Abstract
Photodynamic Therapy (PDT) is a promising paradigm for treating cancer, especially superficial cancers, including skin and oral cancers. However, the effectiveness of PDT is hindered by the hydrophobicity of photosensitizers. Here, chlorin e6 (Ce6), a hydrophobic photosensitizer, was loaded into pluronic F127 micelles to enhance solubility and improve tumor-specific targeting efficiency. The resulting Ce6@F127 Ms demonstrated a significant increase in solubility and singlet oxygen generation (SOG) efficiency in aqueous media compared to free Ce6. The confocal imaging and fluorescence-activated cell sorting (FACS) analysis confirmed the enhanced internalization rate of Ce6@F127 Ms in murine melanoma cell lines (B16F10) and human oral carcinoma cell lines (FaDu). Upon laser irradiation (666 nm), the cellular phototoxicity of Ce6@F127 Ms against B16F10 and FaDu was approximately three times higher than the free Ce6 treatment. The in vivo therapeutic investigations conducted on a murine model of skin cancer demonstrated the ability of Ce6@F127 Ms, when combined with laser treatment, to penetrate solid tumors effectively, which resulted in a significant reduction in tumor volume compared to free Ce6. Further, the Ce6@F127 Ms demonstrated upregulation of TUNEL-positive cells, downregulation of proliferation markers in tumor tissues, and prevention of lung metastasis with insignificant levels of proliferating cells and collagenase, as validated through immunohistochemistry. Subsequent analysis of serum and blood components affirmed the safety and efficacy of Ce6@F127 Ms in mice. Consequently, the developed Ce6@F127 Ms exhibits significant potential for concurrently treating solid tumors and preventing metastasis. The photodynamic formulation holds great clinical translation potential for treating superficial tumors.
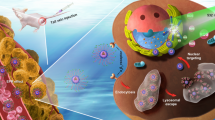
Graphical abstract












Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
References
Gupta N, Gupta R, Acharya AK, Patthi B, Goud V, Reddy S, Garg A, Singla A. Changing trends in oral cancer-a global scenario. Nepal J Epidemiol. 2016;6(4):613.
Rivera C. Essentials of oral cancer. Int J Clin Exp Pathol. 2015;8(9):11884.
Marcazzan S, Varoni EM, Blanco E, Lodi G, Ferrari M. Nanomedicine, an emerging therapeutic strategy for oral cancer therapy. Oral Oncol. 2018;76:1–7.
Saini R, Lee NV, Liu KY, Poh CF. Prospects in the application of photodynamic therapy in oral cancer and premalignant lesions. Cancers. 2016;8(9):83.
Pellosi DS, De Jesus PD, Tedesco AC. Spotlight on the delivery of photosensitizers: different approaches for photodynamic-based therapies. Expert Opin Drug Deliv. 2017;14(12):1395–406.
Shen L, Huang Y, Chen D, Qiu F, Ma C, Jin X, Zhu X, Zhou G, Zhang Z. pH-responsive aerobic nanoparticles for effective photodynamic therapy. Theranostics. 2017;7(18):4537.
Chang MH, Pai CL, Chen YC, Yu HP, Hsu CY, Lai PS. Enhanced antitumor effects of epidermal growth factor receptor targetable cetuximab-conjugated polymeric micelles for photodynamic therapy. Nanomaterials. 2018;8(2):121.
Chen K, Preuß A, Hackbarth S, Wacker M, Langer K, Röder B. Novel photosensitizer-protein nanoparticles for photodynamic therapy: photophysical characterization and in vitro investigations. J Photochem Photobiol B. 2009;96(1):66–74.
Bechet D, Couleaud P, Frochot C, Viriot ML, Guillemin F, Barberi-Heyob M. Nanoparticles as vehicles for delivery of photodynamic therapy agents. Trends Biotechnol. 2008;26(11):612–21.
Lee SJ, Koo H, Lee DE, Min S, Lee S, Chen X, et al. Tumor-homing photosensitizer-conjugated glycol chitosan nanoparticles for synchronous photodynamic imaging and therapy based on cellular on/off system. Biomaterials. 2011;32:4021e9.
Bonnett R. Photosensitizers of the porphyrin and phthalocyanine series for photodynamic therapy. Chem Soc Rev. 1995;24(1):19–33.
Lukyanov AN, Torchilin VP. Micelles from lipid derivatives of water-soluble polymers as delivery systems for poorly soluble drugs. Adv Drug Deliv Rev. 2004;56(9):1273–89.
Hamblin MR, Miller JL, Rizvi I, Ortel B, Maytin EV, Hasan T. Pegylation of a chlorin e6 polymer conjugate increases tumor targeting of photosensitizer. Cancer Res. 2001;61(19):7155–62.
Wang M, Lu P, Wu B, Tucker JD, Cloer C, Lu Q. High efficiency and low toxicity of polyethyleneimine modified Pluronics (PEI–Pluronic) as gene delivery carriers in cell culture and dystrophic mdx mice. J Mater Chem. 2012;22(13):6038–46.
Alakhov V, Lemieux P, Klinski E, Kabanov A, Pietrzynski G. Block copolymeric biotransport carriers as versatile vehicles for drug delivery. Expert Opin Biol Ther. 2001;1(4):583–602.
