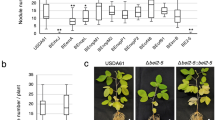
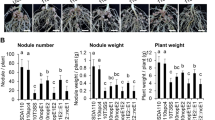
Abstract
Biological nitrogen fixation through a symbiosis between the legume and free-living Bradyrhizobium is a crucial step to fulfill the nitrogenous demand of the host plant. Bradyrhizobium establishes successful symbiosis with the legumes by virtue of having a set of genes that code for nod-factors (NFs). The NFs are very much species-specific and thus, the symbionts have their own choice of host. Recently it has been reported that even in absence of NF Bradyrhizobia can establish successful nodulation in their cognate hosts. But nod-independent (NI) nodulation is only limited in some species of Bradyrhizobium. This review emphasized on types of Bradyrhizobium species, types of nodule formation, regulation of nodulation, comparative mechanism of both nod-dependent (ND) and independent nitrogen fixation in legume plants, relationship with type 3 secretion systems (T3SS) and NI nitrogen fixation by Bradyrhizobium, the role of NF receptor homolog and NF in symbiosis. Thus, this review predicts the shift of one group of symbionts to another unique type, where pathogen-associated machinery like T3SS plays a role in symbiosis.





Similar content being viewed by others
References
Arrighi JF, Cartieaux F, Brown SC, Rodier-Goud M, Boursot M, Fardoux J, Patrel D, Gully D, Fabre S, Chaintreuil C, Giraud E (2012) Aeschynomene evenia, a model plant for studying the molecular genetics of the nod-independent rhizobium-legume symbiosis. Mol Plant Microbe Interact 25:851–861. https://doi.org/10.1094/MPMI-02-12-0045-TA
Avontuur JR, Palmer M, Beukes CW, Chan WY, MPA C, Blom J, Stępkowski T, Kyrpides NC, Woyke T, Shapiro N, Whitman WB, Venter SN, Steenkamp ET (2019) Genome-informed Bradyrhizobium taxonomy: where to from here? Syst Appl Microbiol 2:427–439. https://doi.org/10.1016/j.syapm.2019.03.006
Bolzan de Campos S, Deakin WJ, Broughton WJ, Passaglia LMP (2011) Roles of flavonoids and the transcriptional regulator TtsI in the activation of the type III secretion system of Bradyrhizobium elkanii SEMIA587. Microbiol Read Engl 157:627–635. https://doi.org/10.1099/mic.0.040873-0
Bonaldi K, Gargani D, Prin Y, Fardoux J, Gully D, Nouwen N, Goormachtig S, Giraud E (2011) Nodulation of Aeschynomene afraspera and A. indica by photosynthetic Bradyrhizobium sp. strain ORS285: the nod-dependent versus the nod-independent symbiotic interaction. Mol Plant-Microbe Interact 24:1359–1371. https://doi.org/10.1094/MPMI-04-11-0093
Broghammer A, Krusell L, Blaise M, Sauer J, Sullivan JT, Maolanon N, Vinther M, Lorentzen A, Madsen EB, Jensen KJ, Roepstorff P, Thirup S, Ronson CW, Thygesen MB, Stougaard J (2012) Legume receptors perceive the rhizobial lipochitin oligosaccharide signal molecules by direct binding. PNAS 109:13859–13864
Brottier L, Chaintreuil C, Simion P, Scornavacca C, Rivallan R, Mournet P, Moulin L, Lewis GP, Fardoux J, Brown SC, Gomez-Pacheco M, Bourges M, Hervouet C, Gueye M, Duponnois R, Ramanankierana H, Randriambanona H, Vandrot H, Zabaleta M et al (2018) A phylogenetic framework of the legume genus Aeschynomene for comparative genetic analysis of the Nod-dependent and Nod-independent symbioses. BMC Plant Biol 18:333. https://doi.org/10.1186/s12870-018-1567-z
