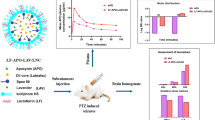
Abstract
Epilepsy is a condition in the brain cause frequent seizures and related comorbidities. The major hindrance during the design of antiepileptic drugs is their permeability through the blood–brain barrier (BBB). Recently, advancements in biology and engineering have led to the development of nanocomposites for drug delivery through the BBB. In this study, we have fabricated a novel type of nanoparticles using a plant compound, naringenin, with graphene oxide (NGO). The mean size of the fabricated nanoparticles was obtained as 73.1 nm and the polydispersity value was obtained as 3.286 through dynamic light scattering measurement. The average zeta potential value, − 49.6 mV, showed that the nanoparticles were strongly anionic in nature. A fluorescent imaging experiment demonstrated that the fabricated fluorescein isothiocyanice (FITC) tagged NGO nanoparticles localised successfully in adult zebrafish brains. The in vitro antioxidant assay showed that NGO nanoparticles have reduced the levels of 2,2-diphenyl-1-picrylhydrazyl (DPPH) and 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) free radicals upto 41.5 ± 0.51 and 64.25 ± 1.62%, respectively. In vivo developmental toxicity assessment indicated that NGO nanoparticles were non-toxic to the developing embryo of zebrafish. The percentage of ROS and apoptosis in zebrafish larvae due to pentylenetetrazole (PTZ) exposure was significantly reduced by NGO pre-treatment. Additionally, the behavioural analysis showed that NGO could suppress the PTZ-induced convulsant behaviour in adult zebrafish. From the results of both in vitro and in vivo experiments, we concluded that NGO nanoparticles could be used as promising medicine to treat free radical-induced neuronal damage and epilepsy, provided the results need to be reconfirmed in the mammalian model as well as a clinical trial.







Similar content being viewed by others
Data availability
Data will be made available on reasonable request.
References
Moshé SL, Perucca E, Ryvlin P, Tomson T (2015) Epilepsy: new advances. Lancet 385:884–898. https://doi.org/10.1016/S0140-6736(14)60456-6
Trinka E, Kwan P, Lee B, Dash A (2019) Epilepsy in Asia: disease burden, management barriers, and challenges. Epilepsia 60:7–21. https://doi.org/10.1111/epi.14458
Kumar M, Kumar P (2017) Protective effect of spermine against pentylenetetrazole kindling epilepsy induced comorbidities in mice. Neurosci Res 120:8–17. https://doi.org/10.1016/j.neures.2017.02.003
Aguiar CCT, Almeida AB, Araújo PVP et al (2012) Oxidative stress and epilepsy: literature review. Oxid Med Cell Longev 2012:1–12. https://doi.org/10.1155/2012/795259
Malinska D, Kulawiak B, Kudin AP et al (2010) Complex III-dependent superoxide production of brain mitochondria contributes to seizure-related ROS formation. Biochim Biophys Acta - Bioenerg 1797:1163–1170. https://doi.org/10.1016/j.bbabio.2010.03.001
Zhu X, Dong J, Shen K et al (2015) NMDA receptor NR2B subunits contribute to PTZ-kindling-induced hippocampal astrocytosis and oxidative stress. Brain Res Bull 114:70–78. https://doi.org/10.1016/j.brainresbull.2015.04.002
Kovács R, Rabanus A, Otáhal J et al (2009) Endogenous nitric oxide is a key promoting factor for initiation of seizure-like events in hippocampal and entorhinal cortex slices. J Neurosci 29:8565–8577. https://doi.org/10.1523/JNEUROSCI.5698-08.2009
Zhu X, Dong J, Han B et al (2017) Neuronal nitric oxide synthase contributes to PTZ kindling epilepsy-induced hippocampal endoplasmic reticulum stress and oxidative damage. Front Cell Neurosci 11:1–16. https://doi.org/10.3389/fncel.2017.00377
Vauzour D, Vafeiadou K, Rodriguez-Mateos A et al (2008) The neuroprotective potential of flavonoids: a multiplicity of effects. Genes Nutr 3:115–126. https://doi.org/10.1007/s12263-008-0091-4
Hassan HM, Elnagar MR, Abdelrazik E et al (2022) Neuroprotective effect of naringin against cerebellar changes in Alzheimer’s disease through modulation of autophagy, oxidative stress and tau expression: an experimental study. Front Neuroanat 16:1012422. https://doi.org/10.3389/fnana.2022.1012422
Pardridge WM (2012) Drug transport across the blood–brain barrier. J Cereb Blood Flow Metab 32:1959–1972. https://doi.org/10.1038/jcbfm.2012.126
