Abstract
The purpose of this study was to conduct the kinetic assessment of iontophoretic delivery of niosomal tetracycline-HCl formulated in an electroconductive gel. Tween-80 and Span-80 were used to obtain tetracycline-HCl niosomes with an average diameter of 101.9 ± 3.3 nm, a polydispersity index of 0.247 ± 0.004, a zeta potential of − 34.1 mV, and an entrapment efficiency of 70.08 ± 0.16%. Four different gel preparations, two of which contained niosomal tetracycline-HCl, were transdermally delivered using Franz diffusion cells under the trigger effect of iontophoresis, applied at 0.2, 0.5, and 1 mA/cm2 current density. The control group was the passive diffusion results of the preparation made using a tetracycline-HCl-based drug marketed in Turkey. The control group was compared with the groups that contained (a) tetracycline-HCl in an electroconductive gel, (b) the niosomal tetracycline-HCl formulation in water, and (c) the niosomal tetracycline-HCl formulation in the electroconductive gel. The group with the niosomal formulation in the electroconductive gel displayed the highest increase in iontophoretic transdermal delivery relative to the control group, displaying a 2-, 2.1-, and 2.2-fold increase, respectively, by current density. The experimental results of transdermal delivery using the synergistic effect of niosomal formulation in electroconductive gel and the trigger effect of iontophoresis appeared to divert slightly from zero-order kinetics, demonstrating a statistically significant increase in the rate of controlled transdermal drug delivery. Considering that about 20% of the formulation is transdermally delivered in the first half-hour, the iontophoretic transdermal delivery of niosomal tetracycline-HCl can be efficiently used in local iontophoretic therapy.
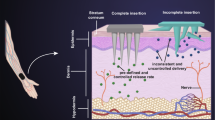
Graphical Abstract







Similar content being viewed by others
Availability of data and materials
If needed, the datasets used in this study can be obtained from the corresponding author upon reasonable request.
References
Lee H, Song C, Baik S, Kim D, Hyeon T, Kim DH. Device-assisted transdermal drug delivery. Adv Drug Deliver Rev. 2018;127:35–45. https://doi.org/10.1016/j.addr.2017.08.009.
Leite-Silva VR, de Almeida MM, Fradin A, Grice JE, Roberts MS. Delivery of drugs applied topically to the skin. Expert Rev Dermatol. 2012;7:383–97. https://doi.org/10.1586/edm.12.32.
Schreier H, Bouwstra J. Liposomes and niosomes as topical drug carriers: dermal and transdermal drug delivery. J Control Rel. 1994;30:1–15. https://doi.org/10.1016/0168-3659(94)90039-6.
Goyal R, Macri LK, Kaplan HM, Kohn J. Nanoparticles and nanofibers for topical drug delivery. J Control Rel. 2016;240:77–92. https://doi.org/10.1016/j.jconrel.2015.10.049.
Petrilli R, Lopez RF. Physical methods for topical skin drug delivery: concepts and applications. Brazilian J Pharm Sci. 2018;54. https://doi.org/10.1590/s2175-97902018000001008.
Mitragotri S. Devices for overcoming biological barriers: the use of physical forces to disrupt the barriers. Adv Drug Deliver Rev. 2013;65:100–3. https://doi.org/10.1016/j.addr.2012.07.016.
Kauscher U, Holme MN, Björnmalm M, Stevens MM. Physical stimuli-responsive vesicles in drug delivery: beyond liposomes and polymersomes. Adv Drug Deliver Rev. 2019;138:259–75. https://doi.org/10.1016/j.addr.2018.10.012.
Jain S, Chaudhari BH, Swarnakar NK. Preparation and characterization of niosomal gel for iontophoresis mediated transdermal delivery of isosorbide dinitrate. Drug Deliver Trans Res. 2011;1:309–21. https://doi.org/10.1007/s13346-011-0035-1.
Seleci DA, Seleci M, Walter J-G, Stahl F, Scheper T. Niosomes as nanoparticular drug carriers: fundamentals and recent applications. J Nanomater. 2016;1. https://doi.org/10.1155/2016/7372306.
Hamishehkar H, Hasanpouri A, Lotfipour F, Ghanbarzadeh S. Improvement of dermal delivery of tetracycline using vesicular nanostructures. Res Pharm Sci. 2018;13:385. https://doi.org/10.4103/1735-5362.236831.
Hua S, Wu SY. The use of lipid-based nanocarriers for targeted pain therapies. Front Pharmacol. 2013;4:143. https://doi.org/10.3389/fphar.2013.00143.
Zylberberg C, Matosevic S. Pharmaceutical liposomal drug delivery: a review of new delivery systems and a look at the regulatory landscape. Drug Deliver. 2016;23:3319–29. https://doi.org/10.1080/10717544.2016.1177136.
Metselaar JM, Storm G. Liposomes in the treatment of inflammatory disorders. Expert Opin Drug Deliv. 2005;2:465–76.
Abdelkader H, Alani AW, Alany RG. Recent advances in non-ionic surfactant vesicles (niosomes): self-assembly, fabrication, characterization, drug delivery applications and limitations. Drug Deliv. 2013;21:87–100.