Chaintreuil C, Rivallan R, Bertioli DJ, Klopp C, Gouzy J, Courtois B, Leleux P, Martin G, Rami JF, Gully D, Parrinello H, Séverac D, Patrel D, Fardoux J, Ribière W, Boursot M, Cartieaux F, Czernic P, Ratet P et al (2016) A gene-based map of the nod-factor-independent Aeschynomene evenia sheds new light on the evolution of nodulation and legume genomes. DNA Res 23:365–376. https://doi.org/10.1093/dnares/dsw020
Compant S, Samad A, Faist H, Sessitsch A (2019) A review on the plant microbiome: ecology, functions, and emerging trends in microbial application. J Adv Res 19:29–37. https://doi.org/10.1016/j.jare.2019.03.004
Fabre S, Gully D, Poitout A, Patrel D, Arrighi JF, Giraud E, Czernic P, Cartieaux F (2015) Nod-factor-independent nodulation in Aeschynomene evenia required the common plant-microbe symbiotic ‘toolkit’. Plant Physiol 169:2654–2664. https://doi.org/10.1104/pp.15.01134
Ferguson BJ, Indrasumunar A, Hayashi S, Lin MH, Lin YH, Reid DE, Gresshoff PM (2010) Molecular analysis of legume nodule development and autoregulation. J Int Plant Biol 52:61–76. https://doi.org/10.1111/j.1744-7909.2010.00899.x
Ferro M, Lorquin J, Ba S, Sanon K, Promé JC, Boivin C (2000) Bradyrhizobium sp. strains that nodulate the leguminous tree Acacia albida produce fucosylated and partially sulfated Nod-factors. Appl Environ Microbiol 66:5078–5082. https://doi.org/10.1128/AEM.66.11.5078-5082.2000
Fiume E, Fletcher JC (2012) Regulation of Arabidopsis embryo and endosperm development by the polypeptide signaling molecule CLE8. Plant Cell 24:1000–1012. https://doi.org/10.1105/tpc.111.094839
Gage DJ (2004) Infection and invasion of roots by symbiotic, nitrogen-fixing rhizobia during nodulation of temperate legumes. Microbiol Mol Biol Rev 68:280–300. https://doi.org/10.1128/MMBR.68.2.280-300.2004
Gazi AD, Sarris PF, Fadouloglou VE, Charova SN, Mathioudakis N, Panopoulos NJ, Kokkinidis M (2012) Phylogenetic analysis of a gene cluster encoding an additional, rhizobial-like type III secretion system that is narrowly distributed among Pseudomonas syringae strains. BMC Microbiol 12:188. https://doi.org/10.1186/1471-2180-12-188
Giraud E, Moulin L, Vallenet D, Barbe V, Cytryn E, Avarre JC, Jaubert M, Simon D, Cartieaux F, Prin Y, Bena G, Hannibal L, Fardoux J, Kojadinovic M, Vuillet L, Lajus A, Cruveiller S, Rouy Z, Mangenot S et al (2007) Legumes symbioses: absence of Nod genes in photosynthetic bradyrhizobia. Science 316:1307–1312. https://doi.org/10.1126/science.1139548
Giraud E, Xu L, Chaintreuil C, Gargani D, Gully D, Sadowsky MJ (2013) Photosynthetic Bradyrhizobium sp. strain ORS285 is capable of forming nitrogen-fixing root nodules on soybeans (Glycine max). Appl Environ Microbiol 79(7):2459–2462. https://doi.org/10.1128/AEM.03735-12
Gourion B, Berrabah F, Ratet P, Stacey G (2014) Rhizobium-legume symbioses: the crucial role of plant immunity. Trends Plant Sci 20:186–194. https://doi.org/10.1016/j.tplants.2014.11.008
Guha S, Sarkar M, Ganguly P, Uddin MR, Mandal S, DasGupta M (2016) Segregation of nod-containing and nod-deficient bradyrhizobia as endosymbionts of Arachis hypogaea and as endophytes of Oryza sativa in intercropped fields of Bengal Basin, India. Environ Microbiol 18:2575–2590. https://doi.org/10.1111/1462-2920.13348
Gully D, Czernic P, Cruveiller S, Mahé F, Longin C, Vallenet D, François P, Nidelet S, Rialle S, Giraud E, Arrighi JF, DasGupta M, Cartieaux F (2018) Transcriptome profiles of nod factor-independent symbiosis in the tropical legume aeschynomene evenia. Sci Rep 8:10934. https://doi.org/10.1038/s41598-018-29301-0
