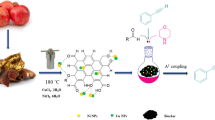
Abstract
On the anvil of the world of catalysis, the role of nanoparticles (NPs) as well as graphene oxide (GO) are well acknowledged as a new marvel experienced in various fields specially catalyzing the organic reactions. Herein, we report the use of biogenically synthesized Cu2O NPs decorated on reduced graphene oxide (Cu2O/RGO) nanocomposites (NCs) as a selective hydrogenation catalyst to efficiently transform E-cinnamaldehyde to E-cinnamyl alcohol. The transformation of cinnamaldehyde with 10 mg of Cu2O/RGO nanocatalyst and NaBH4 was achieved in 5 min in water at room temperature in excellent yield (~ 95%). The detailed mechanism of the formation of cinnamyl alcohol was also discussed. Cu2O/RGO NCs were synthesized utilizing an agricultural waste i.e., beetroot peel extract (BRPE) where NCs were stabilized by BRPE on the surface of reduced GO, and simultaneously the conjugation between the GO and copper ions is also responsible for the stabilization of Cu2O NPs. Incorporating the agricultural waste for the synthesis of Cu2O/RGO NCs validates an economic, efficient and ecofriendly approach. Novel nanoscale materials Cu2O/RGO are capable of offering several opportunities as heterogeneous catalysts thereby arousing wider interest and enthusiasm of scientists for exploring more reactions for their efficient catalytic performances in future.
Graphical abstract











Similar content being viewed by others
References
Allen JM, Vincent TC, Richard KB (2010) Honeycomb carbon : a review of graphene what is graphene? Chem Rev 110:132–145
Butt M, Feng X, Yamamoto Y, Almansour AI, Arumugam N, Kumar RS, Bao M (2017) Unsupported nanoporous gold-catalyzed chemoselective reduction of α, β-unsaturated aldehydes using formic acid as hydrogen source. Asian J Org Chem 6(7):867–872. https://doi.org/10.1002/ajoc.201700130
Cahyana AH, Liandi AR, Maghdalena M, Yunarti RT, Wendari TP (2022) Magnetically separable Fe3O4/graphene oxide nanocomposite: an efficient heterogenous catalyst for spirooxindole derivatives synthesis. Ceram Int 48(13):18316–18323. https://doi.org/10.1016/j.ceramint.2022.03.090
Coman SN, Parvulescu VI, De Bruyn M, De Vos DE, Jacobs PA (2002) Reduction of prostaglandin unsaturated ketones to secondary allylic alcohols by hydrogen transfer over mesoporous-supported PtSn catalysts. J Catal 206(2):218–229. https://doi.org/10.1006/jcat.2001.3432
Cova CM, Zuliani A, Muñoz-Batista MJ, Luque R (2019) Efficient Ru-based scrap waste automotive converter catalysts for the continuous-flow selective hydrogenation of cinnamaldehyde. Green Chem 21(17):4712–4722. https://doi.org/10.1039/c9gc01596e
Dai Y, Gao X, Chu X, Jiang C, Yao Y, Guo Z et al (2018) On the role of water in selective hydrogenation of cinnamaldehyde to cinnamyl alcohol on PtFe catalysts. J Catal 364:192–203. https://doi.org/10.1016/j.jcat.2018.05.008
Fang X, Zhang M, Jackstell R, Beller M (2013) Selective palladium-catalyzed hydroformylation of alkynes to α, β-unsaturated aldehydes. Angew Chem Int Ed Engl 52(17):4645–4649. https://doi.org/10.1002/anie.201300759
Fortunato JM, Ganem B (1976) Lithium and potassium trialkylborohydrides. Reagents for direct reduction of α, β-unsaturated carbonyl compounds to synthetically versatile enolate anions1. J Org Chem 41(12):2194–2200. https://doi.org/10.1021/jo00874a028
Fuller JC, Stangeland EL, Goralski CT, Singaram B (1993) Aminoborohydrides. 2. Regiospecific reductions of α, β-unsaturated carbonyl compounds with lithium pyrrolidinoborohydride. A facile conversion of α, β-unsaturated aldehydes and ketones to the corresponding allylic alcohols in high purity. Tetrahedron Lett 34(2):257–260. https://doi.org/10.1016/S0040-4039(00)60561-0
