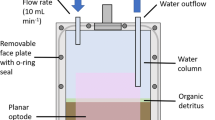
Abstract
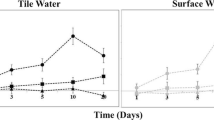
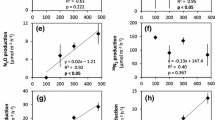
Microbial nitrate reduction in wetland soils and sediments plays an important role mitigating the excess of nitrogen released into the environment. The reduction of nitrate to its reduced gaseous form, i.e. denitrification, is tightly linked to the oxidation of organic carbon. In intertidal wetlands, the organic carbon derived from aquatic or terrestrial primary producers strongly differs in composition, which in turn might affect nitrate reduction rates. In the current study the impact of carbon quality on benthic nitrate reduction in a temperate intertidal wetland sediment was determined. To this end, nitrate reduction rates were determined over a three-week period using an anoxic controlled flow-through reactor approach using sediments amended with carbon from different vegetation types. Organic carbon from algal or plants was mixed with the sediment at the same absolute quantity of carbon. A significant initial increase in nitrate reduction rates was observed in the presence of algal (microphytobenthos) derived organic carbon, most likely due to its labile structure; however, rates decreased rapidly, most likely due to a depletion of the labile source. This indicates the dependence of denitrifiers to the microalgae turnover in non-vegetated sediment. Despite a delay, the addition of plant derived organic carbon, increased nitrate reduction rates with a higher amount of total nitrate reduced over the duration of experiment compared to the microphytobenthos addition. These results highlight that presence of both labile and recalcitrant carbon sources in sediment is essential to support an efficient nitrogen biogeochemical cycle.






Similar content being viewed by others
Data Availability
All relevant data are within the manuscript or in supplementary material.
Code Availability
Not Applicable.
References
Alldred M, Baines SB (2016) Effects of wetland plants on denitrification rates: a meta-analysis. Ecol Appl 26:676–685
Anderson DM, Glibert PM, Burkholder JM (2002) Harmful algal blooms and eutrophication nutrient sources, composition, and consequences. Estuaries 25:704–726
Barbier E, Hacker S (2011) The value of estuarine and coastal ecosystem services. Ecol Monogr 81:169–193
Bastian F, Bouziri L, Nicolardot B, Ranjard L (2009) Impact of wheat straw decomposition on successional patterns of soil microbial community structure. Soil Biol Biochem 41:262–275. https://doi.org/10.1016/j.soilbio.2008.10.024
Bastviken SK, Eriksson PG, Ekström A, Tonderski K (2007) Seasonal denitrification potential in wetland sediments with organic matter from different plant species. Water Air Soil Pollut 183:25–35. https://doi.org/10.1007/s11270-007-9352-x
Bastviken SK, Eriksson PG, Premrov A, Tonderski K (2005) Potential denitrification in wetland sediments with different plant species detritus. Ecol Eng 25:183–190. https://doi.org/10.1016/j.ecoleng.2005.04.013
Baumann K, Marschner P, Smernik RJ, Baldock JA (2009) Residue chemistry and microbial community structure during decomposition of eucalypt, wheat and vetch residues. Soil Biol Biochem 41:1966–1975. https://doi.org/10.1016/j.soilbio.2009.06.022
Bouillon S, Boschker HTS (2006) Bacterial carbon sources in coastal sediments: a review based on stable isotope data of biomarkers. Biogeosciences 3:175–185. https://doi.org/10.5194/bgd-2-1617-2005
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Burdige DJ (2007) Preservation of organic matter in marine sediments: Controls, mechanisms, and an imbalance in sediment organic carbon budgets? Chem Rev 107:467–485. https://doi.org/10.1021/cr050347q
