian, K. Alimohammadi, S. Yeganegi, J. Mol. Struct. 1030, 168 (2012)
M. Ghandi, A. Taheri, A. Abbasi, Tetrahedron 66, 6744 (2010)
K. Alimohammadi, Y. Sarrafi, B. Rajabpour, C. R. Chim. 17, 156 (2014)
A. Alizadeh, A. Roosta, M. Halvagar, ChemistrySelect 4, 71 (2019)
M.R. Mohammadizadeh, ARKIVOC 11, 47 (2006)
R. Meghyasi, J. Safaei-Ghomi, M.A. Sharif, J. Chem. Res. 40, 397 (2016)
A. Alizadeh, N. Zohreh, Synlett 2012, 428 (2012)
N. Zohreh, A. Alizadeh, ACS Comb. Sci. 15, 278 (2013)
A. Khazaei, M.A. Zolfigol, F. Karimitabar, I. Nikokar, A.R. Moosavi-Zare, RSC Adv. 5, 71402 (2015)
S.M. Sadeghzadeh, M.A. Nasseri, Catal. Today 217, 80 (2013)
M. Divar, S. Khabnadideh, R. Sabet, K. Zomorodian, N. Ershadi, F. Hassanpour, Chem. Methodol. 3, 237 (2019)
A.A. Esmaeili, M. Darbanian, Tetrahedron 59, 5545 (2003)
X.-J. Zou, L.-H. Lai, G.-Y. Jin, Z.-X. Zhang, J. Agric. Food Chem. 50, 3757 (2002)
H. Khalilullah, M.J. Ahsan, M. Hedaitullah, S. Khan, B. Ahmed, Mini Rev. Med. Chem. 12, 789 (2012)
A. Alizadeh, L. Moafi, Helv. Chim. Acta 99, 457 (2016)
M. Mehrdad, L. Faraji, K. Jadidi, P. Eslami, H. Sureni, Monatshefte Für Chem. Chem. Mon. 142, 917 (2011)
H. Yazdani, A. Bazgir, Synthesis 51, 1669 (2019)
R. Baharfar, S. Asghari, F. Zaheri, N. Shariati, C. R. Chim. 20, 359 (2017)
R. Baharfar, D. Zareyee, S.L. Allahgholipour, Appl. Organomet. Chem. 33, e4805 (2019)
A. Yazdani-Elah-Abadi, N. Simin, R. Morekian, H. Heydari-Dahoei, Polycycl. Aromat. Compd. 39, 1 (2019)
Z. Rashid, T. Moadi, R. Ghahremanzadeh, New J. Chem. 40, 3343 (2016)
S.T. Roudsari, K. Rad-Moghadam, H. Hosseinjani-Pirdehi, Appl. Organomet. Chem. 33, e4993 (2019)
H. Sepehrmansourie, M. Zarei, R. Taghavi, M.A. Zolfigol, ACS Omega 4, 17379 (2019)
M. Dabiri, Z.N. Tisseh, M. Bahramnejad, A. Bazgir, Ultrason. Sonochem. 18, 1153 (2011)
M. Dabiri, Z. Noroozi Tisseh, M. Nobahar, A. Bazgir, Helv. Chim. Acta 94, 824 (2011)
S.H. Ghorbani, M.M. Hashemi, Orient. J. Chem. 31, 181 (2015)
G. Imani Shakibaei, A. Feiz, H. Reza Khavasi, A. Abolhasani Soorki, A. Bazgir, ACS Comb. Sci. 13, 96 (2010)
B. Mirhosseini-Eshkevari, M.A. Ghasemzadeh, M. Esnaashari, Appl. Organomet. Chem. 33, e5027 (2019)
S. Ahadi, R. Ghahremanzadeh, P. Mirzaei, A. Bazgir, Tetrahedron 65, 9316 (2009)
H. Naeimi, Z. Rashid, A.-H. Zarnani, R. Ghahremanzadeh, New J. Chem. 38, 5527 (2014)
K. Jadidi, R. Ghahremanzadeh, P. Mirzaei, A. Bazgir, J. Heterocycl. Chem. 48, 1014 (2011)
R. Baharfar, R. Azimi, J. Chem. Sci. 127, 1389 (2015)
R. Ghahremanzadeh, Z. Rashid, A.H. Zarnani, H. Naeimi, Appl. Catal. Gen. 467, 270 (2013)
A.R. Khorrami, P. Kiani, A. Bazgir, Monatshefte Für Chem. Chem. Mon. 142, 287 (2011)
A. Khalafi-Nezhad, S. Mohammadi, ACS Comb Sci. 15, 512 (2013)
G. Mohammadi Ziarani, F. Aleali, N. Lashgari, A. Badiei, Iran. J. Chem. Chem. Eng. IJCCE 35, 17 (2016)