Greene EFD, Herz W, Benkeser RA, Brauman JI, Evans DA, Gassman PG et al (1977) J Org Chem 18042(215):20036
Hao CH, Guo XN, Pan YT, Chen S, Jiao ZF, Yang H, Guo XY (2016) Visible-light-driven selective photocatalytic hydrogenation of cinnamaldehyde over Au/SiC catalysts. J Am Chem Soc 138(30):9361–9364. https://doi.org/10.1021/jacs.6b04175
Hapsari M, Cahyana AH, Oktavia SH, Liandi AR (2020) Synthesis of spirooxindole-pyrrolizidine compounds using Fe3O4-GO catalyst and their bioactivity assays. Rasayan J Chem 13(4):2317–2324. https://doi.org/10.31788/RJC.2020.1345583
Kelsen V, Wendt B, Werkmeister S, Junge K, Beller M, Chaudret B (2013) The use of ultrasmall iron(0) nanoparticles as catalysts for the selective hydrogenation of unsaturated C-C bonds. Chem Commun 49(33):3416–3418. https://doi.org/10.1039/c3cc00152k
Khan M, Tahir MN, Adil SF, Khan HU, Siddiqui MRH, Al-Warthan AA, Tremel W (2015) Graphene based metal and metal oxide nanocomposites: synthesis, properties and their applications. J Mater Chem A 3(37):18753–18808. https://doi.org/10.1039/c5ta02240a
Kim S, Moon YC, Ahn KH (1982) Lithium n-butylborohydride as a selective reducing agent for the reduction of enones, cyclic ketones, and selected carbonyl compounds. J Org Chem. https://doi.org/10.1021/jo00138a021
Kurmarayuni CM, Kurapati S, Akhil S, Chandu B, Khandapu BMK, Koya PR, Bollikolla HB (2020) Synthesis of multifunctional graphene exhibiting excellent sonochemical dye removal activity, green and regioselective reduction of cinnamaldehyde. Mater Lett 263:127224. https://doi.org/10.1016/j.matlet.2019.127224
Llabre FX, Corma A, Garcia H, Valencia D, Vera C De (2007). <2007_JPC_Applications for Metal Organic Frameworks as Quantum Dot Semiconductors.pdf>, 80–85.
Lu W, Sun W, Tan X, Wang B, Yang H, Gao L, Zheng G (2019) Stabilized Cu2O nanoparticles on rGO highly catalyzed direct oxidative coupling synthesis of Α-ketoamides with molecular oxygen. Catal Commun 132:105806. https://doi.org/10.1016/j.catcom.2019.105806
Marcano DC, Kosynkin DV, Berlin JM, Sinitskii A, Sun Z, Slesarev A et al (2010) Improved synthesis of graphene oxide. ACS Nano 4(8):4806–4814. https://doi.org/10.1021/nn1006368
Mertens PGN, Vandezande P, Ye X, Poelman H, Vankelecom IFJ, De Vos DE (2009) Recyclable Au0, Ag0 and Au0-Ag0 nanocolloids for the chemoselective hydrogenation of α, β-unsaturated aldehydes and ketones to allylic alcohols. Appl Catal A Gen 355(1–2):176–183. https://doi.org/10.1016/j.apcata.2008.12.013
Miao X, Bidange J, Dixneuf PH, Fischmeister C, Bruneau C, Dubois JL, Couturier JL (2012) Ruthenium-benzylidenes and ruthenium-indenylidenes as efficient catalysts for the hydrogenation of aliphatic nitriles into primary amines. ChemCatChem 4(12):1911–1916. https://doi.org/10.1002/cctc.201200511
Mohamadi M, Setamdideh D, Khezri B (2013) Regioselective and chemoselective reduction of α, β-unsaturated carbonyl compounds by NaBH 4/Ba (OAc) 2 as a reducing system. Org Chem Int 2013:1–5. https://doi.org/10.1155/2013/127585
Mohr C, Hofmeister H, Radnik J, Claus P (2003) Identification of active sites in gold-catalyzed hydrogenation of acrolein. J Am Chem Soc 125(7):1905–1911. https://doi.org/10.1021/ja027321q
Nongwe I, Ravat V, Meijboom R, Coville NJ (2016) Pt supported nitrogen doped hollow carbon spheres for the catalysed reduction of cinnamaldehyde. Appl Catal A Gen 517:30–38. https://doi.org/10.1016/j.apcata.2016.02.025
Ralphs K, McCourt É, Ormandy C, Carneiro de Souza TA, Nockemann P, Jacquemin J, Manyar HG (2018) Highly selective reduction of α, β-unsaturated aldehydes and ketones under ambient conditions using tetraalkylphosphonium-based ionic liquids. ChemistrySelect 3(42):11706–11711. https://doi.org/10.1002/slct.201801092
