Abstract
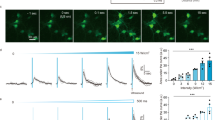
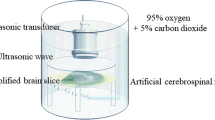
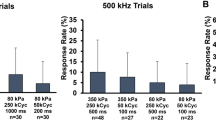
Ultrasound is becoming an emerging and promising method for neuromodulation due to its advantage of noninvasiveness and its high spatial resolution. However, the underlying principles of ultrasound neuromodulation have not yet been elucidated. We have herein developed a new in vitro setup to study the ultrasonic neuromodulation, and examined various parameters of ultrasound to verify the effective conditions to evoke the neural activity. Neurons were stimulated with 0.5 MHz center frequency ultrasound, and the action potentials were recorded from rat hippocampal neural cells cultured on microelectrode arrays. As the intensity of ultrasound increased, the neuronal activity also increased. There was a notable and significant increase in both the spike rate and the number of bursts at 50% duty cycle, 1 kHz pulse repetition frequency, and the acoustic intensities of 7.6 W/cm2 and 3.8 W/cm2 in terms of spatial-peak pulse-average intensity and spatial-peak temporal-average intensity, respectively. In addition, the impact of ultrasonic neuromodulation was assessed in the presence of a gamma-aminobutyric acid A (GABAA) receptor antagonist to exclude the effect of activated inhibitory neurons. Interestingly, it is noteworthy that the predominant neuromodulatory effects of ultrasound disappeared when the GABAA blocker was introduced, suggesting the potential of ultrasonic stimulation specifically targeting inhibitory neurons. The experimental setup proposed herein could serve as a useful tool for the clarification of the mechanisms underlying the electrophysiological effects of ultrasound.








Similar content being viewed by others
References
Levy R, Deer TR, Henderson J. Intracranial neurostimulation for pain control: a review. Pain Phys. 2010;13(2):157–65.
Yu X, Nagai J, Khakh BS. Improved tools to study astrocytes. Nat Rev Neurosci. 2020;21(3):121–38.
Gunaydin LA, et al. Ultrafast optogenetic control. Nat Neurosci. 2010;13(3):387–92.
Boyden ES, et al. Millisecond-timescale, genetically targeted optical control of neural activity. Nat Neurosci. 2005;8(9):1263–8.
Eom K, et al. Synergistic combination of near-infrared irradiation and targeted gold nanoheaters for enhanced photothermal neural stimulation. Biomed Opt Express. 2016;7(4):1614–25.
Tyler WJ. Noninvasive neuromodulation with ultrasound? A continuum mechanics hypothesis. Neuroscientist. 2010;17(1):25–36.
George MS, Lisanby SH, Sackeim HA. Transcranial magnetic stimulation: applications in neuropsychiatry. Arch Gen Psychiatry. 1999;56(4):300–11.
Nitsche MA, et al. Treatment of depression with transcranial direct current stimulation (tDCS): a review. Exp Neurol. 2009;219(1):14–9.
Bystritsky A, Korb AS. A review of low-intensity transcranial focused ultrasound for clinical applications. Curr Behav Neurosci Rep. 2015;2(2):60–6.
Rezayat E, Toostani IG. A review on brain stimulation using low intensity focused ultrasound. Basic Clin Neurosci. 2015;7(3):187–94.
Baek H, Pahk KJ, Kim H. A review of low-intensity focused ultrasound for neuromodulation. Biomed Eng Lett. 2016;7(2):135–42.
Khraiche ML, et al. Ultrasound induced increase in excitability of single neurons. Conf Proc IEEE Eng Med Biol Soc. 2008;2008:4246–9. https://doi.org/10.1109/IEMBS.2008.4650147.
Tyler WJ, et al. Remote excitation of neuronal circuits using low-intensity, low-frequency ultrasound. PLoS ONE. 2008;3(10):e3511.
Muratore, R. et al.: Bioeffects of low dose ultrasound on neuronal cell function. In 2009 38th Annual Symposium of the Ultrasonic Industry Association (UIA). 2009.
Tufail Y, et al. Ultrasonic neuromodulation by brain stimulation with transcranial ultrasound. Nat Protoc. 2011;6(9):1453–70.
Kim H, et al. Focused ultrasound-mediated non-invasive brain stimulation: examination of sonication parameters. Brain Stimul. 2014;7(5):748–56.