R. Ghahremanzadeh, Z. Rashid, A.-H. Zarnani, H. Naeimi, Dalton Trans. 43, 15791 (2014)
S.A. Shirvan, R. Ghahremanzadeh, M.M. Moghaddam, A. Bazgir, A.H. Zarnani, M.M. Akhondi, J. Heterocycl. Chem. 49, 951 (2012)
A. Rahmati, Z. Khalesi, Tetrahedron 68, 8472 (2012)
M. Esmaeilpour, J. Javidi, M. Divar, J. Magn. Magn. Mater. 423, 232 (2017)
A. Khalafi-Nezhad, M. Divar, F. Panahi, RSC Adv. 5, 2223 (2015)
M. Esmaeilpour, A.R. Sardarian, H. Firouzabadi, ChemistrySelect 3, 9236 (2018)
R. Ghahremanzadeh, Z. Rashid, A.-H. Zarnani, H. Naeimi, RSC Adv. 4, 43661 (2014)
A. Alizadeh, L. Moafi, Helv. Chim. Acta 98, 546 (2015)
K. Jadidi, R. Ghahremanzadeh, A. Bazgir, Tetrahedron 65, 2005 (2009)
M. Dabiri, S.C. Azimi, H.R. Khavasi, A. Bazgir, Tetrahedron 64, 7307 (2008)
M. Heravi, Z. Teymuri, N. Karimi, Y. Beheshtiha, N. Tavakoli, Gazi Univ. J. Sci. 28, 195 (2015)
M.A. Ghasemzadeh, M.H. Abdollahi-Basir, B. Mirhosseini-Eshkevari, Green Chem. Lett. Rev. 11, 47 (2018)
R. Ghahremanzadeh, F. Fereshtehnejad, A. Bazgir, J. Heterocycl. Chem. 47, 1031 (2010)
R. Ghahremanzadeh, S. Ahadi, G.I. Shakibaei, A. Bazgir, Tetrahedron Lett. 51, 499 (2010)
A. Feiz, G.I. Shakibaei, Z. Yasaei, H. Reza Khavasi, A. Bazgir, Helv. Chim. Acta 94, 1628 (2011)
A. Alizadeh, T. Firuzyar, A. Mikaeili, Synthesis 2010, 3913 (2010)
A.A. Mohammadi, S. Taheri, A. Amouzegar, J. Heterocycl. Chem. 54, 2085 (2017)
F. Rahimi, M. Bayat, H. Hosseini, RSC Adv. 9, 16384 (2019)
F.A. Tameh, J. Safaei-Ghomi, J. Iran. Chem. Soc. 15, 1633 (2018)
S. Khojasteh-Khosro, H. Shahbazi-Alavi, J. Chem. Res. 43, 107 (2019)
L. Rong, H. Han, H. Jiang, S. Tu, Synth. Commun. 39, 3493 (2009)
M.N. Elinson, A.I. Ilovaisky, V.M. Merkulova, F. Barba, B. Batanero, Tetrahedron 69, 7125 (2013)
T.H. Babu, A.A. Joseph, D. Muralidharan, P.T. Perumal, Tetrahedron Lett. 51, 994 (2010)
F.M. Abdelrazek, N.H. Metwally, N.A. Kassab, N.A. Sobhy, P. Metz, A. Jaeger, J. Heterocycl. Chem. 47, 384 (2010)
H. Karlsen, P.H. Songe, L.K. Sunsby, L.C. Hagen, P. Kolsaker, C. Rømming, J. Chem. Soc. Perkin 1, 497 (2001)
P. Das, T. Chaudhuri, C. Mukhopadhyay, ACS Comb. Sci. 16, 606 (2014)
M.-S. Tu, Y. Li, X. Wang, B. Jiang, S.-L. Wang, S.-J. Tu, RSC Adv. 3, 3877 (2013)
C. Mukhopadhyay, P. Das, R.J. Butcher, Organ. Lett. 13, 4664 (2011)
G.I. Shakibaei, A. Bazgir, RSC Adv. 6, 22306 (2016)
Z. Yasaei, P. Mirzaei, A. Bazgir, C. R. Chim. 13, 1308 (2010)
Z. Jamshidi, A.A. Esmaeili, J.T. Mague, J. Chem. Res. 40, 471 (2016)
A.A. Mohammadi, S. Askari, H. Rohi, A.A. Soorki, Synth. Commun. 44, 457 (2014)
M. Abdi, S. Rostamizadeh, N. Zekri, Polycycl. Aromat. Compd. 39(5), 413 (2017)
G.M. Ziarani, Z.K. Asl, P. Gholamzadeh, A. Badiei, M. Afshar, J. Sol-Gel Sci. Technol. 85, 103 (2018)
