Abstract
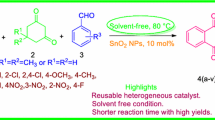
An efficient green approach for the synthesis of N-cyclohexyl-3-aryl-quinoxaline-2-amine derivatives, via a three-component one-pot condensation reaction of o-phenylenediamine, aromatic aldehydes and cyclohexyl isocyanide in the presence of perlite–SO3H nanoparticles (diameter/thickness of platelets < 100 nm) under ultrasound irradiation has been demonstrated. The present method offers advantages such as shorter reaction time, easy work-up, excellent yields, recovery and reusability of catalyst. In addition, the methodology has been prosperous in getting the green chemistry purposes such as natural catalyst, using ultrasound irradiation instead of conventional heating and stirring, and a non-hazardous products in the thus combining the features of both economic and environmental advantages.






Similar content being viewed by others
References
I. Akritopoulou-Zanze, Isocyanide-based multicomponent reactions in drug discovery. Curr. Opin. Chem. Biol. 12, 324–331 (2008)
S. Sadjadi, M. Heravi, N. Nazari, Isocyanide-based multicomponent reactions in the synthesis of heterocycles. RSC Adv 6, 53203–53272 (2016)
A. Domling, Recent developments in isocyanide based multicomponent reactions in applied chemistry. Chem. Rev. 106, 17–89 (2006)
M. Jida, M. Soueidan, N. Willand, F. Agbossou-Niedercorn, L. Pelinski, G. Laconde, R. Deprez-Poulain, B. Deprez, A facile and rapid synthesis of N-benzyl-2-substituted piperazines. Tetrahedron Lett. 52, 1705–1708 (2011)
M. Jida, S. Malaquin, R. Deprez-Poulain, G. Laconde, B. Deprez, Synthesis of five- and six-membered lactams via solvent-free microwave Ugi reaction. Tetrahedron Lett. 51, 5109–5111 (2010)
S. Malaquin, M. Jida, G.D. Gesquiere, R. Deprez-Poulain, B. Deprez, G. Laconde, Ugi reaction for the synthesis of 4-aminopiperidine-4-carboxylic acid derivatives. Application to the synthesis of carfentanil and remifentanil. Tetrahedron Lett. 51, 2983–2985 (2010)
A.E.A. Porter, in Comprehensive Heterocyclic Chemistry, 3 ed. by A.R. Katritzky, C.W. Rees eds. (Pergamon Press, New York, 1984), pp. 191–196
A. Varadi, T.C. Palmer, R. Notis Dardashti, S. Majumdar, Isocyanide-based multicomponent reactions for the synthesis of heterocycles. Molecules 21, 19–41 (2015)
G.W.H. Cheeseman, R.F. Cookson, in The Chemistry of Heterocyclic Compounds, 1 ed. by A. Weissberger, E.C. Taylor eds. 2nd edn. : (Wiley, New York, 1970)
S. Tariq, K. Somakala, M. Amir, Quinoxaline: an insight into the recent pharmacological advances. Eur. J. Med. Chem. 143, 542–557 (2018)
S. Ucar, S. Essiz, A. Dastan, Bromination of quinoxaline and derivatives: effective synthesis of some new brominated quinoxalines. Tetrahedron 73, 1618–1632 (2017)
R.N. Lima, A.L.M. Porto, Facile synthesis of new quinoxaline from ethyl gallate by green chemistry protocol. Tetrahedron Lett. 58, 825–828 (2017)
M. Sato, T. Nakazawa, Y. Tsunematsu, K. Hotta, K. Watanabe, Echinomycin biosynthesis. Curr. Opin. Chem. Biol. 17, 537–545 (2013)
H. Oveisi, M. Adharvana Chari, Ch.V. Nguyen, J.E. Chen, S.M. Alshehri, E. Ynmaz, Md.Sh. Hossein, Y Yamauchi, K.C.W. Wu, ZnO-loaded mesoporous silica (KIT-6) as an efficient solid catalyst for production of various substituted quinoxalines. Catal. Commun. 90, 111–115 (2017)
