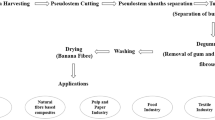
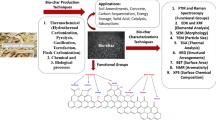
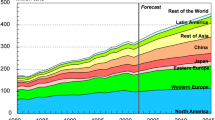
Abstract
Lignin is an aromatic complex polymer that constitutes a major part of biomass, along with cellulose and hemicellulose. Due to its abundant and renewable nature, it can be an ideal candidate for reducing the dependence on fossil resources. However, extraction and further valorization of lignin to useful chemicals has been an uphill task due to its highly branched and recalcitrant nature. Lignin-based biorefineries can be a major boost to a highly populated country like India where there is an ever-rising demand for chemicals. India being an agricultural country produces an exorbitant amount of lignin in the form of agro-residues, which can be properly valorized to achieve the aim of lignin-based biorefineries. In this review, various processes of lignin extraction from biomass residues are discussed in brief, and a major part of the review focuses on recent techniques developed to valorize this aromatic polymer into useful chemicals. These techniques have been compared based on the type of catalyst used, the extent of depolymerization, and the monomeric yield and selectivity of the process. Finally, the potential of lignin as a sustainable source of useful chemicals in India is discussed.






Similar content being viewed by others
References
Stefanidis SD, Kalogiannis KG, Iliopoulou EF et al (2014) A study of lignocellulosic biomass pyrolysis via the pyrolysis of cellulose, hemicellulose and lignin. J Anal Appl Pyrolysis 105:143–150. https://doi.org/10.1016/j.jaap.2013.10.013
Ralph J, Lapierre C, Boerjan W (2019) Lignin structure and its engineering. Curr Opin Biotechnol 56:240–249. https://doi.org/10.1016/j.copbio.2019.02.019
Wang H, Pu Y, Ragauskas A, Yang B (2019) From lignin to valuable products–strategies, challenges, and prospects. Bioresour Technol 271:449–461
Tang PL, Hassan O, Yue CS, Abdul PM (2020) Lignin extraction from oil palm empty fruit bunch fiber (OPEFBF) via different alkaline treatments. Biomass Convers Biorefinery 10:125–138. https://doi.org/10.1007/s13399-019-00413-5
Başakçılardan Kabakcı S, Tanış MH (2020) Pretreatment of lignocellulosic biomass at atmospheric conditions by using different organosolv liquors: a comparison of lignins. Biomass Convers Biorefinery. https://doi.org/10.1007/s13399-020-00677-2
Watkins D, Nuruddin M, Hosur M et al (2015) Extraction and characterization of lignin from different biomass resources. J Mater Res Technol 4:26–32. https://doi.org/10.1016/j.jmrt.2014.10.009
Fernández-Rodríguez J, Erdocia X, Hernández-Ramos F, et al (2019) Chapter 7 - Lignin separation and fractionation by ultrafiltration. In: Galanakis CMBT-S of FM in F by MT (ed). Academic Press, pp 229–265, ISBN 9780128150566, https://doi.org/10.1016/B978-0-12-815056-6.00007-3.
Tian D, Chandra RP, Lee JS et al (2017) A comparison of various lignin-extraction methods to enhance the accessibility and ease of enzymatic hydrolysis of the cellulosic component of steam-pretreated poplar. Biotechnol Biofuels 10:1–10. https://doi.org/10.1186/s13068-017-0846-5
Cequier E, Aguilera J, Balcells M, Canela-Garayoa R (2019) Extraction and characterization of lignin from olive pomace: a comparison study among ionic liquid, sulfuric acid, and alkaline treatments. Biomass Convers Biorefinery 9:241–252. https://doi.org/10.1007/s13399-019-00400-w
Cao X, Chen F, Cen K, et al (2020) Effect of torrefaction on the pyrolysis behavior, kinetics, and phenolic products of lignin. Biomass Convers Biorefinery.https://doi.org/10.1007/s13399-020-01136-8
Wu Z, Wang F, Xu J, et al (2020) Improved lignin pyrolysis over attapulgite-supported solid acid catalysts.Biomass Convers Biorefinery. https://doi.org/10.1007/s13399-020-00667-4
Chen C, Liu P, Sharma BK, et al (2020) Insights into catalytic valorization of different lignin feedstocks into liquid fuels with microwave heating in hydrogen-donor solvents. Biomass Convers Biorefinery 19–22. https://doi.org/10.1007/s13399-020-00849-0
Liu X, Bouxin FP, Fan J et al (2020) Recent advances in the catalytic depolymerization of lignin towards phenolic chemicals: a review. Chemsuschem 13:4296–4317. https://doi.org/10.1002/cssc.202001213
Cabral Almada C, Kazachenko A, Fongarland P, et al (2020) Oxidative depolymerization of lignins for producing aromatics: variation of botanical origin and extraction methods. Biomass Convers Biorefinery.https://doi.org/10.1007/s13399-020-00897-6
Hafezisefat P, Lindstrom JK, Brown RC, Qi L (2020) Non-catalytic oxidative depolymerization of lignin in perfluorodecalin to produce phenolic monomers. Green Chem 22:6567–6578. https://doi.org/10.1039/d0gc02505d
Zeng J, Tong Z, Bao H, et al (2020) Controllable depolymerization of lignin using carbocatalyst graphene oxide under mild conditions. Fuel 267:117100. https://doi.org/10.1016/j.fuel.2020.117100
Carrozza CF, Leonardi G, Vasso M, et al (2020) Novel in-situ preparation of nano sized Ni (0) catalyst for depolymerization of lignin-rich waste from industrial biorefinery. Bioresour Technol Reports 10:100355. https://doi.org/10.1016/j.biteb.2019.100355