Ceña V, Játiva P (2018) Nanoparticle crossing of blood–brain barrier: a road to new therapeutic approaches to central nervous system diseases. Nanomedicine 13:1513–1516. https://doi.org/10.2217/nnm-2018-0139
Tabish TA, Narayan RJ (2021) Crossing the blood–brain barrier with graphene nanostructures. Mater Today 51:393–401. https://doi.org/10.1016/j.mattod.2021.08.013
Tapeinos C (2021) Graphene-based nanotechnology in neurodegenerative disorders. Adv NanoBiomed Res 1:2000059. https://doi.org/10.1002/anbr.202000059
Vinodhkumar G, Ramya R, Vimalan M et al (2018) Reduced graphene oxide based on simultaneous detection of neurotransmitters. Prog Chem Biochem Res 1:40–49. https://doi.org/10.29088/SAMI/PCBR.2018.1.4049
Bellet P, Gasparotto M, Pressi S et al (2021) Graphene-based scaffolds for regenerative medicine. Nanomaterials 11:404. https://doi.org/10.3390/nano11020404
Rahmanian N, Hamishehkar H, Dolatabadi JEN, Arsalani N (2014) Nano graphene oxide: a novel carrier for oral delivery of flavonoids. Colloids Surf B Biointerfaces 123:331–338. https://doi.org/10.1016/j.colsurfb.2014.09.036
Priya PS, Vaishnavi S, Pavithra V et al (2023) Graphene oxide decorated daidzein as an oral drug to ameliorate the oxidative stress and glucocorticoid-induced osteoporosis in vivo zebrafish model. J Drug Deliv Sci Technol 81:104278. https://doi.org/10.1016/j.jddst.2023.104278
Rueden CT, Schindelin J, Hiner MC et al (2017) Image J2: ImageJ for the next generation of scientific image data. BMC Bioinformatics 18:529. https://doi.org/10.1186/s12859-017-1934-z
Prabha N, Guru A, Harikrishnan R et al (2022) Neuroprotective and antioxidant capability of RW20 peptide from histone acetyltransferases caused by oxidative stress-induced neurotoxicity in in vivo zebrafish larval model. J King Saud Univ - Sci 34:101861. https://doi.org/10.1016/j.jksus.2022.101861
Guru A, Velayutham M, Arockiaraj J (2022) Lipid-lowering and antioxidant activity of RF13 peptide from vacuolar protein sorting-associated protein 26B (VPS26B) by modulating lipid metabolism and oxidative stress in HFD induced obesity in zebrafish larvae. Int J Pept Res Ther 28:74. https://doi.org/10.1007/s10989-022-10376-3
Pekarova M, Kralova J, Kubala L et al (2009) Carvedilol and adrenergic agonists suppress the lipopolysaccharide-induced no production in raw 264.7 macrophages via the adrenergic receptors. J Physiol Pharmacol 60:143–150
Haridevamuthu B, Manjunathan T, Guru A et al (2022) Hydroxyl containing benzo[b]thiophene analogs mitigates the acrylamide induced oxidative stress in the zebrafish larvae by stabilising the glutathione redox cycle. Life Sci 298:120507. https://doi.org/10.1016/j.lfs.2022.120507
Sudhakaran G, Prathap P, Guru A et al (2022) Reverse pharmacology of Nimbin-N2 attenuates alcoholic liver injury and promotes the hepatoprotective dual role of improving lipid metabolism and downregulating the levels of inflammatory cytokines in zebrafish larval model. Mol Cell Biochem 477:2387–2401. https://doi.org/10.1007/s11010-022-04448-7
Sarkar P, Guru A, Raju SV et al (2021) GP13, an Arthrospira platensis cysteine desulfurase-derived peptide, suppresses oxidative stress and reduces apoptosis in human leucocytes and zebrafish (Danio rerio) embryo via attenuated caspase-3 expression. J King Saud Univ Sci 33:101665. https://doi.org/10.1016/j.jksus.2021.101665
Sari MM (2013) Fluorescein isothiocyanate conjugated graphene oxide for detection of dopamine. Mater Chem Phys 138:843–849. https://doi.org/10.1016/j.matchemphys.2012.12.069
Yamanaka O, Takeuchi R (2018) UMATracker: an intuitive image-based tracking platform. J Exp Biol 221:1–5. https://doi.org/10.1242/jeb.182469
Ferrari M, Downing G (2005) Medical Nanotechnology. BioDrugs 19:203–210. https://doi.org/10.2165/00063030-200519040-00001
Ahmad A, Fauzia E, Kumar M et al (2019) Gelatin-coated polycaprolactone nanoparticle-mediated naringenin delivery rescue human mesenchymal stem cells from oxygen glucose deprivation-induced inflammatory stress. ACS Biomater Sci Eng 5:683–695. https://doi.org/10.1021/acsbiomaterials.8b01081