J.A. Pereira, A.M. Pessoa, M.N.D.S. Cordeiro, R. Fernandes, C. Prudencio, J.P. Noronha, M. Vieira, Quinoxaline, its derivatives and applications: a state of the review. Eur. J. Med. Chem. 97, 664–672 (2015)
J. Azuaje, A. El Maatouguri, X. Garcia-Mera, E. Sotelo, Ugi-based approaches to quinoxaline libraries. ACS Comb. Sci. 16, 403–411 (2014)
S. Achelle, Ch Baudequin, N. Ple, Luminescent materials incorporating pyrazine or quinoxaline moieties. Dyes Pigm. 98, 575–600 (2013)
H. Chavan, L.M. Adsul, B.P. Bandgar, Polyethylene glycol in water: a simple, efficient and green protocol for the synthesis of quinoxalines. J. Chem. Sci. 123, 477–483 (2011)
P.O. Patil, S.B. Bari, Nitrogen heterocycles as potential monoamine oxidase inhibitors: Synthetic aspects. Arab. J. Chem. 7, 857–884 (2014)
M.R. Islami, Z. Hassani, One-pot and efficient protocol for synthesis of quinoxaline derivatives. ARKIVOC 2008, 280–287 (2008)
H.R. Darabi, F. Tahoori, K. Aghapour, F. Taala, F. Mohsenzadeh, NH4Cl–CH3OH: an efficient, acid- and metal-free catalyst system for the synthesis of quinoxalines. J. Braz. Chem. Soc. 19, 1646–1652 (2008)
Ch Li, F. Zhang, Zh Yang, Ch Qi, Chemoselective synthesis of quinoxalines and benzimidazoles by silica gel catalysis. Tetrahedron Lett. 55, 5430–5433 (2014)
J.-F. Chen, Z.-Q. Liu, Synthesis of imidazo[1,2-a]quinoxalines by double Groebke reactions and inhibitory effects on radicals and DNA oxidation. Tetrahedron Lett. 72, 1850–1859 (2016)
I.H. Eissa, A.M. El-Naggar, N.E.A.A. El-Sattar, A.S.A. Youssef, Design and discovery of novel quinoxaline derivatives as dual DNA intercalators and topoisomerase II inhibitors. Anticancer Agents Med. Chem. 18, 195–209 (2018)
A. Dandia, V. Parewa, Sh Maheshwari, K.S. Rathore, Cu doped CdS nanoparticles: a versatile and recoverable catalyst for chemoselective synthesis of indolo[2,3-b]quinoxaline derivatives under microwave irradiation. J. Mol. Catal. A Chem. 394, 244–252 (2014)
M.M. Heravi, B. Baghernejad, H.A. Oskooie, A novel three-component reaction for the synthesis of N-cyclohexyl-3-aryl-quinoxaline-2-amines. Tetrahedron Lett. 50, 767–769 (2009)
J. Safaei-Ghomi, S. Rohani, A. Ziarati, CuI nanoparticles as a reusable heterogeneous catalyst for the one-pot synthesis of N-cyclohexyl-3-aryl-quinoxaline-2-amines under mild conditions. J. Nanostruc. 2, 79–83 (2012)
L.-Y. Fan, L. Wei, H. Wen-Jun, X.-X. Li, Yb modified NaY zeolite: a recyclable and efficient catalyst for quinoxaline synthesis. Chin. Chem. Lett. 25, 1203–1206 (2014)
H. Slimi, Y. Moussaoui, R.B. Salem, Synthesis of 3,4-dihydropyrimidin-2(1H)-ones/thiones via Biginelli reaction promoted by bismuth(III)nitrate or PPh3 without solvent. Aran. J. Chem. 9, 510–514 (2016)
M. Jeganthan, A. Dhakshinamoorthy, K. Pitchumani, One-pot synthesis of 2-substituted quinoxalines using K10-montmorillonite as heterogeneous catalyst. Tetrahedron Lett. 55, 1616–1620 (2014)
S.N. Hosseini, S.M. Borghei, M. Vossoughi, N. Taghavinia, Immobilization of TiO2 on perlite granules for photocatalytic degradation of phenol. Appl. Catalysis B Environ. 18, 53–62 (2007)
Jahanshahi,R.,Akhlaghinia,B. Expanded perlite: an inexpensive natural efficient heterogeneous catalyst for the green and highly accelerated solvent-free synthesis of 5-substituted-1H-tetrazoles using [bmim]N3 and nitriles. RSC Adv. 5: 104087–104096 (2015).