Negi H, Singh RK (2020) A review on lignin utilization in petroleum exploration, petroleum products formulation, bio-fuel production, and oil spill clean-up. Biomass Convers Biorefinery.https://doi.org/10.1007/s13399-020-01126-w
Hao K, Zhang LL, Song L et al (2020) Highly active Mo-V-based bifunctional catalysts for catalytic conversion of lignin dimer model compounds at room temperature. Inorg Chem Commun 116:107910. https://doi.org/10.1016/j.inoche.2020.107910
Qi S, Wang G, Sun H et al (2020) Using lignin monomer as a novel capping agent for efficient acid-catalyzed depolymerization of high molecular weight lignin to improve its antioxidant activity. ACS Sustain Chem Eng 8:9104–9114. https://doi.org/10.1021/acssuschemeng.0c02366
Hernández-Ramos F, Fernández-Rodríguez J, Alriols MG, et al (2020) Study of a renewable capping agent addition in lignin base catalyzed depolymerization process. Fuel 280:118524. https://doi.org/10.1016/j.fuel.2020.118524
Cornejo A, García-Yoldi Í, Alegria-Dallo I et al (2020) Systematic diffusion-ordered spectroscopy for the selective determination of molecular weight in real lignins and fractions arising from base-catalyzed depolymerization reaction mixtures. ACS Sustain Chem Eng 8:8638–8647. https://doi.org/10.1021/acssuschemeng.0c01375
Pineda A, Lee AF (2016) Heterogeneously catalyzed lignin depolymerization. Appl Petrochemical Res 6:243–256. https://doi.org/10.1007/s13203-016-0157-y
Ren T, Qi W, Su R, He Z (2019) Promising techniques for depolymerization of lignin into value-added chemicals. ChemCatChem 11:639–654. https://doi.org/10.1002/cctc.201801428
Chen X, Guan W, Tsang C-W, Hu H, Liang C (2019) Lignin valorizations with Ni catalysts for renewable chemicals and fuels productions. Catalysts 9(6):488. https://doi.org/10.3390/catal9060488
Sun Z, Fridrich B, De Santi A et al (2018) Bright side of lignin depolymerization: toward new platform chemicals. Chem Rev 118:614–678. https://doi.org/10.1021/acs.chemrev.7b00588
Holladay JE, White JF, Bozell JJ, Johnson D (2007) Top value-added chemicals from biomass volume II — results of screening for potential candidates from biorefinery lignin. Prepared for the U.S. Department of Energy under Contract DE-AC05–76RL01830. II:
Bruijnincx PCA, Weckhuysen BM (2014) Biomass conversion: Lignin up for break-down. Nat Chem 6:1035–1036. https://doi.org/10.1038/nchem.2120
Dong NQ, Lin HX (2021) Contribution of phenylpropanoid metabolism to plant development and plant–environment interactions. J Integr Plant Biol 63:180–209. https://doi.org/10.1111/jipb.13054
Munk L, Sitarz AK, Kalyani DC et al (2015) Can laccases catalyze bond cleavage in lignin? Biotechnol Adv 33:13–24. https://doi.org/10.1016/j.biotechadv.2014.12.008
Parthasarathi R, Romero RA, Redondo A, Gnanakaran S (2011) Theoretical study of the remarkably diverse linkages in lignin. J Phys Chem Lett 2:2660–2666. https://doi.org/10.1021/jz201201q
Galkin MV, Samec JSM (2016) Lignin valorization through catalytic lignocellulose fractionation: a fundamental platform for the future biorefinery. Chemsuschem 9:1544–1558. https://doi.org/10.1002/cssc.201600237
Huang JB, Bin WuS, Cheng H et al (2015) Theoretical study of bond dissociation energies for lignin model compounds. Ranliao Huaxue Xuebao/J Fuel Chem Technol 43:429–436. https://doi.org/10.1016/s1872-5813(15)30011-6
Van den Bosch S, Koelewijn SF, Renders T et al (2018) Catalytic strategies towards lignin-derived chemicals. Springer International Publishing. https://link.springer.com/article/10.1007%2Fs41061-018-0214-3
Stücker A, Schütt F, Saake B, Lehnen R (2016) Lignins from enzymatic hydrolysis and alkaline extraction of steam refined poplar wood: utilization in lignin-phenol-formaldehyde resins. Ind Crops Prod 85:300–308. https://doi.org/10.1016/j.indcrop.2016.02.062
Azadi P, Inderwildi OR, Farnood R, King DA (2013) Liquid fuels, hydrogen and chemicals from lignin: a critical review. Renew Sustain Energy Rev 21:506–523. https://doi.org/10.1016/j.rser.2012.12.022
Huang C, Ma J, Zhang W, et al (2018) Preparation of lignosulfonates from biorefinery lignins by sulfomethylation and their application as a water reducer for concrete. Polymers (Basel) 10.https://doi.org/10.3390/polym10080841
Trajano HL, Engle NL, Foston M et al (2013) The fate of lignin during hydrothermal pretreatment. Biotechnol Biofuels 6:1–16. https://doi.org/10.1186/1754-6834-6-110
Villaverde JJ, Ligero P, de Vega A (2014) Miscanthus x giganteus as a source of biobased products through organosolv fractionation: a mini review. Open Agric J 4:102–110. https://doi.org/10.2174/1874331501004010102
Dapía S, Santos V, Parajó JC (2002) Study of formic acid as an agent for biomass fractionation. Biomass Bioenerg 22:213–221. https://doi.org/10.1016/S0961-9534(01)00073-3
Zhang Y, Hou Q, Xu W et al (2017) Revealing the structure of bamboo lignin obtained by formic acid delignification at different pressure levels. Ind Crops Prod 108:864–871. https://doi.org/10.1016/j.indcrop.2017.08.065
Muurinen ESA (2000) A review and distillation study related to peroxyacid. Department of Process Engineering. http://urn.fi/urn:isbn:9514256611
Quesada-Medina J, López-Cremades FJ, Olivares-Carrillo P (2010) Organosolv extraction of lignin from hydrolyzed almond shells and application of the δ-value theory. Bioresour Technol 101:8252–8260. https://doi.org/10.1016/j.biortech.2010.06.011
