Abstract
Objective
The effect of L-ascorbic acid and α-tocopherol on parabenzoquinone(p-BQ)-induced hemato-biochemical and histological alterations in the liver and kidney of male Wistar rats was investigated.
Material and methods
Animals (n = 40) were divided into eight groups, all receiving p-BQ(1.5 mg Kg−1, i.p) for 21 days except the control group I which received equal amount of double-distilled water only. Group II served as positive control group, while all other groups received either L-ascorbic acid or α-tocopherol or combination of both L-ascorbic acid and α-tocopherol for 21 consecutive days.
Results
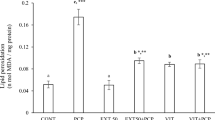
p-BQ significantly (P < 0.05) increased the neutrophil and platelet counts while reducing (P < 0.05) the total count of RBC, WBC, Hb, Hct, and lymphocyte. Animals administered p-BQ were found to have higher levels of hepatic (ALP, SGOT, and SGPT) and renal (creatinine, urea, and BUN) biomarker activity. Additionally, p-BQ treatment also significantly reduces HDL with concurrent elevation in the serum level of (P < 0.05), total cholesterol, triglycerides, and LDL. In the liver and kidney, p-BQ treatment caused significant reduction in total antioxidant capacity (SOD, CAT, GST& GSH) and also increases the level of MDA, marker of lipid peroxidation. The normal histoarchitecture of the liver and kidney tissues was disrupted indicating severe pathological changes after treatment with p-BQ. However, co-treatment with L-ascorbic acid and α-tocopherol, either alone or in combination, was effective in alleviating p-BQ-induced hemato-biochemical, antioxidant enzymes, and histopathological alterations in the treated animals.
Conclusion
To summarize, it appears that combined treatment with L-ascorbic acid and α-tocopherol was much more effective in ameliorating p-BQ-induced hematological and hepato-renal toxicity compared to only single vitamin treatment in Wistar rats.

Similar content being viewed by others
References
Ma Z, Yao D, Zhao J, Li H, Chen Z, Cui P, Zhu Z, Wang L, Wang Y, Ma Y, Gao J (2021) Efficient recovery of benzene and n-propanol from wastewater via vapor recompression assisted extractive distillation based on techno-economic and environmental analysis. Process Saf Environ Prot 1(148):462–472
Spatari G, Allegra A, Carrieri M, Pioggia G, Gangemi S (2021) Epigenetic effects of benzene in hematologic neoplasms: the altered gene expression. Cancers 13(10):2392
Jena AB, Samal RR, Kumari K, Pradhan J, Chainy GBN, Subudhi U, Pal S, Dandapat J (2021) The benzene metabolite p-benzoquinone inhibits the catalytic activity of bovine liver catalase: a biophysical study. Int J Biol Macromol 167:871–80. https://doi.org/10.1016/j.ijbiomac.2020.11.044
Hazardous Substances Data Bank (1993) Bethesda: National Toxicology Information Program, National Library of Medicine
Das A, Dey N, Ghosh A, Das T, Chatterjee IB (2011) NAD (P) H: quinoneoxidoreductase 1 deficiency conjoint with marginal vitamin C deficiency causes cigarette smoke induced myelodysplastic syndromes. PLoS ONE 6(5):e20590. https://doi.org/10.1371/journal.pone.0020590
Ghosh A, Banerjee S, Mitra A, Muralidharan M, Roy B, Banerjee R, Mandal AK, Chatterjee IB (2016) Interaction of p-benzoquinone with hemoglobin in smoker’s blood causes alteration of structure and loss of oxygen binding capacity. Toxicol Rep 3:295–305. https://doi.org/10.1016/j.toxrep.2016.02.001
Hegazy RM, Kamel HF (2014) Oxidant hepatic & /or Haem. Injury on fuel-station workers exposed to benzene vapor, possible protection of antioxidants. Am J Med Med Sci 4(2):34–46. https://doi.org/10.5923/j.ajmms.20140402.01
Snyder R (2007) Benzene’s toxicity: a consolidated short review of human and animal studies by HAKhan. Hum Exp Toxicol 26(9):687–96. https://doi.org/10.1177/0960327107083975
