Abstract
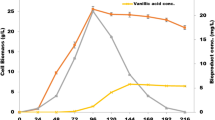
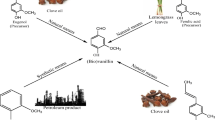
Ferulic acid is a known precursor for vanillin production but the significance of agro waste as substrates for its extraction, in combination with microbes is a less explored option. Various lactic acid bacteria were screened for the production of ferulic acid esterase (FAE) and Enterococcus lactis SR1 was found to produce maximum FAE (7.54 ± 0.15 IU/ml) in the synthetic medium under submerged fermentation. To make the process cost effective, various lignocellulosic agroresidues were evaluated for the production of FAE from the bacterium. It was found that wheat bran serves as the best substrate for FAE production (4.18 ± 0.12 IU/ml) from E. lactis SR1. Further, optimization of fermentation conditions for FAE production from E. lactis SR1 using wheat bran as carbon source led to an increase in the enzyme production (7.09 ± 0.21 IU/ml) by 1.5 fold. The FAE produced was used alone or in combination with commercial holocellulase for biological release of FA from the tested agroresidues. The highest release of FA (mg/g) by enzymatic extraction occurred in sugarbeet pulp (2.56), followed by maize bran (1.45), wheat bran (1.39) and rice bran (0.87), when both the enzymes (FAE and holocellulase) were used together. Alkaline extraction and purification of ferulic acid (FA) from these agro residues also showed that sugarbeet pulp contains the highest amount of FA (5.5 mg/g) followed by maize bran (3.0 mg/g), wheat bran (2.8 mg/g) and rice bran (1.9 mg/g), similar to the trend obtained in biological/enzymatic extraction of FA from these residues. Furthermore, the substrates were found to release higher reducing sugars when both commercial holocellulase and FAE were used in combination than by the use of holocellulase alone. Thus, FAEs not only release FA but also enabled hemicellulase and cellulase to release more sugars from plant material.



Similar content being viewed by others
References
Adom KK, Liu RH (2002) Antioxidant activity of grains. J Agric Food Chem 50:6182–6187
Bailey MJ, Biely P, Poutanen K (1992) Interlaboratory testing of methods for assay of xylanase activity. J Biotechnol 23:257–270. https://doi.org/10.1016/0168-1656(92)90074-J
Buchanan CJ, Wallace G, Fry SC, Eastwood MA (1996) In Vivo Release of 14C-Labelled phenolic groups from intact dietary spinach cell walls during passage through the rat intestine. J Sci Food Agri 71:459–469. https://doi.org/10.1002/(SICI)1097-0010(199608)71:4<459:AID-JSFA602>3.0.CO;2-H
Bunzel M, Ralph J, Brüning P, Steinhart H (2006) Structural identification of dehydrotriferulic and dehydrotetraferulic acids isolated from insoluble maize bran fiber. J Agric Food Chem 54:6409–6418. https://doi.org/10.1021/jf061196a
Chattopadhyay P, Banerjee G, Sen SK (2018) Cleaner production of vanillin through biotransformation of ferulic acid esters from agroresidue by Streptomyces sannanensis. J Clean Prod 182:272–279. https://doi.org/10.1016/j.jclepro.2018.02.043
Converti A, Aliakbarian B, Domínguez JM et al (2010) Microbial production of biovanillin. Braz J Microbiol 41:519–530. https://doi.org/10.1590/S1517-83822010000300001
Couteau D, McCartney AL, Gibson GR et al (2001) Isolation and characterization of human colonic bacteria able to hydrolyse chlorogenic acid. J Appl Microbiol 90:873–881. https://doi.org/10.1046/j.1365-2672.2001.01316.x
Das A, Raychaudhuri U, Chakraborty R (2012) Cereal based functional food of Indian subcontinent: a review. J Food Sci Technol 49:665–672. https://doi.org/10.1007/s13197-011-0474-1
Ding ZT, Xu DM, Bai J et al (2019) Characterization and identification of ferulic acid esterase-producing Lactobacillus species isolated from Elymus nutans silage and their application in ensiled alfalfa. J Appl Microbiol 127:985–995. https://doi.org/10.1111/jam.14374
Donaghy J, Kelly PF, McKay AM (1998) Detection of ferulic acid esterase production by Bacillus spp. and lactobacilli. Appl Microbiol Biotechnol 50:257–260. https://doi.org/10.1007/s002530051286
El-Shishtawy RM, Mohamed SA, Asiri AM, Gomaa AB, Ibrahim IH, Al-Talhi HA (2014) Solid fermentation of wheat bran for hydrolytic enzymes production and saccharification content by a local isolate Bacillus megatherium. BMC Biotechnol 14:29
Esteban-Torres M, Mancheño JM, de Las Rivas B, Muñoz R (2014) Production and characterization of a tributyrin esterase from Lactobacillus plantarum suitable for cheese lipolysis. J Dairy Sci 97:6737–6744. https://doi.org/10.3168/jds.2014-8234
