Abstract
Introduction
Crotalidae immune F(ab’)2 (Fab2AV) became available in the USA in 2019 for treatment of rattlesnake envenomation. In the clinical trial comparing Fab2AV to crotalidae immune polyvalent fab (FabAV), Fab2AV was associated with less late hemotoxicity. The purpose of this study was to describe outcomes following use of Fab2AV in patients with rattlesnake envenomation in Arizona.
Methods
This is an observational study of patients admitted to a medical toxicology service at two hospitals in Arizona between January 1, 2019 and December 31, 2020. Patients with rattlesnake envenomation who received Fab2AV were included. Patients who received FabAV, alone or in combination with Fab2AV, were excluded. The main outcomes of interest were antivenom dose, adverse reactions, late hemotoxicity, and hospital readmission or retreatment.
Results
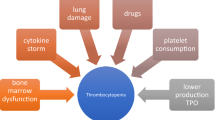
Forty-six patients were included. The mean age was 40 years, with 15% under 12 years of age. All exhibited swelling, 20% thrombocytopenia, and 35% coagulopathy. Median time to treatment was 3 h and median total Fab2AV dose was 20 vials. Three patients had an acute reaction to Fab2AV which was non-life-threatening and resolved with antihistamines and/or steroids. In the follow-up period, one case of delayed thrombocytopenia (platelets = 108 K/mm3) and one case of recurrent thrombocytopenia (platelets = 111 K/mm3) were identified. There was no late coagulopathy. Five patients reported symptoms consistent with mild serum sickness.
Conclusions
In this series of patients with rattlesnake envenomation in Arizona who were treated with Fab2AV, there were no cases of clinically significant late hemotoxicity, and no patients required late retreatment with antivenom. Acute and delayed reactions did occur in some patients but were mild and easily treated.

Similar content being viewed by others
References
Seifert SA, Boyer LV, Benson BE, Rogers JJ. AAPCC database characterization of native U.S. venomous snake exposures, 2001-2005. Clin Toxicol (Phila). 2009;47(4):327–35. https://doi.org/10.1080/15563650902870277.
Bush S, Ruha AM, Seifert S, Morgan D, Lewis B, Arnold T, Clark R, Meggs W, Toschloq E, Borron S, Figge G, Sollee D, Shirazi F, Wolk R, Chazal I, Quan D, Garcia-Ubbelohde W, Alagon A, Gerkin R, Boyer L. Comparison of F(ab’)2 versus Fab antivenom for pit viper envenomation: a prospective, blinded, multicenter, randomized clinical trial. Clin Toxicol. 2015;53(1):37–45.
Ruha AM, Curry SC, Beuhler M, Katz K, Brooks DE, Graeme KA, Wallace K, Gerkin R, LoVecchio F, Wax P, Selden B. Initial postmarketing experience with crotalidae polyvalent immune Fab for treatment of rattlesnake envenomation. Ann Emerg Med. 2002;39(6):609–15.
Ruha AM, Curry SC, Albrecht C, Riley B, Pizon A. Late hematologic toxicity following treatment of rattlesnake envenomation with crotalidae polyvalent immune fab antivenom. Toxicon. 2011;57(1):53–9.
Cannon RD, Ruha AM, Kashani J. Acute hypersensitivity reactions associated with the administration of crotalidae polyvalent immune fab antivenom. Ann Emerg Med. 2008;51(4):407–11.
Spyres MB, Skolnik A, Moore E, Padilla-Jones A, Gerkin RD, Ruha AM. Comparison of antivenom dosing strategies for rattlesnake envenomation. Crit Care Med. 2018. https://doi.org/10.1097/CCM.0000000000003079.
Levine M, Ruha AM, Padilla-Jones A, Gerkin R, Thomas SH. Bleeding following rattlesnake envenomation in patients with pre-envenomation use of antiplatelet or anticoagulant medications. Acad Emerg Med. 2014;21(3):301–7.
Moore EC, Porter LM, Ruha AM. Rattlesnake venom-induced recurrent coagulopathy in first trimester pregnant women - two cases. Toxicon. 2019;163:8–11. https://doi.org/10.1016/j.toxicon.2019.03.006.
CROFAB® crotalidae polyvalent immune fab (ovine) lyophilized powder for solution for intravenous injection initial U.S. Approval: 2000. Available at: https://www.fda.gov/files/vaccines%2C%20blood%20%26%20biologics/published/Package-Insert---CroFab.pdf Accessed on June 18, 2021.
ANAVIP® crotalidae immune F(ab’)2 (equine) lyophilized powder for solution for injection for intravenous use only Available at: https://www.fda.gov/media/92139/download Accessed on June 18, 2021.
LoVecchio F, Klemens J, Roundy EB, Klemens A. Serum sickness following administration of antivenin (Crotalidae) polyvalent in 181 cases of presumed rattlesnake envenomation. Wilderness Environ Med. 2003;14(4):220–1. https://doi.org/10.1580/1080-6032(2003)14[220:ssfaoa]2.0.co;2.
Funding
None
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of Interest
None
Additional information
Supervising Editor: Mark B. Mycyk, MD
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Data in this manuscript were presented at the North American Congress of Clinical Toxicology (NACCT) annual meeting in Atlanta, GA (Virtual) in October 2021.
Rights and permissions
About this article
Cite this article
Ruha, AM., Padilla-Jones, A., Canning, J. et al. Early Experience with Crotalidae Immune F(ab’)2 Antivenom to Treat Arizona Rattlesnake Envenomations. J. Med. Toxicol. 18, 38–42 (2022). https://doi.org/10.1007/s13181-021-00859-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13181-021-00859-y