Abstract
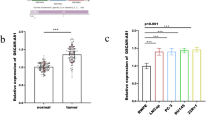
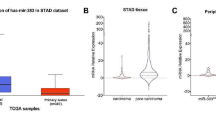
The aim of study was to discuss the correlation and regulatory mechanism of HIF-1 and miR-191 expression in pancreatic tumor. The association between the miR-191 and the clinicopathologic characteristics and the prognosis of pancreatic cancer was further explored. After hypoxic cultured for 6 and 12 h, qRT-PCR and Western blot were practiced to analyze the miR-191 and HIF-1 expression of MIA PaCa-2 and Aspac1 cells. We regulated the HIF-1 expression via plasmid and siRNA transfection to observe the alteration of HIF-1 and miR-191 expression. ChIP sequencing identified the binding sites of HIF-1 and miR-191. Dual luciferase assays were practiced to verify the binding sites. Immunohistochemical staining was practiced to analyze the expression of HIF-1, while qRT-PCR were done for investigating miR-191 in tumor tissues. Then, the association between the expression of them and the clinicopathologic characteristics and prognosis of pancreatic cancer were analyzed. After hypoxic cultured 12 h, the expression of HIF-1 protein, HIF-1mRNA and miR-191 of MIA PaCa-2 and AsPC-1 cells increased significantly (P < 0.05). After HIF-1 overexpressing plasmid transfected to the MIA PaCa-2 and AsPC-1 cells for 48 h, the expression of HIF-1 protein, HIF-1mRNA, and miR-191 upregulated significantly (P < 0.05). While after transfected the MIA PaCa-2 cells by HIF-1 siRNA, the significant decreasing of HIF-1 protein, HIF-1mRNA, and miR-191 expression were observed (P < 0.05). ChIP sequencing showed the protein synthesis of HIF-1 increased in hypoxia situation. Only the HRE5 (−1,169 bp, ChIP4) were significantly brighter in hypoxia in comparing with normoxic cells. In dual luciferase assays, after pGL3-miR-191 and HIF-1 overexpressing plasmid co-transfect the MIAPaCa-2 cells for 48 h, its relative expression of bioluminescence was higher than those co-transfected by mutant miR-191 vectors and HIF-1 overexpressing plasmid or by pGL3-miR-191 and HIF-1 empty plasmid. The expression of miR-191 closely associated with the tumor size, pTNM stage, lymph node metastasis, and perineural invasion (P < 0.05). Patients with higher expression of miR-191 were a risk factor for prognosis of pancreatic cancers. Expression of HIF-1 in pancreatic cancer cells increased under the condition of chronic hypoxia, which may bind to HRE2 in 5'flanking region of miR-191 and initiate transcription of miR-191. Expression of miR-191 was significantly higher in pancreatic tumor tissues. The expression of miR-191 closely associated with the tumor size, pTNM stage, lymph node metastasis and perineural invasion and poor prognosis of pancreatic cancer.





Similar content being viewed by others
References
Sun W, Julie L, Huang HD, Shyy JY, Chien S. microRNA: a master regulator of cellular processes for bioengineering systems. Annu Rev Biomed Eng. 2010;12:1–27.
Cecilia D, Simona G, Fabio M, et al. miR-210: more than a silent player in hypoxia. IUBMB Life. 2011;2(63):94–100.
Berezikov E, Furyev V, van de Belt J, Wienholds E, Plasterk RH, Cuppen E. Phylogenetic shadowing and computational identification of human microRNA genes. Cell. 2005;120(1):21–4.
Ventura A, Jacks T. MicroRNAs and cancer: short RNAs go a long way. Cell. 2009;136:586–91.
Song T, Zhang X, Wang C, et al. MiR-138 suppresses expression of hypoxia-inducible factor 1α (HIF-1α) in clear cell renal cell carcinoma 786-O cells. Asian Pac J Cancer Prev. 2011;12(5):1307–11.