H. Kefayati, K. Rad-Moghadam, M. Zamani, S. Hosseyni, Lett. Organ. Chem. 7, 277 (2010)
B. Baghernejad, M. Khorshidi, Bull. Chem. Soc. Ethiop. 27, 309 (2013)
M. Khorshidi, M.M. Heravi, Y.S. Beheshtia, B. Baghernejad, Synth. Commun. 41, 2899 (2011)
F.A. Tameh, J. Safaei-Ghomi, M. Mahmoudi-Hashemi, H. Shahbazi-Alavi, RSC Adv. 6, 74802 (2016)
F. Alemi-Tameh, J. Safaei-Ghomi, M. Mahmoudi-Hashemi, M. Monajjemi, Polycycl. Aromat. Compd. 38, 199 (2018)
F. Alemi-Tameh, J. Safaei-Ghomi, M. Mahmoudi-Hashemi, R. Teymuri, Res. Chem. Intermed. 42, 6391 (2016)
S. Ahadi, Z. Yasaei, A. Bazgir, J. Heterocycl. Chem. 47, 1090 (2010)
G.M. Ziarani, M. Rahimifard, F. Nouri, A. Badiei, J. Serb. Chem. Soc. 80, 1265 (2015)
M. Rajabi-Salek, M.A. Zolfigol, M. Zarei, Res. Chem. Intermed. 44, 5255 (2018)
F. Rostami-Charati, Z. Hossaini, R. Rostamian, M. Ghambarian, A. Zamani, M. Abdoli, Chem. Heterocycl. Compd. 53, 480 (2017)
A.V. Moradi, J. Chem. Res. 41, 7 (2017)
A. Rahmati, A.H. Rezayan, M. Alizadeh, A. Nikbakht, J. Iran. Chem. Soc. 10, 521 (2013)
G.M. Ziarani, R. Moradi, N. Lashgari, A. Badiei, A.A. Soorki, Quím. Nova 38, 1167 (2015)
A. Bazgir, Z.N. Tisseh, P. Mirzaei, Tetrahedron Lett. 49, 5165 (2008)
S.C. Azimi, E. Abbaspour-Gilandeh, Q. Iran, Chem. Commun. 4, 245 (2016)
S. Asadi, G.M. Ziarani, M. Rahimifard, A.A. Soorki, Res. Chem. Intermed. 41, 6219 (2015)
R. Ghahremanzadeh, T. Amanpour, M. Sayyafi, A. Bazgir, J. Heterocycl. Chem. 47, 421 (2010)
M. Mirhosseini Moghaddam, A. Bazgir, A. Mohammad Mehdi, R. Ghahremanzadeh, Chin. J. Chem. 30, 709 (2012)
S. Jannati, A.A. Esmaeili, Tetrahedron 74, 2967 (2018)
A. Hasaninejad, S. Firoozi, F. Mandegani, Tetrahedron Lett. 54, 2791 (2013)
A. Yazdani-Elah-Abadi, R. Mohebat, M. Kangani, J. Chin. Chem. Soc. 64, 690 (2017)
A. Mohammadi, M. Bayat, S. Nasri, RSC Adv. 9, 16525 (2019)
S. Ghadiri, M. Bayat, F.S. Hosseini, Monatshefte Für Chem. Chem. Mon. 150, 1079 (2019)
R. Ghahremanzadeh, G. Hosseini, R. Akbarzadeh, A. Bazgir, J. Heterocycl. Chem. 50, 272 (2013)
Z.N. Tisseh, F. Ahmadi, M. Dabiri, H.R. Khavasi, A. Bazgir, Tetrahedron Lett. 53, 3603 (2012)
A.A. Esmaeili, S. Amini-Ghalandarabad, F. Mesbah, M. Tasmimi, M. Izadyar, A.R. Fakhari, A.R. Salimi, Tetrahedron 71, 2458 (2015)
N. Azizi, S. Dezfooli, M.M. Hashemi, J. Mol. Liq. 194, 62 (2014)
L. Youseftabar-Miri, Iran. Chem. Commun. 7, 142 (2019)
F. Mohamadpour, M.T. Maghsoodlou, R. Heydari, M. Lashkari, Res. Chem. Intermed. 42, 7841 (2016)
R. Ghahremanzadeh, T. Amanpour, A. Bazgir, J. Heterocycl. Chem. 46, 1266 (2009)
M.M. Heravi, M. Zakeri, A. Moharami, J. Chem. Sci. 124, 865 (2012)
S. Gholizadeh, K. Radmoghadam, Orient. J. Chem. 29, 1637 (2014)
B. Sadeghi, Z. Lasemi, R. Azimi, Orient. J. Chem. 31, 1175 (2015)
A. Hasaninejad, F. Mandegani, M. Beyrati, A. Maryamabadi, G. Mohebbi, ChemistrySelect 2, 6784 (2017)