Magdy BW, Mohamed FE, Amin AS, Rana SS (2016) Ameliorative effect of antioxidants (vitamins C and E) against abamectin toxicity in liver, kidney and testis of male albino rats. J Basic Appl Zool 77:69–82. https://doi.org/10.1016/j.jobaz.2016.10.002
Ganguly S, Chandra A, Chattopadhyay DJ, Chatterjee IB (2017) p-benzoquinoneinitiates non-invasive urothelial cancer through aberrant tyrosine phosphorylation of EGFR, MAP kinase activation and cell cycle deregulation: prevention by vitamin C. Toxicol Rep 4:296–305. https://doi.org/10.1016/j.toxrep.2017.06.005
Abou EMF, Elbaiomy AE, Ahmed MS, Alsharif KF, Dahran N, Elmahallawy EK, Mokhbatly AA (2022) Ameliorative effects of bovine lactoferrin on benzene-induced hematotoxicity in albino rats. Front Vet Sci 9:5558
Liang B, Chen Y, Yuan W, Qin F, Zhang Q, Deng N, Liu X, Ma X, Zhang X, Zhang B, Deng Q, Huang M, Tang H, Liu L, Chen W, Xiao Y (2018) Down-regulation of miRNA-451a and miRNA-486–5p involved in benzene-induced inhibition on erythroidcell differentiation in vitro and in vivo. Arch Toxicol 92(1):259–72. https://doi.org/10.1007/s00204-017-2033-7
Arika WM, Nyamai DW, Musila MN, Ngugi MP, Njagi EN (2016) Hematological markers of in vivo toxicity. J Hematol Thromboembol Dis. https://doi.org/10.4172/2329-8790.1000236
Ovuru SS, Ekweozor IK (2004) Haematological changes associated with crude oil ingestion in experimental rabbits. Afr J Biotech 3(6):346–348
D’Azevedo PA, Tannhauser M, Tannhauser SL, Barros HM (1996) Hematological alterations in rats from xylene and benzene. Vet Hum Toxicol 38(5):340–4
D’Andrea MA, Reddy GK (2014) Hematological and hepatic alterations in nonsmoking residents exposed to benzene following a flaring incident at the British Petroleum plant in Texas City. Environ Health 13(1):115. https://doi.org/10.1186/1476-069X-13-115
Onunogbo CC, Ohaeri OC, Eleazu CO (2012) Effect of mistletoe (Viscum album) extract on the blood glucose, liver enzymes and electrolyte balance in alloxan induced diabetic rats. Am J Biochem Mol Biol 3(1):143–50. https://doi.org/10.3923/ajbmb.2013.143.150
Çavuşoğlu K, Yalçın E, Yapar K, Oruç E, Gür B, Çiçek F (2015) The effects of grape seed extract against toxicity of benzene on liver and kidney tissues of albino mice: biochemical evaluation. Turk J Biochem 40(1):66–73. https://doi.org/10.5505/tjb.2015.22448
Jarsiah P, Nosrati A, Alizadeh A, Hashemi-Soteh SM (2017) Hepatotoxicity and ALT/AST enzymes activities change in therapeutic and toxic doses consumption of acetaminophen in rats. Int Biol Biomed J 3(3):119–124
Adeyemi O, Ajayi JO, Olajuyin AM, Oloyede OB, Oladiji AT, Oluba OM, Adeyemi O, Ololade IA, Adebayo EA (2009) Toxicological evaluation of the effect of water contaminated with lead, phenol and benzene on liver, kidney and colon of Albino rats. Food Chem Toxicol 47(4):885–7. https://doi.org/10.1016/j.fct.2009.01.023
Morgan A, Galal MK, Ogaly HA, Ibrahim MA, Abd-Elsalam RM, Noshy P (2017) Tiron ameliorates oxidative stress and inflammation in titanium dioxide nanoparticles induced nephrotoxicity of male rats. Biomed Pharmacother. 93:779–87. https://doi.org/10.1016/j.biopha.2017.07.006
Halim AB, El-Ahmady O, Hassab-Allah S, Abdel-Galil F, Hafez Y, Darwish A (1997) Biochemical effect of antioxidants on lipids and liver function in experimentally induced liver damage. Ann Clin Biochem 34(6):656–63. https://doi.org/10.1177/000456329703400610
Alshareef A, Ibrahim M (2020) Neurological and biological toxicity of subchronic exposure to inhaled benzene in male rats. J Biochem Technol 11(2):52–9
Hemilä H (1992) Vitamin C and plasma cholesterol. Crit Rev Food Sci Nutr 32(1):33–57
Aluwong T, Ayo JO, Kpukple A, Oladipo OO (2016) Amelioration of hyperglycaemia, oxidative stress and dyslipidaemia in alloxan-induced diabetic wistar rats treated with probiotic and vitamin C. Nutrients 8(5):151