Faulds CB (2010) What can feruloyl esterases do for us? Phytochem Rev 9:121–132. https://doi.org/10.1007/s11101-009-9156-2
Fazary AE, Ju Y-H (2007) Feruloyl esterases as biotechnological tools: current and future perspectives. Acta Biochim Biophys Sin (Shanghai) 39:811–828. https://doi.org/10.1111/j.1745-7270.2007.00348.x
Ferreira P, Diez N, Faulds CB et al (2007) Release of ferulic acid and feruloylated oligosaccharides from sugar beet pulp by Streptomyces tendae. Bioresour Technol 98:1522–1528. https://doi.org/10.1016/j.biortech.2006.06.004
Ghose TK (1987) Measurement of cellulase activities. Pure Appl Chem 59:257–268. https://doi.org/10.1351/pac198759020257
Guglielmetti S, De Noni I, Caracciolo F et al (2008) Bacterial cinnamoyl esterase activity screening for the production of a novel functional food Product. Appl Environ Microbiol 74:1284–1288. https://doi.org/10.1128/AEM.02093-07
Hunt CJ, Antonopoulou I, Tanksale A et al (2017) Insights into substrate binding of ferulic acid esterases by arabinose and methyl hydroxycinnamate esters and molecular docking. Sci Rep 7:1–11. https://doi.org/10.1038/s41598-017-17260-x
Katileviciute A, Plakys G, Budreviciute A et al (2019) A sight to wheat bran: High value-added products. Biomol 9:887
Kaur B, Chakraborty D (2013) Statistical media and process optimization for biotransformation of rice bran to vanillin using Pediococcus acidilactici. Indian J Exp Biol 51(11):935–943
Kheder F, Delaunay S, Abo-Chameh G et al (2009) Production and biochemical characterization of a type B ferulic acid esterase from Streptomyces ambofaciens. Can J Microbiol 55:729–738. https://doi.org/10.1139/W09-027
Kroon PA, Faulds CB, Ryden P et al (1997) Release of covalently bound ferulic acid from fiber in the human colon. J Agric Food Chem 45:661–667. https://doi.org/10.1021/jf9604403
Kumar K, Yadav AN, Kumar V et al (2017) Food waste: a potential bioresource for extraction of nutraceuticals and bioactive compounds. Bioresour Bioprocess 4:18. https://doi.org/10.1186/s40643-017-0148-6
Kumar N, Pruthi V (2014) Potential applications of ferulic acid from natural sources. Biotechnol Rep (Amst) 4:86–93. https://doi.org/10.1016/j.btre.2014.09.002
Lai KK, Lorca GL, Gonzalez CF (2009) Biochemical properties of two cinnamoyl esterases purified from a Lactobacillus johnsonii strain isolated from stool samples of diabetes-resistant rats. Appl Environ Microbiol 75:5018–5024. https://doi.org/10.1128/AEM.02837-08
Lee FH, Wan SY, Foo HL et al (2019) Comparative study of extracellular proteolytic, cellulolytic, and hemicellulolytic enzyme activities and biotransformation of palm kernel cake biomass by lactic acid bacteria isolated from Malaysian foods. IJMS 20:4979. https://doi.org/10.3390/ijms20204979
Lequart C, Nuzillard J-M, Kurek B, Debeire P (1999) Hydrolysis of wheat bran and straw by an endoxylanase: production and structural characterization of cinnamoyl-oligosaccharides. Carbohyd Res 319:102–111. https://doi.org/10.1016/S0008-6215(99)00110-X
Li F, Ding Z, Ke W et al (2019) Ferulic acid esterase-producing lactic acid bacteria and cellulase pretreatments of corn stalk silage at two different temperatures: Ensiling characteristics, carbohydrates composition and enzymatic saccharification. Biores Technol 282:211–221. https://doi.org/10.1016/j.biortech.2019.03.022
Liu S, Bischoff KM, Anderson AM, Rich JO (2016) Novel feruloyl esterase from Lactobacillus fermentum NRRL B-1932 and analysis of the recombinant enzyme produced in Escherichia coli. Appl Environ Microbiol 82:5068–5076. https://doi.org/10.1128/AEM.01029-16
Long L, Tian D, Zhai R et al (2018) Thermostable xylanase-aided two-stage hydrolysis approach enhances sugar release of pretreated lignocellulosic biomass. Bioresour Technol 257:334–338. https://doi.org/10.1016/j.biortech.2018.02.104
Malunga LN, Beta T (2016) Isolation and identification of feruloylated arabinoxylan mono- and oligosaccharides from undigested and digested maize and wheat. Heliyon 2:e00106. https://doi.org/10.1016/j.heliyon.2016.e00106
Meena P, Tripathi A, Srivastava S, Jha A (2013) Utilization of agro-industrial waste (wheat bran) for alkaline protease production by Pseudomonas aeruginosa in SSF using Taguchi (DOE) methodology. Biocatal Agric Biotechnol 2:210–216
Micard V, Grabber JH, Ralph J, Renard CMGC, Thibault JF (1997) Dehydroferulic acids from sugarbeet pulp. Phytochem 44:1365–1368
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Analytical Chem 31:426–428
