Abstract
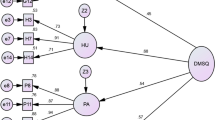
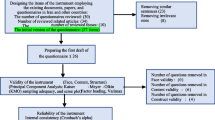
The purpose of this study is to test the validity and reliability of the Barriers to Insulin Treatment Questionnaire and adapt the questionnaire for Turkish society. This was a cross-sectional study. The population of this study consisted of adult patients with type 2 diabetes who were using insulin and being monitored in the Endocrine and Outpatient Clinics of Inonu University Turgut Ozal Medical Centre. The sample of the study consisted of 348 patients. The data were collected by using the Barriers to Insulin Treatment Questionnaire (BITQ) and a survey including demographic characteristics. The 14 items of the questionnaire are scored between 1 and 10. BITQ has five factors as fear of injections and self-testing, expectations regarding positive insulin-related outcomes, expected hardship from insulin treatment, stigmatization by insulin injections, and fear of hypoglycemia. In order to test the construct validity of the questionnaire, Exploratory Factor Analysis (EFA) and Confirmatory Factor Analysis (CFA) were utilized. The reliability of BITQ was evaluated by its Cronbach’s alpha internal consistency coefficient, item total correlation, and test-retest reliability. It was determined that the Cronbach’s alpha reliability coefficients of the questionnaire factors ranged between 0.70 and 0.82, the scale item factor loadings varied from 0.54 to 0.86, and the item total correlation coefficients were between 0.30 and 0.52. CFA supported the 5-factor scale structure obtained as a result of EFA. Good fit index values were obtained as a result of CFA. According to the obtained results, it may be asserted that BITQ is valid and reliable in terms of its application in Turkish society.

Similar content being viewed by others
References
Koski RR. Practical review of oral antihyperglycemic agents for type 2 diabetes mellitus. Diabetes Educ. 2006;32:869–76.
Lebovitz HE. Treating hyperglycemia in type 2 diabetes: new goals and strategies. Cleve Clin J Med. 2002;69:809–20.
Nathan DM. Clinical practice. Initial management of glycemia in type 2 diabetes mellitus. N Engl J Med. 2002;347:1342–9.
Wright A, Burden AC, Paisey RB, Cull CA, Holman RR, U.K. Prospective Diabetes Study Group. Sulfonylurea inadequacy: efficacy of addition of insulin over 6 years in patients with type 2 diabetes in the U.K. prospective diabetes study (UKPDS 57). Diabetes Care. 2002;25:330–6.
Nathan DM, Genuth S, Lachin J, Cleary P, Crofford O, Davis M, et al. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. The diabetes control and complications trial research group. N Engl J Med. 1993;329:977–86.
UK prospective diabetes study (UKPDS) Group. Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). Lancet. 1999;14:602.
Stratton IM, Adler AI, Neil HA, Matthews DR, Manley SE, Cull CA, et al. Association of glycaemia with macrovascular and microvascular complications of type 2 diabetes (UKPDS 35): prospective observational study. BMJ. 2000;321:405–12.
Snoek FJ. Breaking the barriers to optimal glycaemic control-what physicians need to know from patients’ perspectives. Int J Clin Pract Suppl. 2002;129:80–4.
Snoek FJ, Mollema ED, Heine RJ, Bouter LM, van der Ploeg HM. Development and validation of the diabetes fear of injecting and self-testing questionnaire (DFISQ): first findings. Diabet Med. 1997;14:871–6.
Khan H, Lasker SS, Chowdhury TA. Prevalence and reasons for insulin refusal in Bangladeshi patients with poorly controlled type 2 diabetes in East London. Diabet Med. 2008;25:1108–11.
Petrak F, Stridde E, Leverkus F, Crispin AA, Forst T, Pfützner A. Development and validation of a new measure to evaluate psychological resistance to insulin treatment. Diabetes Care. 2007;30(9):2199–204.
Seçer İ. Psychological test development and adaptation process: SPSS and LISREL applications [in Turkish]. Ankara, Anı, 2015, pp. 32–59.
Beaton DE, Bombardier C, Guillemin F, Ferraz MB. Guidelines for the process of cross-cultural adaptation of self-report measures. Spine. 2000;25:3186–91.
Karasar N. Scientific research methods [in Turkish]. Ankara: Nobel; 2012. p. 5–33.
Pallant J. SPSS survival manual: a step by step guide to data analysis using SPSS for windows (version 12). 2nd ed. Maidenhead: Open University Press; 2005. p. 3–179.
Kline RB. Principles and practice of structural equation modeling. Fourth edt. New York: Guilford Press; 2015.
Tezbaşaran AA. Likert-type scale preparation manual [in Turkish] e-book. 2008, pp. Available from http://www.academia.edu/1288035/.2008, pp. 9–51.
Alpar R. Applied multivariate statistical methods [in Turkish]. Ankara: Detay; 2011. p. 10–29.
Burns N, Grove SK. The practice of nursing research appraisal, synthesis, and generation of evidence. 6th ed. St. Louis: Saunders; 2009. p. 76–593.
Şencan H. Sosyal ve davranışsal ölçümlerde güvenilirlik ve geçerlilik. Ankara: Seçkin Yayincilik; 2005. p. 150–7.
Akgül A. A statistical analysis: methods in medical research—SPSS applications [in Turkish]. Ankara: Emek; 2005. p. 6–22.
Büyüköztürk S. A data analysis handbook for the social sciences [in Turkish]. Ankara: Pegem; 2012. p. 1–15.
Harrington D. Confirmatory factor analysis. New York: Oxford University Press; 2009. p. 5–77.
Streiner DL, Kottner J. Recommendations for reporting the results of studies of instrument and scale development and testing. J Adv Nurs. 2014;70(9):1970–9.
Polit DF, Beck CT. Essentials of nursing research: appraising evidence for nursing practice. 7th ed. Philadelphia: Lippincott, Williams & Wilkins; 2010. p. 391–591.
Bland JM, Altman GD. Statistics notes: Cronbach’s alpha. BMJ. 1997;314:572.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the article.
Informed consent
Informed consent was obtained from all participants included in the study.
Ethical approval
The population and sample of the study. The study was approved by the Scientific Research and Ethics Board of Inonu University Health Sciences Institute. Verbal information was provided to all participants in accordance with the principles of the Declaration of Helsinki. Additionally, the participants were informed about the research and notified that their personal data would be kept confidential.
Rights and permissions
About this article
Cite this article
Saritas, S.C., Erci, B., Sahin, I. et al. Validity study of the Turkish version of the barriers to insulin treatment questionnaire. Int J Diabetes Dev Ctries 39, 247–253 (2019). https://doi.org/10.1007/s13410-018-0670-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13410-018-0670-x