Mozos I, Stoian D, Luca CT (2017) Crosstalk between vitamins A, B12, D, K, C, and E status and arterial stiffness. Dis Markers 2017:8784971. https://doi.org/10.1155/2017/8784971
Emmanuel MO, Okwandu N, Dongo B (2015) Comparative effects of vitamin C and vitamin E pre-treatment in acute paracetamol induced toxicity on the liver of rats. World J Pharm Sci 2:407–12
Amin KA, Hameid HA II, AbdElsttar AH (2010) Effect of food azo dyes tartrazine and carmoisine on biochemical parameters related to renal, hepatic function and oxidative stress biomarkers in young male rats. Food Chem Toxicol 48(10):2994–2999. https://doi.org/10.1016/j.fct.2010.07.039
Emara AM, El-Bahrawy H (2008) Green tea attenuates benzene-induced oxidative stress in pump workers. J Immunotoxicol 5(1):69–80. https://doi.org/10.1080/15476910802019029
Kim SH, Kim JH, Park MA, Hwang SD, Kang JC (2015) The toxic effects of ammonia exposure on antioxidant and immune responses in Rockfish, Sebastesschlegelii during thermal stress. Environ Toxicol Pharmacol 40(3):954–9. https://doi.org/10.1016/j.etap.2015.10.006
Kumar A, Siddiqi NJ, Alrashood ST, Khan HA, Dubey A, Sharma B (2021) Protective effect of eugenol on hepatic inflammation and oxidative stress induced by cadmium in male rats. Biomed Pharmacother 1(139):111588. https://doi.org/10.1016/j.biopha.2021.111588
Sargazi S, Galavi H, Zarei S (2018) Evaluation of attenuative effect of tert-butylhydroquinone against diazinon-induced oxidative stress on hematological indices in male Wistar rats. Biomed Rep 8(6):565–70. https://doi.org/10.3892/br.2018.1090
Laouar A, Klibet F, Bourogaa E, Benamara A, Boumendjel A, Chefrour A, Messarah M (2017) Potential antioxidant properties and hepatoprotective effects of Juniperusphoenicea berries against CCl4 induced hepatic damage in rats. Asian Pac J Trop Med 10(3):263–269
Lowry O, Rosebrough N, Farr AL, Randall R (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193(1):265–75. https://doi.org/10.1016/S0021-9258(19)52451-6
Marklund S, Marklund G (1974) Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem 47(3):469–74
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione S-transferases: the first enzymatic step in mercapturic acid formation. J Biol Chem 249(22):7130–7139. https://doi.org/10.1016/S0021-9258(19)42083-8
Sinha AK (1972) Colorimetric assay of catalase. Anal Biochem 47(2):389–94. https://doi.org/10.1016/0003-2697(72)90132-7
Boyne AF, Ellman GL (1972) A methodology for analysis of tissue sulfhydryl components. Anal Biochem 46(2):639–53
Rehncrona S, Smith DS, Åkesson B, Westerberg E, Siesjö BK (1980) Peroxidative changes in brain cortical fatty acids and phospholipids, as characterized during Fe2+-and ascorbic acid-stimulated lipid peroxidation in vitro. J Neurochem 34(6):1630–1638
Suvarna KS, Layton C, Bancroft JD (eds) (2018) Bancroft’s theory and practice of histological techniques E-Book. Elsevier, London
Acknowledgements
The first author acknowledges Institutional Biotech Hub, Department of Biophysics, Pub Kamrup College, Baihata Chariali, Kamrup for support received to conduct a part of the enzymatic assay.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Ritu Mishra, Karabi Dutta and Manuj Kr. Bharali declare that we have no conflictof interest.
Ethical approval
The experiment protocols were approved by IAEC, Gauhati University, Guwahati. (IAEC/Per/2018-19/PP-IAEC/2018-19/48).
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mishra, R., Dutta, K. & Bharali, M.K. L-ascorbic acid and α-tocopherol treatment alleviates parabenzoquinone-induced hemato-biochemical and histopathological changes in Wistar rats. Toxicol. Environ. Health Sci. 14, 379–387 (2022). https://doi.org/10.1007/s13530-022-00152-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13530-022-00152-7