Hanning I, Diaz-Sanchez S (2015) The functionality of the gastrointestinal microbiome in non-human animals. Microbiome 3:51. https://doi.org/10.1186/s40168-015-0113-6
Hayashi S, Reid DE, Lorenc MT, Stiller J, Edwards D, Gresshoff PM, Ferguson BJ (2012) Transient Nod-factor-dependent gene expression in the nodulation-competent zone of soybean (Glycine max [L.] Merr.) roots. Plant Biotechnol J 10:995–1010. https://doi.org/10.1111/j.1467-7652.2012.00729.x
Hedin LO, Brookshire EJ, Menge DN, Barron AR (2009) The nitrogen paradox in tropical forest ecosystems. Annu Rev Ecol Evol Syst 40:613–635, doi.org/https://doi.org/10.1146/annurev.ecolsys.37.091305.110246
Held M, Hou H, Miri M, Huynh C, Ross L, Hossain MS, Sato S, Tabata S, Perry J, Wang TL, Szczyglowski K (2014) Lotus japonicas cytokinin receptors work partially redundantly to mediate nodule formation. Plant Cell 26(2):678–694. https://doi.org/10.1105/tpc.113.119362
Hungria M, Menna P, Delamuta JRM (2015) Bradyrhizobium, the ancestor of all rhizobia: phylogeny of housekeeping and nitrogen-fixation genes. In: de Bruijn F (ed) Biological nitrogen fixation. Wiley, Hoboken, pp 191–202
Jiménez-Guerrero I, Pérez-Montaño F, Monreal JA, Preston GM, Fones H, Vioque B, Ollero FJ, López-Baena FJ (2015) The Sinorhizobium (Ensifer) fredii HH103 Type 3 secretion system suppresses early defense responses to effectively nodulate soybean. Mol Plant-Microbe Interact 28:790–799. https://doi.org/10.1094/MPMI-01-15-0020-R
Jordan DC (1982) Notes: transfer of Rhizobium japonicum Buchanan 1980 to Bradyrhizobium gen. nov., a genus of slow-growing, root nodule bacteria from leguminous plants. Int J Syst Bacteriol 32:136–139
Kouchi H, Imaizumi-Anraku H, Hayashi M, Hakoyama T, Nakagawa T, Umehara Y, Suganuma N, Kawaguchi M (2010) How many peas in a pod? Legume genes responsible for mutualistic symbioses underground. Plant Cell Physiol 51:1381–1397. https://doi.org/10.1093/pcp/pcq107
Lavin M, Herendeen PS, Wojciechowski MF (2005) Evolutionary rates analysis of leguminosae implicates a rapid diversification of lineages during the tertiary. Syst Biol 54:575–594. https://doi.org/10.1080/10635150590947131
Lin JS, Li X, Luo ZL, Mysore KS, Wen J (2018) NIN interacts with NLPs to mediate nitrate inhibition of nodulation in Medicago truncatula. Nat Plants 4:942–952. https://doi.org/10.1038/s41477-018-0261-3
Liu YH, Wang ET, Jiao YS, Tian CF, Wang L (2018) Symbiotic characteristics of Bradyrhizobium diazoefficiens USDA 110 mutants associated with shrubby sophora (Sophora flavescens) and soybean (Glycine max). Microbial Res 214:19–27. https://doi.org/10.1016/j.micres.2018.05.012
Lohar D, Stiller J, Kam J, Stacey G, Gresshoff PM (2009) Ethylene insensitivity conferred by a mutated Arabidopsis ethylene receptor gene alters nodulation in transgenic Lotus japonicus. Ann Bot 104:277–285. https://doi.org/10.1093/aob/mcp132
Madsen LH, Tirichine L, Jurkiewicz A, Sullivan JT, Heckmann AB, Bek AS, Ronson CW, James EK, Stougaard J (2010) The molecular network governing nodule organogenesis and infection in the model legume Lotus japonicus. Nat Commun 1:10. https://doi.org/10.1038/ncomms1009