Muzzalupo R, Tavano L. Niosomal drug delivery for transdermal targeting: recent advances. Res Reports Transdermal Drug Deliv. 2015;4:23–33.
Shirsand SB, Kanani KM, Keerthy D, Nagendrakumar D, Para MS. Formulation and evaluation of ketoconazole niosomal gel drug delivery system. Int J Pharm Invest. 2012;2:201.
Patel KK, Kumar P, Thakkar HP. Formulation of niosomal gel for enhanced transdermal lopinavir delivery and its comparative evaluation with ethosomal gel. AAPS Pharm Sci Tech. 2012;13:1502–10.
Bhardwaj P, Tripathi P, Gupta R, Pandey S. Niosomes: a review on niosomal research in the last decade. J Drug Deliver Sci Technol. 2020;56: 101581.
Wang Y, Thakur R, Fan Q, Michniak B. Transdermal iontophoresis: combination strategies to improve transdermal iontophoretic drug delivery. Europ J Pharm Biopharm. 2005;60:179–91.
Wang Y, Zeng L, Song W, Liu J. Influencing factors and drug application of iontophoresis in transdermal drug delivery: an overview of recent progress. Drug Deliv Trans Res. 2021. https://doi.org/10.1007/s13346-021-00898-6.
Barry BW. Lipid-protein-partitioning theory of skin penetration enhancement. J Control Rel. 1991;15:237–48.
Guy RH, Kalia YN, Delgado-Charro MB, Merino V, López A, Marro D. Iontophoresis: electrorepulsion and electroosmosis. J Control Rel. 2000;64:129–32.
Williams AC, Barry BW. Skin absorption enhancers. Crit Rev Therap Drug Carrier Sys. 1992;9(3–4):305–53.
Qiang Z, Adams C. Potentiometric determination of acid dissociation constants (pKa) for human and veterinary antibiotics. Water Res. 2004;38:2874–90.
Manjunatha RG, Prasad R, Sharma S, Narayan RP, Koul V. Iontophoretic delivery of lidocaine hydrochloride through ex-vivo human skin. J Dermatol Treat. 2019;31:191–9.
Chenwei S, Bu N, Hu X. Recent trends in electronic skin for transdermal drug delivery. Int Pharm. 2023;10. https://doi.org/10.1016/j.ipha.2023.08.001.
Bezruk I, Vrakin V, Savchenko L, Materiienko A, Georgiyants V. Development and validation of tetracycline hydrochloride assay procedure by spectrophotometry in compounded ointment. Scrip Sci Pharm. 2017;4. https://doi.org/10.14748/ssp.v4i1.2117.
Tavano L, Picci N, Ioele G, Muzzalupo R. Tetracycline-niosomes versus tetracycline hydrochloride-niosomes: how to modulate encapsulation and percutaneous permeation properties. J Drug. 2017;11–6. https://doi.org/10.24218/jod.2017.6.
Abdelbary G, El-Gendy N. Niosome-encapsulated gentamicin for ophthalmic controlled delivery. AAPS PharmSciTech. 2008;9(3):740–7. https://doi.org/10.1208/s12249-008-9105-1.
Bakshi P, Vora D, Hemmady K, Banga AK. Iontophoretic skin delivery systems: success and failures. İnt J Pharm. 2023;1–9. https://doi.org/10.1016/j.ipha.2023.08.001
Juárez-Osornio C, Gracia-Fadrique J. Structures similar to lipid emulsions and liposomes. Dipalmitoylphosphatidylcholine, cholesterol, Tween 20–Span 20 or Tween 80–Span 80 in aqueous media. J Liposome Res. 2016;(27):139–50.
Shalaby AR, Salama NA, Abou-Raya SH, Emam WH, Mehaya FM. Validation of HPLC method for determination of tetracycline residues in chicken meat and liver. Food Chem. 2011;124:1660–6.
Yasin U, Bilal M, Bashir H, Amirzada MI, Sumrin A, Asad MH. Preparation and nanoencapsulation of lectin from Lepidium sativum on chitosan-tripolyphosphate nanoparticle and their cytotoxicity against hepatocellular carcinoma cells (HepG2). BioMed Res Int. 2020;1–11:2020.
Wertz PW, Stover PM, Abraham W, Downing DT. Lipids of chicken epidermis. J Lipid Res. 1986;27:427–35.
Reza MI, Goel D, Gupta RK, Warsi MH. Formulation of ketoconazole loaded nano dispersive gel using swollen micelles technique and its in vitro characterization. Int J Pharm Pharmaceut Sci. 2018;10(3):162–6.
Phipps JB, Padmanabhan RV, Lattin GA. Iontophoretic delivery model inorganic and drug ions. J Pharmaceut Sci. 1989;78:365–9.
Mathematical models of drug release. In: Marcos Luciano Bruschi (Ed). Strategies to modify the drug release from pharmaceutical systems. Woodhead Publishing. 2015;63–6. ISBN 9780081000922. https://doi.org/10.1016/B978-0-08-100092-2.00005-9.