Zijlstra DS, De Santi A, Oldenburger B et al (2019) Extraction of lignin with high β-O-4 content by mild ethanol extraction and its effect on the depolymerization yield. J Vis Exp 2019:1–12. https://doi.org/10.3791/58575
Li J, Gellerstedt G, Toven K (2009) Steam explosion lignins; their extraction, structure and potential as feedstock for biodiesel and chemicals. Bioresour Technol. https://doi.org/10.1016/j.biortech.2008.12.004
Zhang X, Zhu J, Sun L et al (2019) Extraction and characterization of lignin from corncob residue after acid-catalyzed steam explosion pretreatment. Ind Crops Prod 133:241–249. https://doi.org/10.1016/j.indcrop.2019.03.027
Jacquet N, Maniet G, Vanderghem C et al (2015) Application of steam explosion as pretreatment on lignocellulosic material: a review. Ind Eng Chem Res 54:2593–2598. https://doi.org/10.1021/ie503151g
Bonini C, D’Auria M, Di Maggio P, Ferri R (2008) Characterization and degradation of lignin from steam explosion of pine and corn stalk of lignin: the role of superoxide ion and ozone. Ind Crops Prod 27:182–188. https://doi.org/10.1016/j.indcrop.2007.07.023
Sun XF, Xu F, Sun RC et al (2004) Characteristics of degraded lignins obtained from steam exploded wheat straw. Polym Degrad Stab 86:245–256. https://doi.org/10.1016/j.polymdegradstab.2004.05.003
Scott M, Deuss PJ, De Vries JG et al (2016) New insights into the catalytic cleavage of the lignin β-O-4 linkage in multifunctional ionic liquid media. Catal Sci Technol 6:1882–1891. https://doi.org/10.1039/c5cy01554e
Mittal A, Katahira R, Donohoe BS et al (2017) Ammonia pretreatment of corn stover enables facile lignin extraction. ACS Sustain Chem Eng 5:2544–2561. https://doi.org/10.1021/acssuschemeng.6b02892
(1956) Extraction of a lignin fraction from maple wood by SIRA Act
Da Costa SL, Jin M, Chundawat SPS et al (2016) Next-generation ammonia pretreatment enhances cellulosic biofuel production. Energy Environ Sci 9:1215–1223. https://doi.org/10.1039/c5ee03051j
Yoo CG, Kim H, Lu F et al (2016) Understanding the physicochemical characteristics and the improved enzymatic saccharification of corn stover pretreated with aqueous and gaseous ammonia. Bioenergy Res 9:67–76. https://doi.org/10.1007/s12155-015-9662-6
Balasubramanian D, Srinivas V, Gaikar VG, Sharma MM (1989) Aggregation behavior of hydrotropic compounds in aqueous solution. J Phys Chem 93:3865–3870. https://doi.org/10.1021/j100346a098
Ansari KB, Gaikar VG (2014) Green hydrotropic extraction technology for delignification of sugarcane bagasse by using alkybenzene sulfonates as hydrotropes. Chem Eng Sci 115:157–166. https://doi.org/10.1016/j.ces.2013.10.042
Rantwijk F Van, Sheldon RA (2007) cr050946x-OK.pdf
Achinivu EC, Howard RM, Li G et al (2014) Lignin extraction from biomass with protic ionic liquids. Green Chem 16:1114–1119. https://doi.org/10.1039/c3gc42306a
Zakaria SM, Idris A, Alias Y (2017) Lignin extraction from coconut shell using aprotic ionic liquids. BioResources 12:5749–5774. https://doi.org/10.15376/biores.12.3.5749-5774
Zhang J, Wang Y, Zhang L et al (2014) Understanding changes in cellulose crystalline structure of lignocellulosic biomass during ionic liquid pretreatment by XRD. Bioresour Technol 151:402–405. https://doi.org/10.1016/j.biortech.2013.10.009
Sun N, Rodríguez H, Rahman M, Rogers RD (2011) Where are ionic liquid strategies most suited in the pursuit of chemicals and energy from lignocellulosic biomass? Chem Commun 47:1405–1421. https://doi.org/10.1039/c0cc03990j
Zhao Z, Chen X, Ali MF et al (2018) Pretreatment of wheat straw using basic ethanolamine-based deep eutectic solvents for improving enzymatic hydrolysis. Bioresour Technol 263:325–333. https://doi.org/10.1016/j.biortech.2018.05.016
Long J, Li X, Guo B et al (2013) Catalytic delignification of sugarcane bagasse in the presence of acidic ionic liquids. Catal Today 200:99–105. https://doi.org/10.1016/j.cattod.2012.08.018
Dinh Vu N, Thi Tran H, Bui ND, et al (2017) Lignin and cellulose extraction from vietnam’s rice straw using ultrasound-assisted alkaline treatment Mmethod. Int J Polym Sci 2017. https://doi.org/10.1155/2017/1063695
Quesada-Medina J, López-Cremades FJ, Olivares-Carrillo P (2010) Bioresource technology organosolv extraction of lignin from hydrolyzed almond shells and application of the d-value theory. 101:8252–8260. https://doi.org/10.1016/j.biortech.2010.06.011
Gillet S, Aguedo M, Petitjean L et al (2017) Lignin transformations for high value applications: towards targeted modifications using green chemistry. Green Chem 19:4200–4233. https://doi.org/10.1039/c7gc01479a
Zhou M, Sharma BK, Liu P et al (2018) Catalytic in situ hydrogenolysis of lignin in supercritical ethanol: effect of phenol, catalysts, and reaction temperature. ACS Sustain Chem Eng 6:6867–6875. https://doi.org/10.1021/acssuschemeng.8b00701
Rashidi M, Beltramini JN, Martin D (2019) The selective cleavage of lignin aliphatic C-O linkages by solvent-assisted fast pyrolysis (SAFP). J Incl Phenom Macrocycl Chem 94:297–307. https://doi.org/10.1007/s10847-019-00905-x