Ramazani,A.,Rouhani,M.,Mirhadi,E.,Sheikhi,M.,Ślepokura,K.,Lis,T, Perlite-SO3H nanoparticles as an efficient and reusable catalyst for one-pot three-component synthesis of 1,2-dihydro-1-aryl-naphtho[1,2-e][1,3]oxazine-3-one derivatives under both microwave-assisted and thermal solvent-free conditions: single crystal x-ray structure analysis and theoretical study, Nano. Chem. Res. 1:87–107 (2016).
Skubiszewska-Zięba,J.,Charmas,B.,Leboda,R.,Gun’ko,V.M., Carbon-mineral adsorbents with a diatomaceous earth/perlite matrix modified by carbon deposits, Micropor. Mesopor. Mat. 156:209–2016 (2012).
E. Kolvari, N. Koukabi, M.M. Hosseini, Perlite, A cheap natural support for immobilization of sulfonic acid as a heterogeneous solid acid catalyst for the heterocyclic multicomponent reaction. J. Mol. Catal. A Chem 397, 68–75 (2015)
G. Cravotto, P. Cintas, Forcing and controlling chemical reactions with ultrasound. Angew. Chem. Int. Ed. 46, 5476–5478 (2007)
A.C.M.P. Da Silva, C.G. Pancote, C.L. Brito, N.B.A.B. Da Silveira, Synthesis 2004, 1557–1558 (2004)
M. Rouhani, A. Ramazani, S.W. Joo, Y.Very Hanifehpour, Efficient and rapid catalyst-free one-pot three component synthesis of 2,5-dihydro-5-imino-2-methylfuran-3,4-dicarboxylate derivatives under ultrasound irradiation. Bull. Korean Chem. Soc. 33, 4127–4131 (2012)
A. Ramazani, M. Rouhani, S.W. Joo, Novel, fast and efficient one-pot sonochemical synthesis of 2-aryl-1,3,4-oxadiazoles. Ultrasoun. Sonochem. 20, 262–267 (2014)
A. Ramazani, M. Rouhani, S.W. Joo, Ultrasonics in isocyanide-based multicomponent reactions: a new, efficient and fast method for the synthesis of fully substituted 1,3,4-oxadiazole derivatives under ultrasound irradiation. Ultrasoun. Sonochem. 21, 391–396 (2015)
A. Ramazani, M. Rouhani, S.W. Joo, Catalyst-free sonosynthesis of highly substituted propanamide derivatives in water. Ultrason. Sonochem. 28, 393–399 (2016)
H. Ahankar, A. Ramazani, K. Slepokura, T. Lis, S.W. Joo, Synthesis of pyrrolidinone derivatives from aniline, an aldehyde and diethyl acetylenedicarboxylate in an ethanolic citric acid solution under ultrasound irradiation. Green Chem. 18, 3582–3593 (2016)
L. Wen, Zh..R. Li, M. Li, H. Cao, Solvent-free and efficient synthesis of imidazo[1,2-a]pyridine derivatives via a one-pot three-component reaction. Green Chem. 14, 707–716 (2012)
A. Palmieri, S. Gabrielli, C. Cimarelli, R. Ballini, Fast, mild, eco-friendly synthesis of polyfunctionalized pyrroles from β-nitroacrylates and β-enaminones. Green Chem. 13, 3333–3336 (2011)
A. Ramazani, A. Mahyari, A. Farshadi, M. Rouhani, Preparation of silica nanoparticles from organic laboratory waste of silica gel HF254 and their use as a highly efficient catalyst for the one-pot synthesis of 2,3-dihydro-1H-isoindolone derivatives. Helv. Chim. Acta. 94, 1831–1838 (2011)
M. Yurdakoc, M. Akcay, Y. Tonbul, K. Yurdakoc, Acidity of silica-alumina catalysts by amine titration using hammett indicators and FT-IR study of pyridine adsorption., Turk. J. Chem., 23: 319–327 (1999)
Acknowledgements
The authors thank Science and Research Branch, Islamic Azad University for the support and guidance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rouhani, M., Ramazani, A. Perlite–SO3H nanoparticles: very efficient and reusable catalyst for three-component synthesis of N-cyclohexyl-3-aryl-quinoxaline-2-amine derivatives under ultrasound irradiation. J IRAN CHEM SOC 15, 2375–2382 (2018). https://doi.org/10.1007/s13738-018-1426-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-018-1426-8