Lee W, et al. Creation of various skin sensations using pulsed focused ultrasound: Evidence for functional neuromodulation. Int J Imag Syst Technol. 2014;24(2):167–74.
Beaudoin GM 3rd, et al. Culturing pyramidal neurons from the early postnatal mouse hippocampus and cortex. Nat Protoc. 2012;7(9):1741–54.
Yao J, Qi J, Chen G. Actin-dependent activation of presynaptic silent synapses contributes to long-term synaptic plasticity in developing hippocampal neurons. J Neurosci. 2006;26(31):8137–47.
Li X, et al. Long-term recording on multi-electrode array reveals degraded inhibitory connection in neuronal network development. Biosens Bioelectron. 2007;22(7):1538–43.
Geissler M, Faissner A. A new indirect co-culture set up of mouse hippocampal neurons and cortical astrocytes on microelectrode arrays. J Neurosci Methods. 2012;204(2):262–72.
Xiang G, et al. Microelectrode array-based system for neuropharmacological applications with cortical neurons cultured in vitro. Biosens Bioelectron. 2007;22(11):2478–84.
Ahn S, et al. Study on the mechanisms of seizure-like events suppression effect by electrical stimulation using a microelectrode array. NeuroReport. 2017;28(9):471–8.
Gross GW, et al. The use of neuronal networks on multielectrode arrays as biosensors. Biosens Bioelectron. 1995;10(6–7):553–67.
Hochman DW, et al. Extracellular chloride and the maintenance of spontaneous epileptiform activity in rat hippocampal slices. J Neurophysiol. 1999;81(1):49–59.
King RL, et al. Effective parameters for ultrasound-induced in vivo neurostimulation. Ultrasound Med Biol. 2013;39(2):312–31.
Tyler WJ, Lani SW, Hwang GM. Ultrasonic modulation of neural circuit activity. Curr Opin Neurobiol. 2018;50:222–31.
ter Haar G. Therapeutic applications of ultrasound. Prog Biophys Mol Biol. 2007;93(1–3):111–29.
Kubanek J. Neuromodulation with transcranial focused ultrasound. Neurosurg Focus. 2018;44(2):E14–E14.
Kubanek J, et al. Ultrasound elicits behavioral responses through mechanical effects on neurons and ion channels in a simple nervous system. J Neurosci. 2018;588:4969.
Plaksin M, Kimmel E, Shoham S. Cell-type-selective effects of intramembrane cavitation as a unifying theoretical framework for ultrasonic neuromodulation. eneuro. 2016;3:ENEURO.0136-15.2016.
Wu YK, Fujishima K, Kengaku M. Differentiation of apical and basal dendrites in pyramidal cells and granule cells in dissociated hippocampal cultures. PLoS ONE. 2015;10(2):e0118482.
Alreja A, Nemenman I, Rozell CJ. Constrained brain volume in an efficient coding model explains the fraction of excitatory and inhibitory neurons in sensory cortices. PLoS Comput Biol. 2022;18(1):e1009642.
Xu J-C, et al. Cultured networks of excitatory projection neurons and inhibitory interneurons for studying human cortical neurotoxicity. Sci Transl Med. 2016;8(333):33348–33348.
Acknowledgements
We thank Prof. Tae-Seong Kim from Department of Biomedical Engineering, College of Electronics and Information, Kyung Hee University for offering ultrasound transducer and helpful comments during the writing of the manuscript. This research was supported by the National Research Foundation of Korea (NRF-2019M3C1B8090805, NRF-2022R1I1A4063209) and by the Korea Medical Device Development Fund grant funded by the Korea government (the Ministry of Science and ICT, the Ministry of Trade, Industry and Energy, the Ministry of Health & Welfare, the Ministry of Food and Drug Safety) (1711139110, KMDF_PR_20210527_0006).
Author information
Authors and Affiliations
Contributions
Conceptualization: all authors. Methodology: HS. Investigation: HS. Writing—original draft preparation: HS. Writing—review and editing: All authors. Approval of final manuscript: all authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to disclose.
Ethical approval
The procedures followed and the care of the animals were approved by the Institutional Animal Care and Use Committee in Ewha Womans University (Approval no. 17-010).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hwang, S., Jun, S.B. Ultrasound neuromodulation of cultured hippocampal neurons. Biomed. Eng. Lett. 14, 79–89 (2024). https://doi.org/10.1007/s13534-023-00314-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13534-023-00314-7