Z. Karimi-Jaberi, A. Fereydoonnezhad, Q. Iran, Chem. Commun. 5, 407 (2017)
M. Dabiri, M. Bahramnejad, M. Baghbanzadeh, Tetrahedron 65, 9443 (2009)
H.A. Oskooie, M.M. Heravi, N. Karimi, H. Hamidi, Synth. Commun. 41, 3344 (2011)
Z. Lasemi, E. Mehrasbi, JACR, Special issue, 79 (2015)
M. Rohi, B. Sadeghi, M. Moslemin, Ciênc. E Nat. 37, 80 (2015)
Z.M. Darvish, B. Mirza, S. Makarem, J. Heterocycl. Chem. 54, 1763 (2017)
M. Zakeri, M.M. Nasef, E. Abouzari-Lotf, A. Moharami, M.M. Heravi, J. Ind. Eng. Chem. 29, 273 (2015)
M. Hosseini-Sarvari, M. Tavakolian, Comb. Chem. High Throughput Screen. 15, 826 (2012)
A. Bazgir, G. Hosseini, R. Ghahremanzadeh, ACS Comb Sci. 15, 530 (2013)
H. Hasani, M. Irizeh, Asian J. Green Chem. 3, 85 (2018)
F. Mohamadpour, M.T. Maghsoodlou, R. Heydari, M. Lashkari, J. Appl. Chem. Res. 11, 46 (2017)
L. Moradi, Z. Ataei, Green Chem. Lett. Rev. 10, 380 (2017)
M.A. Nasseri, F. Kamali, B. Zakerinasab, RSC Adv. 5, 26517 (2015)
A. Rezvanian, V. Zadsirjan, P. Saedi, M.M. Heravi, J. Heterocycl. Chem. 55, 2772 (2018)
M. Taheri, B. Mirza, M. Zeeb, J. Nanostruct. Chem. 8, 421 (2018)
H.R. Safaei, M. Shekouhy, A. Shirinfeshan, S. Rahmanpur, Mol. Divers. 16, 669 (2012)
S. Makarem, B. Mirza, Z. Mohammad Darvish, N. Amiri Notash, S. Ashrafi, Anal. Bioanal. Chem. Res. 6, 231 (2019)
L. Moradi, Z. Ataei, Z. Zahraei, J. Iran. Chem. Soc. 16, 1273 (2019)
B. Sadeghi, M.G. Pirbaluti, P.F. Nezhad, R.A. Nezhad, Res. Chem. Intermed. 41, 4047 (2015)
T. Jazinizadeh, M.T. Maghsoodlou, R. Heydari, A. Yazdani-Elah-Abadi, J. Iran. Chem. Soc. 14, 2117 (2017)
S.S. Khot, P.V. Anbhule, U.V. Desai, P.P. Wadgaonkar, C. R. Chim. 21, 814 (2018)
M. Divar, K. Zomorodian, R. Sabet, M. Moeini, S. Khabnadideh, Polycycl. Aromat. Compd. 39, 1 (2019)
M. Rouhi, B. Sadeghi, M. Moslemin, Bulg. Chem. Commun 50, 23 (2018)
M. Zarei, H. Sepehrmansourie, M.A. Zolfigol, R. Karamian, S.H.M. Farida, New J. Chem. 42, 14308 (2018)
S. Javanshir, N.S. Pourshiri, Z. Dolatkhah, M. Farhadnia, Monatshefte Für Chem. Chem. Mon. 148, 703 (2017)
A. Mobinikhaledi, N. Foroughifar, M.A.B. Fard, Synth. Commun. 41, 441 (2011)
A.R. Moosavi-Zare, M.A. Zolfigol, R. Salehi-Moratab, E. Noroozizadeh, Can. J. Chem. 95, 194 (2017)
H.R. Shaterian, F. Rigi, Res. Chem. Intermed. 40, 2983 (2014)
M. Samadizadeh, Asian J. Chem. 25(2), 926 (2013)
R. Azimi, R. Baharfar, Can. J. Chem. 92, 1163 (2014)
G. Mohammadi Ziarani, N. Hosseini Mohtasham, N. Lashgari, A. Badiei, M. Amanlou, R. Bazl, J. Nanostruct. 2, 489 (2012)
A. Allahresani, B. Taheri, M.A. Nasseri, Res. Chem. Intermed. 44, 1173 (2018)
K. Niknam, A. Piran, Z. Karimi, J. Iran. Chem. Soc. 13, 859 (2016)
M. Samadizadeh, Asian J. Chem. 25, 926 (2013)
G.M. Ziarani, A. Badiei, S. Mousavi, N. Lashgari, A. Shahbazi, Chin. J. Catal. 33, 1832 (2012)