Wyn HK, Konarova M, Beltramini J, et al (2020) Self-sustaining smouldering combustion of waste: a review on applications, key parameters and potential resource recovery. Fuel Process Technol 205:106425. https://doi.org/10.1016/j.fuproc.2020.106425
Kawamoto H (2017) Lignin pyrolysis reactions. J Wood Sci 63:117–132. https://doi.org/10.1007/s10086-016-1606-z
Kotake T, Kawamoto H, Saka S (2014) Mechanisms for the formation of monomers and oligomers during the pyrolysis of a softwood lignin. J Anal Appl Pyrolysis 105:309–316. https://doi.org/10.1016/j.jaap.2013.11.018
Hosoya T, Kawamoto H, Saka S (2009) Role of methoxyl group in char formation from lignin-related compounds. J Anal Appl Pyrolysis 84:79–83. https://doi.org/10.1016/j.jaap.2008.10.024
Mullen CA, Boateng AA (2010) Catalytic pyrolysis-GC/MS of lignin from several sources. Fuel Process Technol 91:1446–1458. https://doi.org/10.1016/j.fuproc.2010.05.022
Ma Z, Custodis V, Hemberger P et al (2015) Chemicals from lignin by catalytic fast pyrolysis, from product control to reaction mechanism. Chimia (Aarau) 69:597–602. https://doi.org/10.2533/chimia.2015.597
Bi Y, Lei X, Xu G, et al (2018) Catalytic fast pyrolysis of kraft lignin over hierarchical HZSM-5 and Hβ zeolites. Catalysts 8.https://doi.org/10.3390/catal8020082
Lazaridis PA, Fotopoulos AP, Karakoulia SA, Triantafyllidis KS (2018) Catalytic fast pyrolysis of kraft lignin with conventional, mesoporous and nanosized ZSM-5 zeolite for the production of alkyl-phenols and aromatics. Front Chem 6. https://doi.org/10.3389/fchem.2018.00295
Chen F, Yan B, Liu N, et al (2019) Bimetallic oriented catalytic fast pyrolysis of lignin research based on PY-GC/MS. Biomass Convers Biorefinery. https://doi.org/10.1007/s13399-019-00464-8
Han T, Ding S, Yang W, Jönsson P (2019) Catalytic pyrolysis of lignin using low-cost materials with different acidities and textural properties as catalysts. Chem Eng J 373:846–856. https://doi.org/10.1016/j.cej.2019.05.125
Cheng C, Wang J, Shen D et al (2017) Catalytic oxidation of lignin in solvent systems for production of renewable chemicals: a review. Polymers (Basel) 9:38–50. https://doi.org/10.3390/polym9060240
Li Y, Karlen SD, Demir B, et al (2020) Mechanistic study of diaryl ether bond cleavage during palladium-catalyzed lignin hydrogenolysis. ChemSusChem n/a: https://doi.org/10.1002/cssc.202000753
Bosque I, Magallanes G, Rigoulet M et al (2017) Redox catalysis facilitates lignin depolymerization. ACS Cent Sci 3:621–628. https://doi.org/10.1021/acscentsci.7b00140
Galkin MV, Sawadjoon S, Rohde V et al (2014) Mild heterogeneous palladium-catalyzed cleavage of β-o-4’-ether linkages of lignin model compounds and native lignin in air. ChemCatChem 6:179–184. https://doi.org/10.1002/cctc.201300540
Magallanes G, Kärkäs MD, Bosque I et al (2019) Selective C-O bond cleavage of lignin systems and polymers enabled by sequential palladium-catalyzed aerobic oxidation and visible-light photoredox catalysis. ACS Catal 9:2252–2260. https://doi.org/10.1021/acscatal.8b04172
Tana T, Zhang Z, Beltramini J et al (2019) Valorization of native sugarcane bagasse lignin to bio-aromatic esters/monomers: Via a one pot oxidation-hydrogenation process. Green Chem 21:861–873. https://doi.org/10.1039/c8gc03655a
Wang H, Han H, Sun E et al (2020) Production of aryl oxygen-containing compounds by catalytic pyrolysis of bagasse lignin over LaTi0.2Fe0.8O3 prepared by different methods. J Rare Earths 38:76–83. https://doi.org/10.1016/j.jre.2019.07.001
Dhar P, Vinu R (2018) Microwave-assisted catalytic solvolysis of lignin to phenols: kinetics and product characterization. ACS Omega 3:15076–15085. https://doi.org/10.1021/acsomega.8b01509
Ansaloni S, Russo N, Pirone R (2018) Wet air oxidation of industrial lignin case study: influence of the dissolution pretreatment and perovskite-type oxides. Waste and Biomass Valorization 9:2165–2179. https://doi.org/10.1007/s12649-017-9947-4
Wang X, Luo Y, Qian M, Qian EW (2020) Catalytic depolymerization of alkali lignin in ionic liquids on Pt-supported La2O3-SO42-/ZrO2 catalysts. Sustain Energy Fuels 4:1409–1416. https://doi.org/10.1039/c9se00682f
Kumar A, Chandra R (2020) Ligninolytic enzymes and its mechanisms for degradation of lignocellulosic waste in environment. Heliyon 6:e03170. https://doi.org/10.1016/j.heliyon.2020.e03170
Watanabe T, Koller K, Messner K (1998) Copper-dependent depolymerization of lignin in the presence of fungal metabolite, pyridine. J Biotechnol 62:221–230. https://doi.org/10.1016/S0168-1656(98)00063-7
Sedai B, Díaz-Urrutia C, Baker RT et al (2013) Aerobic oxidation of β-1 lignin model compounds with copper and oxovanadium catalysts. ACS Catal 3:3111–3122. https://doi.org/10.1021/cs400636k
Mottweiler J, Puche M, Räuber C et al (2015) Copper- and vanadium-catalyzed oxidative cleavage of lignin using dioxygen. Chemsuschem 8:2106–2113. https://doi.org/10.1002/cssc.201500131
Wang M, Li LH, Lu JM et al (2017) Acid promoted C-C bond oxidative cleavage of β-O-4 and β-1 lignin models to esters over a copper catalyst. Green Chem 19:702–706. https://doi.org/10.1039/c6gc02970a
Wang M, Lu J, Li L et al (2017) Oxidative C(OH)[sbnd]C bond cleavage of secondary alcohols to acids over a copper catalyst with molecular oxygen as the oxidant. J Catal 348:160–167. https://doi.org/10.1016/j.jcat.2017.02.017