Marsh JF, Rakocevic A, Mitra RM, Brocard L, Sun J (2007) Medicago truncatula NIN is essential for rhizobial-independent nodule organogenesis induced by autoactive calcium/calmodulin-dependent protein kinase. Plant Physiol 144:324–335. https://doi.org/10.1104/pp.106.093021
Miché L, Moulin L, Chaintreuil C, Contreras-Jimenez JL, Munive-Hernández JA, Del Carmen Villegas-Hernandez M, Crozier F, Béna G (2010) Diversity analyses of Aeschynomene symbionts in Tropical Africa and Central America reveal that nod independent stem nodulation is not restricted to photosynthetic bradyrhizobia. Environ Microbiol 12:2152–2164. https://doi.org/10.1111/j.1462-2920.2009.02090.x
Miyata K, Hayafune M, Kobae Y, Kaku H, Nishizawa Y, Masuda Y, Shibuya N, Nakagawa T (2016) Evaluation of the role of the LysM receptor-like kinase, OsNFR5/OsRLK2 for AM symbiosis in rice. Plant Cell Physiol 57:2283–2290. https://doi.org/10.1093/pcp/pcw144
Mornico D, Miché L, Béna G, Nouwen N, Verméglio A, Vallenet D, Smith AA, Giraud E, Médigue C, Moulin L (2011) Comparative genomics of Aeschynomene symbionts: insights into the ecological lifestyle of Nod-independent photosynthetic bradyrhizobia. Genes 3:35–61. https://doi.org/10.3390/genes3010035
Mortier V, Den-Herder G, Whitford R, Van de Velde W, Rombauts S, D’Haeseleer K, Holsters M, Goormachtig S (2010) CLE peptides control Medicago truncatula nodulation locally and systemically. Plant Physiol 153:222–237. https://doi.org/10.1104/pp.110.153718
Muñoz V, Ibáñez F, Tordable M, Megías M, Fabra A (2015) Role of reactive oxygen species generation and Nod-factors during the early symbiotic interaction between bradyrhizobia and peanut, a legume infected by crack entry. J Appl Microbiol 118:182–192. https://doi.org/10.1111/jam.12669
Nelson MS, Sadowsky MJ (2015) Secretion systems and signal exchange between nitrogen-fixing rhizobia and legumes. Front Plant Sci 6:491. https://doi.org/10.3389/fpls.2015.00491
Ney L, Franklin D, Mahmud K, Cabrera M, Hancock D, Habteselassie M, Newcomer Q, Fatzinger B (2019) Rebuilding soil ecosystems for improved productivity in biosolarized soils. Int J Agron 2019:5827585. https://doi.org/10.1155/2019/5827585
Nguyen HP, STN R, Yasuda M, Teaumroong N, Okazaki S (2020) Identification of Bradyrhizobium elkanii USDA61 type III efectors determining symbiosis with Vigna mungo. Genes 11:474. https://doi.org/10.3390/genes11050474
Nishida H, Tanaka S, Handa Y, Ito M, Sakamoto Y, Matsunaga S, Betsuyaku S, Miura K, Soyano T, Kawaguchi M, Suzaki T (2018) A NIN-LIKE PROTEIN mediates nitrate-induced control of root nodule symbiosis in Lotus japonicus. Nat Commun 9:499. https://doi.org/10.1038/s41467-018-02831-x
Noisangiam R, Teamtisong K, Tittabutr P, Boonkerd N, Toshiki U, Minamisawa K, Teaumroong N (2012) Genetic diversity, symbiotic evolution, and proposed infection process of Bradyrhizobium strains isolated from root nodules of Aeschynomene Americana L. in Thailand. Appl Environ Microbiol 78:6236–6250. https://doi.org/10.1128/AEM.00897-12
Nouwen N, Fardoux J, Giraud E (2016) NodD1 and NodD2 are not required for the symbiotic interaction of Bradyrhizobium ORS285 with Nod-factor-independent Aeschynomene legumes. PLoS One 11:805–811. https://doi.org/10.1371/journal.pone.0157888
Okamoto S, Ohnishi E, Sato S, Takahashi H, Nakazono M, Tabata S, Kawaguchi M (2009) Nod-factor/nitrate-induced CLE genes that drive HAR1-mediated systemic regulation of nodulation. Plant Cell Physiol 50:67–77. https://doi.org/10.1093/pcp/pcn194