G. Mohammadi Ziarani, R. Moradi, N. Lashgari, A. Badiei, A. Abolhassani Soorki, Polycycl. Aromat. Compd. 38, 66 (2018)
S. Sadjadi, M.M. Heravi, M. Malmir, B. Masoumi, Appl. Organomet. Chem. 32, e4113 (2018)
R. Baharfar, R. Azimi, Synth. Commun. 44, 89 (2014)
F. Kianfar, S.R.M. Moghadam, E. Kianfar, Indian J. Sci. Technol. 8, 1 (2015)
K. Niknam, P. Abolpour, Monatshefte Für Chem. Chem. Mon. 146, 683 (2015)
A.R. Moosavi-Zare, M.A. Zolfigol, E. Noroozizadeh, R. Salehi-Moratab, M. Zarei, J. Mol. Catal. Chem. 420, 246 (2016)
M.M. Heravi, E. Hashemi, F. Azimian, J. Iran. Chem. Soc. 12, 647 (2015)
M. Keshavarz, J. Iran. Chem. Soc. 13, 553 (2016)
A.R. Moosavi-Zare, M.A. Zolfigol, E. Noroozizadeh, M. Zarei, R. Karamian, M. Asadbegy, J. Mol. Catal. Chem. 425, 217 (2016)
G. Mohammadi Ziarani, H. Mollabagher, N. Lashgari, A. Badiei, Sci. Iran. 25, 3295 (2018)
M. Zarei, M.A. Zolfigol, A.R. Moosavi-Zare, E. Noroozizadeh, S. Rostamnia, ChemistrySelect 3, 12144 (2018)
A. Gharib, N.N. Pesyan, B.R.H. Khorasani, M. Roshani, J.H.W. Scheeren, Bulg. Chem. Commun. 45, 371 (2013)
M.A. Nasseri, S.M. Sadeghzadeh, J. Iran. Chem. Soc. 10, 1047 (2013)
N. Mohammadian, B. Akhlaghinia, Res. Chem. Intermed. 45(10), 4737 (2019)
M.A. Ghasemzadeh, B. Mirhosseini-Eshkevari, M.H. Abdollahi-Basir, BMC Chem. 13, 119 (2019)
M. Baghernejad, S. Khodabakhshi, S. Tajik, New J. Chem. 40, 2704 (2016)
L. Jalili-Baleh, N. Mohammadi, M. Khoobi, L. Ma’mani, A. Foroumadi, A. Shafiee, Helv. Chim. Acta 96, 1601 (2013)
N. Ahadi, M.A. Bodaghifard, A. Mobinikhaledi, Appl. Organomet. Chem. 33, e4738 (2019)
H. Hassani, B. Zakerinasab, A. Nozarie, Asian J. Green Chem. 2, 59 (2018)
M.A. Nasseri, K. Hemmat, A. Allahresani, Appl. Organomet. Chem. 33, e4743 (2019)
K. Hemmat, M.A. Nasseri, A. Allahresani, S. Ghiami, J. Organomet. Chem. 903, 120996 (2019)
B. Zamani-Ranjbar-Garmroodi, M.A. Nasseri, A. Allahresani, K. Hemmat, Res. Chem. Intermed. 45(11), 5665 (2019)
N.H. Nasab, J. Safari, J. Mol. Struct. 1193, 118 (2019)
M.S. Mirhosseyni, F. Nemati, A. Elhampour, Comb. Chem. High Throughput Screen. 21, 487 (2018)
E. Safari, A. Maryamabadi, A. Hasaninejad, RSC Adv. 7, 39502 (2017)
Acknowledgements
We would like to thank our colleagues in the Research Council of the University of Islamic Azad University, Tehran Medical Sciences, for providing a good contribution to all aspects of the project.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Youseftabar-Miri, L., Hosseinjani-Pirdehi, H., Akrami, A. et al. Recent investigations in the synthesis of spirooxindole derivatives by Iranian researchers. J IRAN CHEM SOC 17, 2179–2231 (2020). https://doi.org/10.1007/s13738-020-01921-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-020-01921-2