Batrakova EV, Li S, Elmquist WF, Miller DW, Alakhov VY, Kabanov AV. Mechanism of sensitization of MDR cancer cells by Pluronic block copolymers: selective energy depletion. Br J Cancer. 2001;85(12):1987–97.
Batrakova EV, Li S, Brynskikh AM, Sharma AK, Li Y, Boska M, Gong N, Mosley RL, Alakhov VY, Gendelman HE, Kabanov AV. Effects of pluronic and doxorubicin on drug uptake, cellular metabolism, apoptosis and tumor inhibition in animal models of MDR cancers. J Control Release. 2010;143(3):290–301.
Batrakova EV, Han HY, Alakhov VY, Miller DW, Kabanov AV. Effects of pluronic block copolymers on drug absorption in Caco-2 cell monolayers. Pharm Res. 1998;15:850–5.
Kumari P, Jain S, Ghosh B, Zorin V, Biswas S. Polylactide-based block copolymeric micelles loaded with chlorin e6 for photodynamic therapy: in vitro evaluation in monolayer and 3D spheroid models. Mol Pharm. 2017;14(11):3789–800.
Vysyaraju NR, Paul M, Ch S, Ghosh B, Biswas S. Olaparib@ human serum albumin nanoparticles as sustained drug-releasing tumour-targeting nanomedicine to inhibit growth and metastasis in the mouse model of triple-negative breast cancer. J Drug Target. 2022;30(10):1088–105.
Das S, Paul M, Ghosh B, Biswas S. Synergistic anticancer response via docetaxel-and oleanolic acid-loaded albumin/poly (lactide) nanoparticles in triple-negative breast cancer. ACS Appl Nano Mater. 2023;6(21):19710–26.
Itoo AM, Paul M, Ghosh B, Biswas S. Polymeric graphene oxide nanoparticles loaded with doxorubicin for combined photothermal and chemotherapy in triple negative breast cancer. Biomaterials Adv. 2023;153:213550.
Paul M, Itoo AM, Ghosh B, Biswas S. Hypoxia alleviating platinum (IV)/chlorin e6-based combination chemotherapeutic-photodynamic nanomedicine for oropharyngeal carcinoma. J Photochem Photobiol B. 2023;238:112627.
Jain R, Paul M, Padaga SG, Dubey SK, Biswas S, Singhvi G. Dual-drug-loaded assisted topical delivery of photodynamically active lipid-based formulation for combination therapy of cutaneous melanoma. Mol Pharm. 2023;20(7):3653–71.
Swetha KL, Paul M, Maravajjala KS, Kumbham S, Biswas S, Roy A. Overcoming drug resistance with a docetaxel and disulfiram loaded pH-sensitive nanoparticle. J Control Release. 2023;356:93–114.
Patel T, Itoo AM, Paul M, Kondapaneni LP, Ghosh B, Biswas S. Block HPMA-based pH-sensitive gemcitabine pro-drug nanoaggregates for cancer treatment. Eur Polymer J. 2023;186:111843.
Kumbham S, Paul M, Itoo A, Ghosh B, Biswas S. Oleanolic acid-conjugated human serum albumin nanoparticles encapsulating doxorubicin as synergistic combination chemotherapy in oropharyngeal carcinoma and melanoma. Int J Pharm. 2022;614:121479.
Pulya S, Patel T, Paul M, Adhikari N, Banerjee S, Routholla G, Biswas S, Jha T, Ghosh B. Selective inhibition of histone deacetylase 3 by novel hydrazide based small molecules as therapeutic intervention for the treatment of cancer. Eur J Med Chem. 2022;238:114470.
Pulya S, Himaja A, Paul M, Adhikari N, Banerjee S, Routholla G, Biswas S, Jha T, Ghosh B. Selective HDAC3 inhibitors with potent in vivo antitumor efficacy against triple-negative breast cancer. J Med Chem. 2023;66(17):12033–58.
Acknowledgements
Mr. Milan Paul acknowledges the Indian Council of Medical Research (ICMR) for providing a senior research fellowship. The authors are extremely grateful to the Central Analytical Laboratory at the Birla Institute of Technology and Science (BITS), Pilani, Hyderabad Campus, India, for providing the instrumentation facilities such as XRD, DSC, FACS, and SEM for various characterizations.
Funding
The work was funded by the Indian Council of Medical Research, Government of India, to Swati Biswas through grant number 2021/8945.
Author information
Authors and Affiliations
Contributions
Milan Paul: Methodology, Validation, Investigation, Data curation, Writing-Original draft preparation, Editing. Balaram Ghosh: Conceptualization, Validation, Writing- Reviewing and Editing, Supervision. Swati Biswas: Conceptualization, Validation, Writing- Reviewing and Editing, Supervision.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
In vivo experiments adhered to CPCSEA guidelines (BITS-Hyd/IAEC/2023/36) approved by IAEC, Department of Pharmacy, BITS Pilani, Hyderabad.
Consent for publication
The authors declare that all authors have been actively involved in the work leading to this paper, and all take responsibility for the published work.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Paul, M., Ghosh, B. & Biswas, S. F127/chlorin e6-nanomicelles to enhance Ce6 solubility and PDT-efficacy mitigating lung metastasis in melanoma. Drug Deliv. and Transl. Res. (2024). https://doi.org/10.1007/s13346-024-01619-5
Accepted:
Published:
DOI: https://doi.org/10.1007/s13346-024-01619-5