Mukherjee G, Singh RK, Mitra A, Sen SK (2007) Ferulic acid esterase production by Streptomyces sp. Biores Technol 98:211–213. https://doi.org/10.1016/j.biortech.2005.12.001
Nagar S, Mittal A, Kumar D, Kumar L, Kuhad RC, Gupta VK (2011) Hyper production of alkali stable xylanase in lesser duration by Bacillus pumilus SV-85S using wheat bran under solid state fermentation. New Biotechnol 28:581–587
Napolitano A, Costabile A, Martin-Pelaez S et al (2009) Potential prebiotic activity of oligosaccharides obtained by enzymatic conversion of durum wheat insoluble dietary fibre into soluble dietary fibre. Nutr Metab Cardiovas Dis 19:283–290. https://doi.org/10.1016/j.numecd.2008.07.005
Ou S, Zhang J, Wang Y, Zhang N (2011) Production of feruloyl esterase from Aspergillus niger by solid-state fermentation on different carbon sources. In: Enzyme Research. https://www.hindawi.com/journals/er/2011/848939/. Accessed 10 Apr 2020
Perez-Rodriguez N, Moreira CD, Torrado Agrasar A, Dominguez JM (2016) Feruloyl esterase production by Aspergillus terreus CECT 2808 and subsequent application to enzymatic hydrolysis. Enz Microb Technol 91:52–58
Ruiz Rodríguez LG, Mohamed F, Bleckwedel J et al (2019) Diversity and functional properties of lactic acid bacteria isolated from wild fruits and flowers present in Northern Argentina. Front Microbiol. https://doi.org/10.3389/fmicb.2019.01091
Sakamoto T, Nishimura S, Kato T et al (2005) Efficient extraction of ferulic acid from sugar beet pulp using the culture supernatant of Penicillium chrysogenum. J Appl Glycosci 52:115–120. https://doi.org/10.5458/jag.52.115
Salmon DN, Piva LC, Binati RL, Rodrigues C, Vandenberghe LP, Soccol CR, Spier MR (2012) A bioprocess for the production of phytase from Schizophyllum commune: Studies of its optimization, profile of fermentation parameters, characterization and stability. Bioprocess Biosyst 35:1067–1079
Saulnier L, Marot C, Elgorriaga M et al (2001) Thermal and enzymatic treatments for the release of free ferulic acid from maize bran. Carbohyd Polym 45:269–275. https://doi.org/10.1016/S0144-8617(00)00259-9
Smith MM, Hartley RD (1983) Occurrence and nature of ferulic acid substitution of cell-wall polysaccharides in graminaceous plants. Carbohyd Res 118:65–80. https://doi.org/10.1016/0008-6215(83)88036-7
Su R, Ni K, Wang T et al (2019) Effects of ferulic acid esterase-producing Lactobacillus fermentum and cellulase additives on the fermentation quality and microbial community of alfalfa silage. PeerJ. https://doi.org/10.7717/peerj.7712
Szwajgier D, Jakubczyk A (2011) Production of extracellular ferulic cid esterases by Lactobacillus strains using natural and synthetic carbon sources. Acta Sci Pol Technol Aliment 10(3):287–302
Tilay A, Bule M, Kishenkumar J, Annapure U (2008) Preparation of ferulic acid from agricultural wastes: Its improved extraction and purification. J Agric Food Chem 56:7644–7648. https://doi.org/10.1021/jf801536t
Wang X, Geng X, Egashira Y, Sanada H (2004) Purification and characterization of a feruloyl esterase from the intestinal bacterium Lactobacillus acidophilus. Appl Environ Microbiol 70:2367–2372. https://doi.org/10.1128/AEM.70.4.2367-2372.2004
Xiros C, Moukouli M, Topakas E, Christakopoulos P (2009) Factors affecting ferulic acid release from Brewer’s spent grain by Fusarium oxysporum enzymatic system. Biores Technol 100:5917–5921. https://doi.org/10.1016/j.biortech.2009.06.018
Xu Z, He H, Zhang S et al (2017) Characterization of feruloyl esterases produced by the four Lactobacillus Species: L. amylovorus, L. acidophilus, L. farciminis and L. fermentum, isolated from ensiled corn stover. Front Microbiol. https://doi.org/10.3389/fmicb.2017.00941
Zhang Q, Li X, Zhao M, Yu Z (2016) Lactic acid bacteria strains for enhancing the fermentation quality and aerobic stability of Leymus chinensis silage. Grass Forage Sci 71:472–481. https://doi.org/10.1111/gfs.12190
Acknowledgements
Abha Sharma acknowledges fellowship received from Department of Science and Technology (File No. LS/700/2016) under Wos-A scheme. All the authors thank ICAR-IARI, Pusa, New Delhi for providing essential facilities for the research work. The authors acknowledge Dr. Radha Prasanna, Professor, IARI, New Delhi for English editing of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sharma, A., Sharma, A., Singh, J. et al. A biorefinery approach for the production of ferulic acid from agroresidues through ferulic acid esterase of lactic acid bacteria. 3 Biotech 10, 367 (2020). https://doi.org/10.1007/s13205-020-02360-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13205-020-02360-9