Sales FG, Maranhão LCA, Filho NML, Abreu CAM (2007) Experimental evaluation and continuous catalytic process for fine aldehyde production from lignin. Chem Eng Sci 62:5386–5391. https://doi.org/10.1016/j.ces.2007.02.018
Zhang J, Deng H, Lin L (2009) Wet aerobic oxidation of lignin into aromatic aldehydes catalysed by a perovskite-type oxide: LaFe1-xCuxO3 (x=0, 0.1, 0.2). Molecules 14:2747–2757. https://doi.org/10.3390/molecules14082747
Deng H, Lin L, Sun Y et al (2008) Perovskite-type oxide LaMnO3: an efficient and recyclable heterogeneous catalyst for the wet aerobic oxidation of lignin to aromatic aldehydes. Catal Letters 126:106–111. https://doi.org/10.1007/s10562-008-9588-0
Deng H, Lin L, Sun Y et al (2009) Activity and stability of perovskite-type oxide LaCoO 3 catalyst in lignin catalytic wet oxidation to aromatic aldehydes process. Energy Fuels 23:19–24. https://doi.org/10.1021/ef8005349
Gu X, Cheng K, He M et al (2012) La-modified SBA-15/H2O2 systems for the microwave assisted oxidation of organosolv beech wood lignin. Maderas Cienc y Tecnol 14:31–41. https://doi.org/10.4067/S0718-221X2012000100003
Jha A, Patil KR, Rode CV (2013) Mixed Co-Mn oxide-catalysed selective aerobic oxidation of vanillyl alcohol to vanillin in base-free conditions. ChemPlusChem 78:1384–1392
Tarasov AL, Kustov LM, Isaeva VI et al (2011) Platinum-containing catalyst supported on a metal-organic framework structure in the selective oxidation of benzyl alcohol derivatives into aldehydes. Kinet Catal 52:273–276. https://doi.org/10.1134/S0023158411020212
Truong T, Hoang TM, Nguyen CK et al (2015) Expanding applications of zeolite imidazolate frameworks in catalysis: synthesis of quinazolines using ZIF-67 as an efficient heterogeneous catalyst. RSC Adv 5:24769–24776. https://doi.org/10.1039/c4ra16168h
Valange S, Védrine JC (2018) General and prospective views on oxidation reactions in heterogeneous catalysis. Catalysts 8.https://doi.org/10.3390/catal8100483
Crestini C, Caponi MC, Argyropoulos DS, Saladino R (2006) Immobilized methyltrioxo rhenium (MTO)/H2O2 systems for the oxidation of lignin and lignin model compounds. Bioorganic Med Chem 14:5292–5302. https://doi.org/10.1016/j.bmc.2006.03.046
Van Den Bosch S, Schutyser W, Vanholme R et al (2015) Reductive lignocellulose fractionation into soluble lignin-derived phenolic monomers and dimers and processable carbohydrate pulps. Energy Environ Sci 8:1748–1763. https://doi.org/10.1039/c5ee00204d
Figueirêdo MB, Keij FW, Hommes A, et al (2019) Efficient depolymerization of lignin to biobased chemicals using a two-step approach involving ozonation in a continuous flow microreactor followed by catalytic hydrotreatment. ACS Sustain Chem Eng. https://doi.org/10.1021/acssuschemeng.9b04020
Schutyser W, Renders T, Van Den Bosch S et al (2018) Chemicals from lignin: an interplay of lignocellulose fractionation, depolymerisation, and upgrading. Chem Soc Rev 47:852–908. https://doi.org/10.1039/c7cs00566k
Oasmaa A, Alén R, Meier D (1993) Catalytic hydrotreatment of some technical lignins. Bioresour Technol 45:189–194. https://doi.org/10.1016/0960-8524(93)90111-N
Li C, Zhao X, Wang A et al (2015) Catalytic transformation of lignin for the production of chemicals and fuels. Chem Rev 115:11559–11624. https://doi.org/10.1021/acs.chemrev.5b00155
Kloekhorst A, Heeres HJ (2016) Catalytic hydrotreatment of Alcell lignin fractions using a Ru/C catalyst. Catal Sci Technol 6:7053–7067. https://doi.org/10.1039/c6cy00523c
Mukundan S, Atanda L, Beltramini J (2019) Thermocatalytic cleavage of C-C and C-O bonds in model compounds and kraft lignin by NiMoS2/C nanocatalysts. Sustain Energy Fuels 3:1317–1328. https://doi.org/10.1039/c8se00576a
Robinson AM, Hensley JE, Will Medlin J (2016) Bifunctional catalysts for upgrading of biomass-derived oxygenates: a review. ACS Catal 6:5026–5043. https://doi.org/10.1021/acscatal.6b00923
Zhang J, Asakura H, Van Rijn J et al (2014) Highly efficient, NiAu-catalyzed hydrogenolysis of lignin into phenolic chemicals. Green Chem 16:2432–2437. https://doi.org/10.1039/c3gc42589d
Renders T, Van Den Bosch S, Koelewijn SF et al (2017) Lignin-first biomass fractionation: The advent of active stabilisation strategies. Energy Environ Sci 10:1551–1557
Bouxin FP, McVeigh A, Tran F et al (2015) Catalytic depolymerisation of isolated lignins to fine chemicals using a Pt/alumina catalyst: Part 1 - Impact of the lignin structure. Green Chem 17:1235–1242. https://doi.org/10.1039/c4gc01678e
Torr KM, van de Pas DJ, Cazeils E, Suckling ID (2011) Mild hydrogenolysis of in-situ and isolated Pinus radiata lignins. Bioresour Technol 102:7608–7611. https://doi.org/10.1016/j.biortech.2011.05.040
Margellou A, Triantafyllidis KS (2019) Catalytic transfer hydrogenolysis reactions for lignin valorization to fuels and chemicals. Catalysts 9:.https://doi.org/10.3390/catal9010043
Song Q, Wang F, Cai J, et al (2013) Lignin depolymerization (LDP) in alcohol over nickel-based catalysts via a fragmentation-hydrogenolysis process. Energy Environ Sci. https://doi.org/10.1039/c2ee23741e
Kim JY, Park J, Kim UJ, Choi JW (2015) Conversion of lignin to phenol-rich oil fraction under supercritical alcohols in the presence of metal catalysts. Energy Fuels 29:5154–5163. https://doi.org/10.1021/acs.energyfuels.5b01055