Srinivas V, David L, Anders K, et al. MiRNA expression in urothelial carcinomas: important roles of miR-10a, miR-222, miR-125b, miR-7 and miR-452 for tumor stage and metastasis, and frequent homozygous losses of miR-31. Int J Cancer. 2009;124(9):2236–42.
Jun Y, Kenoki O, Kazuhiro M, Jun Y, Kenoki O, Kazuhiro M, et al. MicroRNA miR-17-5p is overexpressed in pancreatic cancer, associated with a poor prognosis, and involved in cancer cell proliferation and invasion. Cancer Biol Ther. 2010;8(10):748–57.
Kizaka-Kondoh S, Tanaka S, Harada H, et al. The HIF-1-active microenvironment: an environmental target for cancer therapy. Adv Drug Deliv Rev. 2009;61(7–8):623–32.
Loscalzo J. The cellular response to hypoxia: tuning the system with microRNAs. J Clin Invest. 2010;120(11):3815–7.
Stephen YC, Joseph L. MicroRNA-210 a unique and pleiotropic hypoxamir. Cell Cycle. 2010;6(9):1072–83.
Rane S et al. Downregulation of miR-199a derepresses hypoxia-inducible factor-1alpha and Sirtuin 1 and recapitulates hypoxic preconditioning in cardiac myocytes. Circ Res. 2009;104(7):879–86.
Strimpakos AS, Syrigos KN, Saif MW. The molecular targets for the diagnosis and treatment of pancreatic cancer[J]. Gut Liver. 2010;12(4):433–49. doi:10.5009/gnl.2010.4.4.433.
Greither T, Grochola LF, Udelnow A, et al. Elevated expression of microRNAs 155, 203, 210 and 222 in pancreatic tumors is associated with poorer survival[J]. Int J Cancer. 2010;126(1):73–80. doi:10.1002/ijc.24687.
Liu R, Chen X, Du Y, et al. Serum MicroRNA Expression Profile as a Biomarker in the Diagnosis and Prognosis of Pancreatic Cancer[J]. Clin Chem. 2012;58(3):610–8. doi:10.1373/clinchem.2011.172767.
Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2- △△ Ct method[J]. Methods. 2001;25(4):402–8.
Zhao T, Gao S, Wang X, Liu J, Duan Y, et al. Hypoxia-inducible factor-1α regulates chemotactic migration of pancreatic ductal adenocarcinoma cells through directly transactivating the CX3CR1 gene. PLoS One. 2012;7(8):e43399. doi:10.1371/journal.pone.0043399.
Kulshreshthar R, Ferracin M, Wojcik SE, et al. A microRNA signature of hypoxia[J]. Mol Cell Biol. 2007;27:1859–67.
Garzon R, Volinia S, Liu CG, et al. MicroRNA signatures associated with cytogenetics and prognosis in acute myeloid leukemia[J]. Blood. 2008;111(6):3183–9. doi:10.1182/blood-2007-07-098749.
Xi Y, Formentini A, Chien M, et al. Prognostic values of microRNAs in colorectal cancer[J]. Biomark Insights. 2006;2:113–21.
Volinia S, Calin GA, Liu CG, et al. A microRNA expression signature of human solid tumors defines cancer gene targets[J]. Proc Natl Acad Sci. 2006;103(7):2257–61. doi:10.1073/pnas.0510565103.
Calin GA, Liu CG, Sevignani C, Ferracin M, Felli N, et al. MicroRNA profiling reveals distinct signatures in B cell chronic lymphocytic leukemias[J]. Proc Natl Acad Sci. 2004;101(32):11755–60. doi:10.1073/pnas.0404432101.
He Y, Cui Y, Wang W, et al. Hypomethylation of the hsa-miR-191 locus causes high expression of hsa-miR-191 and promotes the epithelial-to-mesenchymal transition in hepatocellular carcinoma[J]. Neoplasia. 2011;13(9):841–53.