Okazaki S, Kaneko T, Sato S, Saeki K (2013) Hijacking of leguminous nodulation signaling by the rhizobial type III secretion system. PNAS USA 110:17131–17136. https://doi.org/10.1073/pnas.1302360110
Okazaki S, Tittabutr P, Teulet A, Thouin J, Fardoux J, Chaintreuil C, Gully D, Arrighi JF, Furuta N, Miwa H, Yasuda M, Nouwen N, Teaumroong N, Giraud E (2016) Rhizobium-legume symbiosis in the absence of Nod-factors: two possible scenarios with or without the T3SS. ISME J 1:64–74. https://doi.org/10.1038/ismej.2015.103
Okubo T, Fukushima S, Minamisawa K (2012) Evolution of bradyrhizobium–aeschynomene mutualism: living testimony of the ancient world or highly evolved state? Plant Cell Physiol 53:2000–2007. https://doi.org/10.1093/pcp/pcs150
Oldroyd GED, Murray JD, Poole PS, Downie JA (2011) The rules of engagement in the legume-rhizobial symbiosis. Annu Rev Genet 45:119–144
Passaglia LMP (2017) Bradyrhizobium elkanii nod regulon: insights through genomic analysis. Gen Mol Biol 40:703–716. https://doi.org/10.1590/1678-4685-GMB-2016-0228
Piromyou P, Songwattana P, Greetatorn T, Okubo T, Kakizaki KC, Prakamhang J, Tittabutr P, Boonkerd N, Teaumroong N, Minamisawa K (2015) The type III secretion system (T3SS) is a determinant for rice-endophyte colonization by non-photosynthetic Bradyrhizobium. Microbe Environ 30:291–300. https://doi.org/10.1264/jsme2.ME15080
Podlešáková K, Fardoux J, Patrel D, Bonaldi K, Novák O, Strnad M, Giraud E, Spíchal L, Nouwen N (2013) Rhizobial synthesized cytokinins contribute to but are not essential for the symbiotic interaction between photosynthetic Bradyrhizobia and Aeschynomene legumes. Mol Plant Microbe Interact 26:1232–1238. https://doi.org/10.1094/MPMI-03-13-0076-R
Prithiviraj B, Souleimanov A, Zhou X, Smith DL (2000) Differential response of soybean (Glycine max (L.) Merr.) genotypes to lipo-chito-oligosaccharide Nod Bj-V (C18:1, MeFuc). J Exp Bot 51:2045–2051. https://doi.org/10.1093/jexbot/51.353.2045
Quilbé J, Lamy L, Brottier L, Leleux P, Fardoux J, Rivallan R, Benichou T, Guyonnet R, Becana M, Villar I, Garsmeur O, Hufnagel B, Delteil A, Gully D, Chaintreuil C, Pervent M, Cartieaux F, Bourge M, Valentin N et al (2021) Genetics of nodulation in Aeschynomene evenia uncovers mechanisms of the rhizobium-legume symbiosis. Nat Commun 12:829. https://doi.org/10.1038/s41467-021-21094-7
Renier A, Maillet F, Fardoux J, Poinsot V, Giraud E, Nouwen N (2011) Photosynthetic Bradyrhizobium sp. strain ORS285 synthesizes 2-O-methyl-fucosylated lipochitooligo-saccharides for nod gene-dependent interaction with Aeschynomene plants. Mol Plant Microbe Interact 24:1440–1447. https://doi.org/10.1094/MPMI-05-11-0104
Santi C, Bogusz D, Franche C (2013) Biological nitrogen fixation in non-legume plants. Ann Bot 111:743–767. https://doi.org/10.1093/aob/mct048
Sergeevich SA, Alexandrovich ZV, Yurievna SO, Yurievich BA, Anatolievich TI (2015) Nod-factor signaling in legume-rhizobial symbiosis. In: El-Shemy H, editor. Plants for the future. InTECH, pp. 135–60
Sharma V, Bhattacharyya S, Kumar R, Kumar A, Ibañez F (2020) Molecular basis of root nodule symbiosis between Bradyrhizobium and ‘Crack-Entry’ legume groundnut (Arachis hypogaea L.). Plants (Basel, Switzerland) 9:276. https://doi.org/10.3390/plants9020276