Liguori L, Barth T (2011) Palladium-Nafion SAC-13 catalysed depolymerisation of lignin to phenols in formic acid and water. J Anal Appl Pyrolysis 92:477–484. https://doi.org/10.1016/j.jaap.2011.09.004
Cheng S, Wilks C, Yuan Z et al (2012) Hydrothermal degradation of alkali lignin to bio-phenolic compounds in sub/supercritical ethanol and water-ethanol co-solvent. Polym Degrad Stab 97:839–848. https://doi.org/10.1016/j.polymdegradstab.2012.03.044
Toledano A, Serrano L, Pineda A et al (2012) Microwave-assisted depolymerisation of organosolv lignin via mild hydrogen-free hydrogenolysis: catalyst screening. Appl Catal B Environ 145:43–55. https://doi.org/10.1016/j.apcatb.2012.10.015
McVeigh A, Bouxin FP, Jarvis MC, Jackson SD (2016) Catalytic depolymerisation of isolated lignin to fine chemicals: part 2-process optimisation. Catal Sci Technol 6:4142–4150. https://doi.org/10.1039/c5cy01896j
Kong J, He M, Lercher JA, Zhao C (2015) Direct production of naphthenes and paraffins from lignin. Chem Commun 51:17580–17583. https://doi.org/10.1039/c5cc06828b
Zhang J, Teo J, Chen X et al (2014) A series of NiM (M = Ru, Rh, and Pd) bimetallic catalysts for effective lignin hydrogenolysis in water. ACS Catal 4:1574–1583. https://doi.org/10.1021/cs401199f
Kasakov S, Shi H, Camaioni DM et al (2015) Reductive deconstruction of organosolv lignin catalyzed by zeolite supported nickel nanoparticles. Green Chem 17:5079–5090. https://doi.org/10.1039/c5gc02160j
Qin Y, Wang H, Ruan H et al (2018) High catalytic efficiency of lignin depolymerization over low Pd-zeolite Y loading at mild temperature. Front Energy Res 6:1–7. https://doi.org/10.3389/fenrg.2018.00002
Regmi YN, Mann JK, McBride JR et al (2018) Catalytic transfer hydrogenolysis of organosolv lignin using B-containing FeNi alloyed catalysts. Catal Today 302:190–195. https://doi.org/10.1016/j.cattod.2017.05.051
Sergeev AG, Hartwig JF (2011) Selective, nickel-catalyzed hydrogenolysis of aryl ethers. Science (80- ) 332:439–443. https://doi.org/10.1126/science.1200437
Zhang JW, Cai Y, Lu GP, Cai C (2016) Facile and selective hydrogenolysis of β-O-4 linkages in lignin catalyzed by Pd-Ni bimetallic nanoparticles supported on ZrO2. Green Chem 18:6229–6235. https://doi.org/10.1039/c6gc02265k
Zhang B, Li W, Dou X, et al (2019) Sustainable energy & fuels catalytic depolymerization of Kraft lignin to produce liquid fuels via Ni–Sn metal oxide. https://doi.org/10.1039/c9se01089k
Guo H, Zhang B, Li C et al (2016) Tungsten carbide: a remarkably efficient catalyst for the selective cleavage of lignin C−O bonds. Chemsuschem 9:3220–3229. https://doi.org/10.1002/cssc.201600901
Guo H, Zhang B, Qi Z et al (2017) Valorization of lignin to simple phenolic compounds over tungsten carbide: impact of lignin structure. Chemsuschem 10:523–532. https://doi.org/10.1002/cssc.201601326
Wang J, Li W, Wang H, et al (2017) Liquefaction of kraft lignin by hydrocracking with simultaneous use of a novel dual acid-base catalyst and a hydrogenation catalyst. Bioresour Technol. https://doi.org/10.1016/j.biortech.2017.06.024
Jin L, Li W, Liu Q et al (2018) Liquefaction of kraft lignin over the composite catalyst HTaMoO6 and Rh/C in dioxane-water system. Fuel Process Technol 178:62–70. https://doi.org/10.1016/j.fuproc.2018.05.014
Galkin M V, Dahlstrand C, Samec JSM (2015) Mild and robust redox-neutral Pd/C-catalyzed Lignol b-O-4’ bond cleavage through a low-energy-barrier pathway. 2187–2192. https://doi.org/10.1002/cssc.201500117
Verziu M, Tirsoaga A, Cojocaru B et al (2018) Hydrogenolysis of lignin over Ru-based catalysts: the role of the ruthenium in a lignin fragmentation process. Mol Catal 450:65–76. https://doi.org/10.1016/j.mcat.2018.03.004
Huang X, Morales Gonzalez OM, Zhu J et al (2017) Reductive fractionation of woody biomass into lignin monomers and cellulose by tandem metal triflate and Pd/C catalysis. Green Chem 19:175–187. https://doi.org/10.1039/c6gc02962k
Harris EE, D’Ianni J, Adkins H (1938) Reaction of hardwood lignin with hydrogen. J Am Chem Soc 60:1467–1470. https://doi.org/10.1021/ja01273a056
Song Q, Wang F, Xu J (2012) Hydrogenolysis of lignosulfonate into phenols over heterogeneous nickel catalysts. Chem Commun 48:7019–7021. https://doi.org/10.1039/c2cc31414b
Narani A, Chowdari RK, Cannilla C et al (2015) Efficient catalytic hydrotreatment of Kraft lignin to alkylphenolics using supported NiW and NiMo catalysts in supercritical methanol. Green Chem 17:5046–5057. https://doi.org/10.1039/c5gc01643f
Raikwar D, Majumdar S, Shee D (2019) Thermocatalytic depolymerization of kraft lignin to guaiacols using HZSM-5 in alkaline water-THF co-solvent: a realistic approach. Green Chem 21:3864–3881. https://doi.org/10.1039/c9gc00593e
Klein I, Saha B, Abu-omar MM (2015) Catalysis science & technology methanol, a continuation: effect of substrate and. Catal Sci Technol 6–9. https://doi.org/10.1039/C5CY00490J
Zhang X, Zhang Q, Long J et al (2014) Phenolics production through catalytic depolymerization of alkali lignin with metal chlorides. BioResources 9:3347–3360. https://doi.org/10.15376/biores.9.2.3347-3360