Krishnakumar N, Sulfikkarali N, RajendraPrasad N, Karthikeyan S (2011) Enhanced anticancer activity of naringenin-loaded nanoparticles in human cervical (HeLa) cancer cells. Biomed Prev Nutr 1:223–231. https://doi.org/10.1016/j.bionut.2011.09.003
Jacox ME (2003) Vibrational and electronic energy levels of polyatomic transient molecules. Supplement B. J Phys Chem Ref Data 32:1–441. https://doi.org/10.1063/1.1497629
Sengupta I, Kumar SSS, Gupta K, Chakraborty S (2021) In-vitro release study through novel graphene oxide aided alginate based pH-sensitive drug carrier for gastrointestinal tract. Mater Today Commun 26:101737. https://doi.org/10.1016/j.mtcomm.2020.101737
Francenia Santos-Sánchez N, Salas-Coronado R, Villanueva-Cañongo C, Hernández-Carlos B (2019) Antioxidant compounds and their antioxidant mechanism. Antioxidants 1–28 https://doi.org/10.5772/intechopen.85270
Hersh AM, Alomari S, Tyler BM (2022) Crossing the blood-brain barrier: advances in nanoparticle technology for drug delivery in neuro-oncology. Int J Mol Sci 23:4153. https://doi.org/10.3390/ijms23084153
Zhang X, Zhou Q, Zou W, Hu X (2017) Molecular mechanisms of developmental toxicity induced by graphene oxide at predicted environmental concentrations. Environ Sci Technol 51:7861–7871. https://doi.org/10.1021/acs.est.7b01922
Cebadero-Domínguez O, Ferrández-Gómez B, Sánchez-Ballester S et al (2022) In vitro toxicity evaluation of graphene oxide and reduced graphene oxide on Caco-2 cells. Toxicol Rep 9:1130–1138. https://doi.org/10.1016/j.toxrep.2022.05.010
Zhang J, Cao H-Y, Wang J-Q, et al (2021) Graphene oxide and reduced graphene oxide exhibit cardiotoxicity through the regulation of lipid peroxidation, oxidative stress, and mitochondrial dysfunction. Front Cell Dev Biol 9 https://doi.org/10.3389/fcell.2021.616888
Lim YJ, Kim JH, Pan JH et al (2018) Naringin protects pancreatic β-cells against oxidative stress-induced apoptosis by inhibiting both intrinsic and extrinsic pathways in insulin-deficient diabetic mice. Mol Nutr Food Res 62:1–47. https://doi.org/10.1002/mnfr.201700810
Lockman PR, Koziara JM, Mumper RJ, Allen D (2004) Nanoparticle surface charges alter blood-brain barrier integrity and permeability. J Drug Target 12:635–641. https://doi.org/10.1080/10611860400015936
Myren-Svelstad S, Jamali A, Ophus SS et al (2022) Elevated photic response is followed by a rapid decay and depressed state in ictogenic networks. Epilepsia 63:2543–2560. https://doi.org/10.1111/epi.17380
Martinc B, Grabnar I, Vovk T (2015) Antioxidants as a preventive treatment for epileptic process: a review of the current status. Curr Neuropharmacol 12:527–550. https://doi.org/10.2174/1570159X12666140923205715
Fontana BD, Ziani PR, Canzian J et al (2019) Taurine protects from pentylenetetrazole-induced behavioral and neurochemical changes in zebrafish. Mol Neurobiol 56:583–594. https://doi.org/10.1007/s12035-018-1107-8
Acknowledgements
The authors express their sincere appreciation to the Researchers Supporting Project Number (RSP2023R414), King Saud University, Riyadh, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
Raghul Murugan: Conceptualization, Methodology, Formal analysis, Investigation, Visualisation, Writing—Original Draft, Writing—Review & Editing. G. Mukesh: Methodology, Formal analysis, Investigation, Visualisation, Writing—Review & Editing. B. Haridevamuthu: Methodology, Formal analysis, Investigation, Writing—Review & Editing. P. Snega Priya: Methodology, Formal analysis, Investigation. Raman Pachaiappan, Bader O. Almutairi, Selvaraj Arokiyaraj: Methodology, Formal analysis, Investigation, Resources. Ajay Guru, Jesu Arockiaraj: Conceptualisation, Methodology, Funding acquisition, Formal analysis, Supervision, Writing – review & editing, Project administration.
Corresponding authors
Ethics declarations
Ethical approval
The experiments were performed following the guidelines of the institutional animal ethical approval (No. SAF/IAEC/211215/004).
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Murugan, R., Mukesh, G., Haridevamuthu, B. et al. Plausible antioxidant and anticonvulsant potential of brain targeted naringenin-conjugated graphene oxide nanoparticles. Biomass Conv. Bioref. (2023). https://doi.org/10.1007/s13399-023-04343-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13399-023-04343-1