Espinal-Ruiz M, Restrepo-Sánchez L-P, Narváez-Cuenca C-E. Effect of pectins on the mass transfer kinetics of monosaccharides, amino acids, and a corn oil-in-water emulsion in a Franz diffusion cell. Food Chem. 2016;209:144–53.
Duman G, Aslan I, Ozer AY, Inanc I, Taralp A. Liposome, gel and lipogelosome formulations containing sodium hyaluronate. J Liposome Res. 2014;24(4):259–69. https://doi.org/10.3109/08982104.2014.907305.
Yaghoobian M, Haeri A, Bolourchian N, Shahhosseni S, Dadashzadeh S. The impact of surfactant composition and surface charge of niosomes on the oral absorption of repaglinide as a BCS II model drug. Int J Nanomedicine. 2020;15:8767–878. https://doi.org/10.2147/IJN.S261932.
Croitoru C, Roata IC, Alexandru P, Elena-Manuela S. Diffusion and controlled release in physically crosslinked poly (vinyl alcohol)/iota-carrageenan hydrogel blends. Polymers. 2020;7(12):1544. https://doi.org/10.3390/polym12071544.
Awad D, Tabod I, Lutz S, Wessolowski H, Gabel D. Interaction of Na2B12H11SH with liposomes: influence on zeta potential and particle size. J Organometal Chem. 2005;690(11):2732–5.
Kumar R, Bhupinder S, Bakshi G, Prakash O. Development of liposomal systems of finasteride for topical applications: design, characterization, and in vitro evaluation. Pharmaceut Develop Technol. 2007;12(6):591–1.
Akbarzadeh A, Rezaei-Sadabady R, Davaran S, Joo SW, Zarghami N, Hanifehpour Y, Samiei M, Kouhi M, Nejati-Koshki K. Liposome: classification, preparation, and applications. Nanoscale Res Lett. 2013;8. https://doi.org/10.1186/1556-276x-8-102.
Guerrini L, Alvarez-Puebla RA, Pazos-Perez N. Surface modifications of nanoparticles for stability in biological fluids. Materials. 2018;11:1154.
Guy RH, Delgado-Charro MB, Kalia YN. Iontophoretic transport across the skin. Skin Pharmacol Appl Skin Physiol. 2001;14:35–40. https://doi.org/10.1159/000056388.
Rouser G, Fleischer S, Yamamoto A. Two dimensional thin layer chromatographic separation of polar lipids and determination of phospholipids by phosphorous analysis of spots. Lipids. 1970;5:494–6.
Fang J-Y, Hong C-T, Chiu W-T, Wang Y-Y. Effect of liposomes and niosomes on skin permeation of enoxacin. Int J Pharmaceut. 2001;219:61–72.
Chang W-K, Tai Y-J, Chiang C-H, Hu C-S, Hong P-D, Yeh M-K. The comparison of protein-entrapped liposomes and lipoparticles: preparation, characterization, and efficacy of cellular uptake. Int J Nanomed. 2011;6:2403–17.
Vinograd AM, Fasina A, Dean AJ, Shofer F, Panebianco NL, Lewiss RE, Gupta S, Rao AK, Henwood PC. Evaluation of noncommercial ultrasound gels for use in resource-limited settings. J Ultrasound Med. 2018;38:371–7.
Wang Y, Qu Z, Wang W, Yu D. PVA/CMC/PEDOT: PSS mixture hydrogels with high response and low impedance electronic signals for ECG monitoring. Colloids Surf B: Biointerfaces. 2021;208: 112088.
Li SK, Peck KD. Passive and iontophoretic transport through the skin polar pathway. Skin Pharmacol Physiol. 2013;26:243–53. https://doi.org/10.1159/000351926.
Nugroho AK, Della Pasqua O, Danhof M, Bouwstra JA. Compartmental modeling of transdermal iontophoretic transport: I. In vitro model derivation and application. Pharm Res. 2004;21:1974–84. https://doi.org/10.1023/B:PHAM.0000048187.54125.ac.
Takeuchi I, Takeshita T, Suzuki T, Makino K. Iontophoretic transdermal delivery using chitosan-coated PLGA nanoparticles for positively charged drugs. Colloids Surf B Biointerfaces. 2017;160:520–6.
Funding
No funding agency, whether public, commercial, or not-for-profit, provided any specific grant for this research.
Author information
Authors and Affiliations
Contributions
SS, EG, MM, BU: methodology, validation, data curation, formal analysis. BU: imaging and instrumentation. MM: statistical analysis. EG: iontophoresis, kinetics, draft writing. MO: kinetics; GD, FSU: conceptualization, methodology, kinetics, draft writing, model writing, writing—review and editing, supervision, project administration, formal analysis.
Corresponding author
Ethics declarations
Consent for publication
Not applicable. No human studies have been performed in this research.
Competing interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Duman, G., Gucu, E., Utku, F.S. et al. Kinetic assessment of iontophoretic delivery efficiency of niosomal tetracycline hydrochloride incorporated in electroconductive gel. Drug Deliv. and Transl. Res. 14, 1206–1217 (2024). https://doi.org/10.1007/s13346-023-01452-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13346-023-01452-2