Yang J, Zhao L, Liu S et al (2016) High-quality bio-oil from one-pot catalytic hydrocracking of kraft lignin over supported noble metal catalysts in isopropanol system. Bioresour Technol 212:302–310. https://doi.org/10.1016/j.biortech.2016.04.029
Hao Z, Li S, Sun J et al (2018) Efficient visible-light-driven depolymerization of oxidized lignin to aromatics catalyzed by an iridium complex immobilized on mesocellular silica foams. Appl Catal B Environ 237:366–372. https://doi.org/10.1016/j.apcatb.2018.05.072
Jastrzebski R, Constant S, Lancefield CS et al (2016) Tandem catalytic depolymerization of lignin by water-tolerant Lewis acids and rhodium complexes. Chemsuschem 9:2074–2079. https://doi.org/10.1002/cssc.201600683
Subbotina E, Galkin MV, Samec JSM (2017) Pd/C-catalyzed hydrogenolysis of dibenzodioxocin lignin model compounds using silanes and water as hydrogen source. ACS Sustain Chem Eng 5:3726–3731. https://doi.org/10.1021/acssuschemeng.7b00428
Brand S, Kim J (2015) Liquefaction of major lignocellulosic biomass constituents in supercritical ethanol. Energy 80:64–74. https://doi.org/10.1016/j.energy.2014.11.043
Ma R, Hao W, Ma X et al (2014) Catalytic ethanolysis of kraft lignin into high-value small-molecular chemicals over a nanostructured α-molybdenum carbide catalyst. Angew Chemie - Int Ed 53:7310–7315. https://doi.org/10.1002/anie.201402752
Luo L, Yang J, Yao G, Jin F (2018) Controlling the selectivity to chemicals from catalytic depolymerization of kraft lignin with in-situ H2. Bioresour Technol 264:1–6. https://doi.org/10.1016/j.biortech.2018.03.062
Jongerius AL, Copeland JR, Foo GS et al (2013) Stability of Pt/γ-Al2O3 catalysts in lignin and lignin model compound solutions under liquid phase reforming reaction conditions. ACS Catal 3:464–473. https://doi.org/10.1021/cs300684y
Blanksby SJ, Ellison GB (2003) Bond dissociation energies of organic molecules. Acc Chem Res 36:255–263. https://doi.org/10.1021/ar020230d
Abdelaziz OY, Brink DP, Prothmann J et al (2016) Biological valorization of low molecular weight lignin. Biotechnol Adv 34:1318–1346. https://doi.org/10.1016/j.biotechadv.2016.10.001
Yan N, Zhao C, Dyson PJ, et al (2008) Selective degradation of wood lignin over noble-metal catalysts in a two-step process. 626–629.https://doi.org/10.1002/cssc.200800080
Kim JY, Park SY, Choi IG, Choi JW (2018) Evaluation of RuxNi1-x/SBA-15 catalysts for depolymerization features of lignin macromolecule into monomeric phenols. Chem Eng J 336:640–648. https://doi.org/10.1016/j.cej.2017.11.118
Hita I, Deuss PJ, Bonura G et al (2018) Biobased chemicals from the catalytic depolymerization of Kraft lignin using supported noble metal-based catalysts. Fuel Process Technol 179:143–153. https://doi.org/10.1016/j.fuproc.2018.06.018
Wang H, Ruan H, Feng M et al (2017) One-pot process for hydrodeoxygenation of lignin to alkanes using Ru-based bimetallic and bifunctional catalysts supported on zeolite Y. Chemsuschem 10:1846–1856. https://doi.org/10.1002/cssc.201700160
Wang X, Rinaldi R (2016) Bifunctional Ni catalysts for the one-pot conversion of organosolv lignin into cycloalkanes. Catal Today 269:48–55. https://doi.org/10.1016/j.cattod.2015.11.047
Jongerius AL, Bruijnincx PCA, Weckhuysen BM (2013) Liquid-phase reforming and hydrodeoxygenation as a two-step route to aromatics from lignin. Green Chem 15:3049–3056. https://doi.org/10.1039/c3gc41150h
Zakzeski J, Weckhuysen BM (2011) Lignin solubilization and aqueous phase reforming for the production of aromatic chemicals and hydrogen. Chemsuschem 4:369–378. https://doi.org/10.1002/cssc.201000299
Zakzeski J, Jongerius AL, Bruijnincx PCA, Weckhuysen BM (2012) Catalytic lignin valorization process for the production of aromatic chemicals and hydrogen. Chemsuschem 5:1602–1609. https://doi.org/10.1002/cssc.201100699
Toledano A, Serrano L, Labidi J et al (2013) Heterogeneously catalysed mild hydrogenolytic depolymerisation of lignin under microwave irradiation with hydrogen-donating solvents. ChemCatChem 5:977–985. https://doi.org/10.1002/cctc.201200616
Jiang B, Hu J, Qiao Y et al (2019) Depolymerization of lignin over a Ni-Pd bimetallic catalyst using isopropanol as an in situ hydrogen source. Energy Fuels 33:8786–8793. https://doi.org/10.1021/acs.energyfuels.9b01976
Ferrini P, Rinaldi R (2014) Catalytic biorefining of plant biomass to non-pyrolytic lignin bio-oil and carbohydrates through hydrogen transfer reactions. Angew Chemie 126:8778–8783. https://doi.org/10.1002/ange.201403747
Jongerius AL, Jastrzebski R, Bruijnincx PCA, Weckhuysen BM (2012) CoMo sulfide-catalyzed hydrodeoxygenation of lignin model compounds: an extended reaction network for the conversion of monomeric and dimeric substrates. J Catal 285:315–323. https://doi.org/10.1016/j.jcat.2011.10.006
Huang S, Mahmood N, Zhang Y et al (2017) Reductive de-polymerization of kraft lignin with formic acid at low temperatures using inexpensive supported Ni-based catalysts. Fuel 209:579–586. https://doi.org/10.1016/j.fuel.2017.08.031
Wanmolee W, Beltramini JN, Atanda L et al (2019) Effect of HCOOK/ethanol on Fe/HUSY, Ni/HUSY, and Ni-Fe/HUSY catalysts on lignin depolymerization to benzyl alcohols and bioaromatics. ACS Omega 4:16980–16993. https://doi.org/10.1021/acsomega.9b02413