Rao RG, Blume R, Hansen TW, Fuentes E, Dreyer K, Moldovan S et al (2017) Interfacial charge distributions in carbon-supported palladium catalysts. Nat Commun. https://doi.org/10.1038/s41467-017-00421-x
Rojas H, Díaz G, Martínez JJ, Castañeda C, Gómez-Cortés A, Arenas-Alatorre J (2012) Hydrogenation of α, β-unsaturated carbonyl compounds over Au and Ir supported on SiO 2. J Mol Catal A Chem 363–364:122–128. https://doi.org/10.1016/j.molcata.2012.05.025
Shi J, Nie R, Chen P, Hou Z (2013) Selective hydrogenation of cinnamaldehyde over reduced graphene oxide supported Pt catalyst. Catal Commun 41:101–105. https://doi.org/10.1016/j.catcom.2013.07.012
Simion AM, Arimura T, Simion C (2013) Reaction of cinnamaldehyde and derivatives with Raney Ni-Al alloy and Al powder in water. Reduction or oxido-reduction? C R Chim 16(5):476–481. https://doi.org/10.1016/j.crci.2012.11.022
Sun M, Liu H, Liu Y, Qu J, Li J (2015) Graphene-based transition metal oxide nanocomposites for the oxygen reduction reaction. Nanoscale 7(4):1250–1269. https://doi.org/10.1039/c4nr05838k
White RJ, Luque R, Budarin VL, Clark JH, Macquarrie DJ (2009) Supported metal nanoparticles on porous materials. Methods and applications. Chem Soc Rev 38(2):481–494. https://doi.org/10.1039/b802654h
Wruss J, Waldenberger G, Huemer S, Uygun P, Lanzerstorfer P, Müller U et al (2015) Compositional characteristics of commercial beetroot products and beetroot juice prepared from seven beetroot varieties grown in Upper Austria. J Food Composition Anal 42(3):46–55. https://doi.org/10.1016/j.jfca.2015.03.005
Wu B, Huang H, Yang J, Zheng N, Fu G (2012) Selective hydrogenation of α, β-unsaturated aldehydes catalyzed by amine-capped platinum-cobalt nanocrystals. Angew Chem Int Ed Engl 51(14):3440–3443. https://doi.org/10.1002/anie.201108593
Xiao Z (2017) Insight into the Meerwein-Ponndorf-Verley reduction of cinnamaldehyde over MgAl oxides catalysts. Mol Catal 436:1–9. https://doi.org/10.1016/j.mcat.2017.04.016
Yadav S, Jain A, Malhotra P (2019) A review on the sustainable routes for the synthesis and applications of cuprous oxide nanoparticles and their nanocomposites. Green Chem 21(5):937–955. https://doi.org/10.1039/c8gc03303j
Yadav S, Chauhan M, Mathur D, Jain A, Malhotra P (2021) Sugarcane bagasse-facilitated benign synthesis of Cu2O nanoparticles and its role in photocatalytic degradation of toxic dyes: a trash to treasure approach. Environ Dev Sustain 23(2):2071–2091. https://doi.org/10.1007/s10668-020-00664-7
Yuan K, Song T, Wang D, Zhang X, Gao X, Zou Y et al (2018) Effective and selective catalysts for cinnamaldehyde hydrogenation: hydrophobic hybrids of metal-organic frameworks, metal nanoparticles, and micro- and mesoporous polymers. Angew Chem 130(20):5810–5815. https://doi.org/10.1002/ange.201801289
Zhao M, Yuan K, Wang Y, Li G, Guo J, Gu L et al (2016) Metal-organic frameworks as selectivity regulators for hydrogenation reactions. Nature 539(7627):76–80. https://doi.org/10.1038/nature19763
Zhu Y, Qian H, Drake BA, Jin R (2010) Atomically precise Au 25 (SR) 18 nanoparticles as catalysts for the selective hydrogenation of α, β-unsaturated ketones and aldehydes. Angew Chem 122(7):1317–1320. https://doi.org/10.1002/ange.200906249
Acknowledgements
M Chauhan and S Yadav thankful to University Grant Commission India for providing financial help.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On the behalf of all the authors, there is no conflict of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chauhan, M., Yadav, S. & Malhotra, P. In-situ biogenically synthesized Cu2O/RGO composite using beetroot peel extract for selective and efficient reduction of cinnamaldehyde in water. Appl Nanosci 13, 4331–4342 (2023). https://doi.org/10.1007/s13204-022-02699-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13204-022-02699-w