Abstract
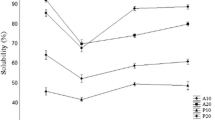
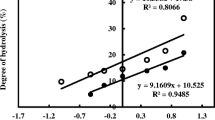
Fish protein hydrolysates (FPH) from fresh water carps Catla catla, Labeo rohita and Cirrhinus mrigala were prepared with 5, 10, 15 and 20% degree of hydrolysis (DH) using papain enzyme. FPH were evaluated for antioxidant properties using in vitro assays such as DPPH free radical scavenging activity (at 10 mg/ml), ferric reducing antioxidant power assay (at 20 mg/ml) and linoleic acid peroxidation inhibition activity (at 10 mg/ml). Antioxidant properties of FPH varied with species and DH. The DPPH radical scavenging activity, linoleic acid peroxidation inhibition activity and ferric reducing antioxidant power (as absorbance at 700 nm) of FPH from carps was in the range of 59–92%, 52–85% and 0.388–0.663 respectively. Based on the overall antioxidant activity, FPH from C. catla with 20% DH was added to oil sardine mince at different concentration (0.1, 0.2 and 0.4%) and found to inhibit effectively the formation of peroxides and malonaldehyde in dose dependent manner. FPH from C. catla with 20% DH was fractionated using size exclusion chromatography and had three different peptide fractions with the approximate molecular weight of 6561–2106 Da (fraction 1), 1942–994 Da (fraction 2) and 935–383 Da (fraction 3). The present study showed promising results that the fish protein hydrolysates from fresh water carps muscle proteins can be used as natural antioxidants in food system. Production of fish protein hydrolysates with nutraceutical properties could be the way forward for better utilization and value addition.




Similar content being viewed by others
Availability of data and material
On appropriate request the data will be shared on public and profit interest.
Abbreviations
- AOAC:
-
Association of Analytical Chemists International
- CPH:
-
Catla protein hydrolysates
- Da:
-
Dalton
- DH:
-
Degree of hydrolysis
- DPPH:
-
2,2 Diphenyl-1-picrylhydrazyl
- E/S:
-
Enzyme to substrate ratio
- FAO:
-
Food and Agriculture organization
- FPH:
-
Fish protein hydrolysates
- FRAP:
-
Ferric reducing antioxidant power
- IMC:
-
Indian Major Carps
- LAPI:
-
Linoleic acid peroxidation inhibition (LAPI) activity
- MPH:
-
Mrigal protein hydrolysates
- pH:
-
Hydrogen ion potential
- PV:
-
Peroxide value
- RPH:
-
Rohu protein hydrolysates
- TBA:
-
Thiobarbituric acid
- TBARS:
-
Thiobarbituric acid-reactive substances
- TCA:
-
Trichloro acetic acid
- TVE:
-
Total volume of extract
References
AOAC (2000) Official Methods of Analysis of Association of Analytical Chemists International, 17th edn. Washington, USA
Chalamaiah M, Dinesh Kumar B, Hemalatha R, Jyothirmayi T (2012) Fish protein hydrolysates: proximate composition, amino acid composition, antioxidant activities and applications: a review. Food Chem 135:3020–3038
Chung YC, Chang CT, Chao WW, Lin CF, Chou ST (2002) Antioxidative activity and safety of the 50% ethanolic extract from red bean fermented by Bacillus subtilis IMR-NK1. J Agric Food Chem 50:2454–2458
Decker EA, Diaz M (2004) Antioxidant mechanisms of caseinophosphopeptides and casein hydrolysates and their application in ground beef. J Agric Food Chem 52:8208–8213
Elavarasan K, Shamasundar BA (2014) Angiotensin I-converting enzyme inhibitory activity of protein hydrolysates prepared from three freshwater carps (Catla catla, Labeo rohita and Cirrhinus mrigala) using Flavorzyme. Int J Food Sci Technol 49:1344–1350
Elias RJ, Kellerby SS, Decker EA (2008) Antioxidant activity of proteins and peptides. Crit Rev Food Sci Nutr 48:430–441
FAO (Food and Agricultural Organisation) (2020) The state of the world fisheries and aquaculture. Rome
FAO, Fisheries and aquaculture software. FishStatJ—Software for Fishery and Aquaculture Statistical Time Series. In: FAO Fisheries Division [online]. Rome. Updated 14 September 2020. (Cited 11 December 2020). http://www.fao.org/fishery/
Gbogouri GA, Linder M, Fanni J, Parmentier M (2004) Influence of hydrolysis degree on the functional properties of salmon byproducts hydrolysates. J Food Sci 69:C615–C622
Ha M, Aea B, Carne A, Hopkins DL (2012) Characterisation of commercial papain, bromelain, actinidin and zingibain protease preparations and their activities toward meat proteins. Food Chem 134:95–105
Kim S, Je J, Kim S (2007) Purification and characterization of antioxidant peptide from hoki (Johnius belengerii) frame protein by gastrointestinal digestion. J Nutr Biochem 18:31–38
Kong BH, Xiong YL (2006) Antioxidant activity of zein hydrolysates in a liposome system and the possible mode of action. J Agric Food Chem 54:6059–6068
Kristinsson HG, Rasco BA (2000) Fish Protein hydrolysates: production, biochemical and functional properties. Crit Rev Food Sci Nutr 40:43–81