Licence CC (2015) Effects of mesostructured silica catalysts on the depolymerization of organosolv lignin fractionated from woody eucalyptus The Joint Graduate School of Energy and Environment ( JGSEE ). King Mongkut’s University of Technology Thonburi, Prachauthit Road
Cox BJ, Jia S, Zhang ZC, Ekerdt JG (2011) Catalytic degradation of lignin model compounds in acidic imidazolium based ionic liquids: Hammett acidity and anion effects. Polym Degrad Stab 96:426–431. https://doi.org/10.1016/j.polymdegradstab.2011.01.011
Jia S, Cox BJ, Guo X et al (2010) Decomposition of a phenolic lignin model compound over organic N-bases in an ionic liquid. Holzforschung 64:577–580. https://doi.org/10.1515/HF.2010.075
Barta K, Matson TD, Fettig ML et al (2010) Catalytic disassembly of an organosolv lignin via hydrogen transfer from supercritical methanol. Green Chem 12:1640–1647. https://doi.org/10.1039/c0gc00181c
Huang X, Atay C, Zhu J et al (2017) Catalytic depolymerization of lignin and woody biomass in supercritical ethanol: influence of reaction temperature and feedstock. ACS Sustain Chem Eng 5:10864–10874. https://doi.org/10.1021/acssuschemeng.7b02790
Singh R, Prakash A, Dhiman SK et al (2014) Hydrothermal conversion of lignin to substituted phenols and aromatic ethers. Bioresour Technol 165:319–322. https://doi.org/10.1016/j.biortech.2014.02.076
Yoshikawa T, Yagi T, Shinohara S et al (2013) Production of phenols from lignin via depolymerization and catalytic cracking. Fuel Process Technol 108:69–75. https://doi.org/10.1016/j.fuproc.2012.05.003
Canakci H, Aziz A, Celik F (2015) Soil stabilization of clay with lignin, rice husk powder and ash. Geomech Eng. https://doi.org/10.12989/gae.2015.8.1.067
Sietske Boschma D, Kees I, Kwant W. Rice straw and wheat straw - potential feedstocks for the biobased economy. Netherlands Programmes Sustainable Biomass. https://english.rvo.nl/sites/default/files/2013/12/Straw%20report%20AgNL%20June%202013.pdf
Kumar RS, Binu NK, Nishant N et al (2014) A review of bamboo based agroforestry models developed in different parts of India, productivity and marketing aspects. Bamboo Product For Non- For Areas 45–52. https://www.researchgate.net/publication/280029314_Bamboo_Productivity_in_Forest_and_Non_-_Forest_Areas_A_review_of_bamboo_based_agroforestry_models_developed_in_different_parts_of_India_productivity_and_marketing_aspects
Mustard A, Singh S (2012) India grain and feed annual. Igarss 2014(13):1–5. https://doi.org/10.1007/s13398-014-0173-7.2
Garlock RJ, Chundawat SPS, Balan V, Dale BE (2009) Optimizing harvest of corn stover fractions based on overall sugar yields following ammonia fiber expansion pretreatment and enzymatic hydrolysis. Biotechnol Biofuels 2:1–14. https://doi.org/10.1186/1754-6834-2-29
Majumder AB, Bera B, Rajan A (2010) Tea statistics: global scenario. Int J Tea Sci 8:121–124
Global data (2019) India’s phenol capacity expected to grow tenfold to 2022
Ministry of Textiles Brief/Background Note On Jute Sector. 1–23
Sharma L Sugarcane in India (Package of Practices for Different Agro-climatic Zones)
Sánchez Moral P, Ruiz Méndez MV (2006) Production of pomace olive oil. Grasas y Aceites 57.https://doi.org/10.3989/gya.2006.v57.i1.21
Chandrasekharan S, Li A (2012) Fermion bag solutions to some sign problems in four-fermion field theories. Phys Rev D - Part Fields, Gravit Cosmol 85.https://doi.org/10.1103/PhysRevD.85.091502
Plan IB (2013) GOURMET
Edrees M, Shaulkamy MS, Mabrouk SS, Abdel-Fattah AF (1976) Studies on the composition of sunflower seed heads. Qual Plant Plant Foods Hum Nutr 25:247–252. https://doi.org/10.1007/BF02590302
Asia SE, Foods G Banana 1. 1–7
Sankaran M (2016) Improvement and varietal wealth in pomegranate
Del Río JC, Rencoret J, Marques G et al (2009) Structural characterization of the lignin from jute (corchorus capsuiaris) fibers. J Agric Food Chem 57:10271–10281. https://doi.org/10.1021/jf900815x
Production S (2012) COTTON 2020 Roadmap for Sustainable Production
Ciriminna R, Fidalgo A, Meneguzzo F et al (2019) Vanillin: the case for greener production driven by sustainability megatrend. ChemistryOpen 8:660–667. https://doi.org/10.1002/open.201900083
Wendisch VF, Kim Y, Lee JH (2018) Chemicals from lignin: recent depolymerization techniques and upgrading extended pathways. Curr Opin Green Sustain Chem 14:33–39
Fache M, Boutevin B, Caillol S (2016) Vanillin production from lignin and its use as a renewable chemical. ACS Sustain Chem Eng 4:35–46. https://doi.org/10.1021/acssuschemeng.5b01344
Rawat S, Gupta P, Singh B, et al (2020) Molybdenum-catalyzed oxidative depolymerization of alkali lignin: selective production of vanillin. Appl Catal A Gen 598:117567.https://doi.org/10.1016/j.apcata.2020.117567
Das L, Kolar P, Sharma-Shivappa R et al (2017) Oxidative depolymerization of lignin using supported niobium catalysts. ChemEngineering 1:1–13. https://doi.org/10.3390/chemengineering1020017
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rishikesh, M.S., Harish, S., Mahendran Prasanth, S. et al. A comprehensive review on lignin obtained from agro-residues: potential source of useful chemicals. Biomass Conv. Bioref. 13, 5533–5556 (2023). https://doi.org/10.1007/s13399-021-01637-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13399-021-01637-0