Songwattana P, Noisangiam R, Teamtisong K, Prakamhang J, Teulet A (2017) Type 3 secretion system (T3SS) of Bradyrhizobium sp. DOA9 and its roles in legume symbiosis and rice endophytic association. Front Microbiol 8:1810. https://doi.org/10.3389/fmicb.2017.01810
Soyano T, Hirakawa H, Sato S, Hayashi M, Kawaguchi M (2014) Nodule inception creates a long-distance negative feedback loop involved in homeostatic regulation of nodule organ production. PNAS USA 111:14607–14612. https://doi.org/10.1073/pnas.1412716111
Suzaki T, Yoro E, Kawaguchi M (2015) Leguminous plants: inventors of root nodules to accommodate symbiotic bacteria. Int Rev Cell Mol Biol 316:111–158
Taiz L, Zeiger E (2010) Plant physiology, 6th edn. Sinauer Associates, Sunderland
Tampakaki AP (2014) Commonalities and differences of T3SSs in rhizobia and plant pathogenic bacteria. Front Plant Sci 5:114. https://doi.org/10.3389/fpls.2014.00114
Teulet A, Busset N, Fardoux J, Gully D, Chaintreuil C, Cartieaux F, Jauneau A, Comorge V, Okazaki S, Kaneko T, Gressent F, Nouwen N, Arrighi JF, Koebnik R, Mergaert P, Deslandes L, Giraud E (2019) The rhizobial type III effector eRNA confers the ability to form nodules in legumes. PNAS USA 116:21758–21768. https://doi.org/10.1073/pnas.1904456116
Tirichine L, Sandal N, Madsen LH, Radutoiu S, Albrektsen AS, Sato S, Asamizu E, Tabata S, Stougaard J (2007) A gain-of-function mutation in a cytokinin receptor triggers spontaneous root nodule organogenesis. Science 315:104–107. https://doi.org/10.1126/science.1132397
Visweswaran GRR, van Roosmalen KLM, Kok J, Buist G (2014) Exploiting the peptidoglycan- binding motif, LysM, for medical and industrial applications. Appl Microbiol Biotechnol 98:4331–4345
Wais RJ, Keating DH, Long SR (2002) Structure-function analysis of nod factor-induced root hair calcium spiking in Rhizobium-legume symbiosis. Plant Physiol 129:211–224. https://doi.org/10.1104/pp.010690
Wang Q, Liu J, Zhu H (2018) Genetic and molecular mechanisms underlying symbiotic specificity in legume-Rhizobium interactions. Front Plant Sci 9:313. https://doi.org/10.3389/fpls.2018.00313
Wei M, Takeshima K, Yokoyama T, Minamisawa K, Mitsui H, Itakura M, Kaneko T, Tabata S, Saeki K, Omori H, Tajima S, Uchiumi T, Abe M, Ishii S, Ohwada T (2010) Temperature dependent expression of type III secretion system genes and its regulation in Bradyrhizobium japonicum. Mol Plant Microbe Interact 23:628–637. https://doi.org/10.1094/MPMI-23-5-0628
Yasuda M, Miwa H, Masuda S, Takebayashi Y, Sakakibara H, Okazaki S (2016) Effector-triggered immunity determines host genotype specific incompatibility in legume-Rhizobium symbiosis. Plant Cell Physiol 57:1791–1800. https://doi.org/10.1093/pcp/pcw104
Author information
Authors and Affiliations
Contributions
DP and SM conceptualize the research, DP performed data curation, wrote the original draft; SM supervised, reviewed and edited the Manuscript.
Corresponding author
Ethics declarations
The authors declare no conflict of interest.
Funding
This work is supported by the Department of Biotechnology, Govt. of India [Grant No- BT/PR23731/BPA/118/344/2017].
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Patra, D., Mandal, S. Nod–factors are dispensable for nodulation: A twist in bradyrhizobia-legume symbiosis. Symbiosis 86, 1–15 (2022). https://doi.org/10.1007/s13199-021-00826-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13199-021-00826-9