Sengupta S, Kim KS, Berk MP, et al. Lysophosphatidic acid downregulates tissue inhibitor of metalloproteinases, which are negatively involved in lysophosphatidic acid-induced cell invasion[J]. Oncogene. 2007;26:2894–901.
Piper M, Barry G, Hawkins J, et al. NFIA controls telencephalic progenitor cell differentiation through repression of the Notch effector Hes1[J]. J Neurosci. 2010;30(27):9127–39. doi:10.1523/JNEUROSCI.6167-09.2010.
Starnes LM, Sorrentino A, Ferracin M, et al. A transcriptome-wide approach reveals the key contribution of NFI-A in promoting erythroid differentiation of human CD34(+) progenitors and CML cells[J]. Leukemia. 2010;24:1220–3. doi:10.1038/leu.2010.78.
Koli K, Ryynanen MJ, Keski-Oja J. Latent TGF-beta binding proteins (LTBPs)-1 and -3 coordinate proliferation and osteogenic differentiation of human mesenchymal stem cells[J]. Bone. 2008;43(4):679–88. doi:10.1016/j.bone.2008.06.016.
Chandramouli A, Simundza J, Pinderhughes A, et al. Choreographing metastasis to the tune of LTBP[J]. J Mammary Gland Biol Neoplasia. 2011;16:67–80. doi:10.1007/s10911-011-9215-3.
Soutar AK. Unexpected roles for PCSK9 in lipid metabolism[J]. Curr Opin Lipidol. 2011;22(3):192–6. doi:10.1097/MOL.0b013e32834622b5.
Grentzmann G, Ingram JA, Kelly PJ, et al. A dual-luciferase reporter system for studying recoding signals[J]. RNA. 1998;4(4):479–86.
Shi X, Su S, Long J, et al. MicroRNA-191 targets N-deacetylase/N-sulfotransferase 1 and promotes cell growth in human gastric carcinoma cell line MGC803[J]. Acta Biochim Biophys Sin. 2011;43(11):849–56. doi:10.1093/abbs/gmr084.
Elyakim E, Sitbon E, Faerman A, et al. hsa- miR-191 is a candidate oncogene target for hepatocellular carcinoma therapy[J]. Cancer Res. 2010;70:8077–87. doi:10.1158/0008-5472.CAN-10-1313.
Di Leva GD, Piovan C, Gasparini P, et al. Estrogen mediated-activation of miR-191/425 cluster modulates tumorigenicity of breast cancer cells depending on estrogen receptor status[EB/OL]. PLoS Genet. 2013;9(3):e1003311. doi:10.1371/journal.pgen.1003311.
Caramuta S, Egyhazi S, Rodolfo M, et al. MicroRNA expression profiles associated with mutational status and survival in malignant melanoma[J]. J Investig Dermatol. 2010;130:2062–70. doi:10.1038/jid.2010.63.
Hui AB, Shi W, Boutros PC, et al. Robust global micro-RNA profiling with formalin-fixed paraffin-embedded breast cancer tissues[J]. Lab Investig. 2009;89:597–606. doi:10.1038/labinvest.2009.12.
Shen J, DiCioccio R, Odunsi K, et al. Novel genetic variants in miR-191 gene and familial ovarian cancer[J]. BMC Cancer. 2010;10:47–56. doi:10.1186/1471-2407-10-47.
Wynendaele J, Bohnke A, Leucci E, et al. An illegitimate microRNA target site within the 3′ UTR of MDM4 affects ovarian cancer progression and chemosensitivity[J]. Cancer Res. 2010;70:9641–9. doi:10.1158/0008-5472.CAN-10-0527.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Song, Z., Ren, H., Gao, S. et al. The clinical significance and regulation mechanism of hypoxia-inducible factor-1 and miR-191 expression in pancreatic cancer. Tumor Biol. 35, 11319–11328 (2014). https://doi.org/10.1007/s13277-014-2452-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-014-2452-5