Lajolina P, Laine J, Linko P (1983) Quality changes in minced fish during cold and frozen storage. In: Zerthan et al (eds) Thermal processing and quality of foods. Elsvier Applied Science Publishers Ltd, Amsterdam
Li X, Luo Y, You J, Shen H, Du J (2012) In vitro antioxidant activity of papain-treated grass carp (Ctenopharyngodon idellus) protein hydrolysate and the preventive effect on fish mince system. Int J Food Sci Tech 47:961–967
Liu Q, Kong B, Xiong YL, Xia X (2010) Antioxidant activity and functional properties of porcine plasma protein hydrolysate as influenced by the degree of hydrolysis. Food Chem 118:403–410
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Maqsood S, Benjakul S (2011) Oxidation of lipid and protein in horse mackerel (Trachurus trachurus) mince and washed minces during processing and storage. Food Chem 124:875–883
Nimse SB, Pal D (2015) Free radicals, natural antioxidants, and their reaction mechanisms. RSC Adv 35:27986–28006
Raghavan S, Hultin HO (2006) Oxidative stability of a cod-canola oil model system: effect of order addition of tocopherol and canola oil to washed, minced cod muscle. J Aquatic Food Prod Technol 158:37–45
Raghavan S, Kristinsson HG (2008) Antioxidative efficacy of alkali-treated tilapia protein hydrolysates: a comparative study of five enzymes. J Agric Food Chem 56:1434–1441
Raghavan S, Kristinsson HG, Leeuwenburgh C (2008) Radical scavenging and reducing ability of tilapia (Oreochromis niloticus) protein hydrolysates. J Agri Food Chem 56:10359–10367
Ren J, Zhao M, Shi J, Wang J, Jiang Y, Cui C, Kukuda Y, Xue JS (2008) Optimization of antioxidant peptide production from grass carp sarcoplasmic protein using response surface methodology. LWT-Food Sci Technol 41:1624–1632
Richards MP, Hultin HO (2002) Contribution of blood and blood components to lipid oxidation in fish muscle. J Agric Food Chem 50:555–564
Shanta NC, Decker EA (1994) Rapid, sensitive, iron-based spectrophotometric methods for determination of peroxide values of food lipids. J Am Oil Chem Soc 77:421–424
Sohn JH, Taki Y, Ushio H, Kohata T, Shioya I, Ohshima T (2005) Lipid oxidations in ordinary and dark muscles of fish: influences on rancid off odor development and color darkening of yellowtail flesh during ice storage. J Food Sci 70:s490–s496
Udenigwe CC, Aluko RE (2011) Chemometric analysis of the amino acid requirements of antioxidant food protein hydrolysates. Int J Mol Sci 12:3148–3161
Vate NK, Benjakul S (2013) Antioxidative activity of melanin free ink from splendid squid (Loligo formosana). Int Aquat Res 5. www.intaquares.com
Wu CH, Chen HM, Shiau CY (2003) Free amino acids and peptides as related to antioxidant properties in protein hydrolysates of mackerel (Scomber austriasi). Food Res Int 36:949–957
Yen GC, Chen HY (1995) Antioxidant activity of various tea extract in relation to their antimutagenicity. J Agric Food Chem 43:27–32
Acknowledgement
The funding support to the authors provided by Indian Council of Agricultural research (ICAR), New Delhi, India under National Agricultural Innovative Project (NAIP), Component II, is gratefully acknowledged. The Director, ICAR-Central Institute of Fisheries Technology and the Dean, College of Fisheries, Mangalore are acknowledged for their kind support rendered.
Funding
The funding support to the authors was received from Indian Council of Agricultural research (ICAR), New Delhi, India under National Agricultural Innovative Project (NAIP), Component II.
Author information
Authors and Affiliations
Contributions
Dr. BAS conceptualized the work, involved in designing of the experiment, analysis and interpretation of the results and drawing of conclusion, correcting the language and proof reading the manuscript. Dr. EK involved in designing and execution of the experiment, collection of data, analysis and interpretation of the results and drafting the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Authors do not have any conflict of interest in the presented research work.
Consent to Participate
I hereby undertake that I shall review at least three manuscripts submitted to JFST in my own specialization.
Consent for publication
I also declare that the work submitted has not been carried out previously and not under submission/consideration for publication in any other journal.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Elavarasan, K., Shamasundar, B.A. Antioxidant properties of papain mediated protein hydrolysates from fresh water carps (Catla catla, Labeo rohita and Cirrhinus mrigala) and its application on inhibition of lipid oxidation in oil sardine mince during ice storage. J Food Sci Technol 59, 636–645 (2022). https://doi.org/10.1007/s13197-021-05053-0
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13197-021-05053-0