Caffrey J, Kemp W (1990) Nitrogen cycling in sediments with estuarine populations of Potamogeton perfoliatus and Zostera marina. Mar Ecol Prog Ser 66:147–160. https://doi.org/10.3354/meps066147
Canuel EA, Martens CS (1996) Reactivity of recently deposited organic matter: degradation of lipid compounds near the sediment-water interface. Geochim Cosmochim Acta 60:1793–1806. https://doi.org/10.1016/0016-7037(96)00045-2
Challinor JM (1995) Characterisation of wood by pyrolysis derivatisation—gas chromatography/mass spectrometry. J Anal Appl Pyrolysis 35:93–107
Chen Y, Wen Y, Cheng J, Xue CH, Yang D, Zhou Q (2011) Effects of dissolved oxygen on extracellular enzymes activities and transformation of carbon sources from plant biomass: Implications for denitrification in constructed wetlands. Bioresour Technol 102:2433–2440. https://doi.org/10.1016/j.biortech.2010.10.122
Decleyre H, Heylen K, Van Colen C, Willems A (2015) Dissimilatory nitrogen reduction in intertidal sediments of a temperate estuary: Small scale heterogeneity and novel nitrate-to-ammonium reducers. Front Microbiol 6. https://doi.org/10.3389/fmicb.2015.01124
Dick TM, Osunkoya OO (2000) Influence of tidal restriction floodgates on decomposition of mangrove litter. Aquat Bot 68:273–280. https://doi.org/10.1016/S0304-3770(00)00119-4
Dodla SK, Wang JJ, DeLaune RD, Cook RL (2008) Denitrification potential and its relation to organic carbon quality in three coastal wetland soils. Sci Total Environ 407:471–480. https://doi.org/10.1016/j.scitotenv.2008.08.022
Dubois M, Gilles KA, Hamilton JK, Rebers Pa, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Ehrenfeld JG (2003) Effects of exotic plant invasions on soil nutrient cycling processes. Ecosystems 6:503–523. https://doi.org/10.1007/s10021-002-0151-3
Ehrenfeld JG, Ravit B, Elgersma K (2005) Feedback in the plant-soil system. Annu Rev Environ Resour 30:75–115. https://doi.org/10.1146/annurev.energy.30.050504.144212
Fabbri D, Helleur R (1999) Characterization of the tetramethylammonium hydroxide thermochemolysis products of carbohydrates. J Anal Appl Pyrolysis 49:277–293. https://doi.org/10.1016/S0165-2370(98)00085-0
Fernandes SO, Dutta P, Gonsalves MJ, Bonin PC, LokaBharathi PA (2016) Denitrification activity in mangrove sediments varies with associated vegetation. Ecol Eng 95:671–681. https://doi.org/10.1016/j.ecoleng.2016.06.102
Fontaine S, Barot S (2005) Size and functional diversity of microbe populations control plant persistence and long-term soil carbon accumulation. Ecol Lett 8:1075–1087. https://doi.org/10.1111/j.1461-0248.2005.00813.x
Fontaine S, Henault C, Aamor A et al (2011) Fungi mediate long term sequestration of carbon and nitrogen in soil through their priming effect. Soil Biol Biochem 43:86–96. https://doi.org/10.1016/j.soilbio.2010.09.017
Fontaine S, Mariotti A, Abbadie L (2003) The priming effect of organic matter: a question of microbial competition? Soil Biol. Biochem 35:837–843. https://doi.org/10.1016/S0038-0717(03)00123-8
Galloway JN, Aber JD, Erisman JW, Seitzinger SP, Howarth RW, Cowling EB, Cosby BJ (2003) The nitrogen cascade. Bioscience 53:341–356. https://doi.org/10.1641/0006-3568(2003)053[0341:TNC]2.0.CO;2
Van de Graaf AA, Mulder A, De Bruijn P, Jetten MSM, Robertson LA, Kuenen JG (1995) Anaerobic oxidation of ammonium is a biologically mediated process. Appl Environ Microbiol 61:1246–1251. https://doi.org/10.1128/aem.61.4.1246-1251.1995
Granéli E, Weberg M, Salomon PS (2008) Harmful algal blooms of allelopathic microalgal species: The role of eutrophication. Harmful Algae 8:94–102. https://doi.org/10.1016/j.hal.2008.08.011
Hardison AK, Canuel EA, Anderson IC, Tobias CR, Veuger B, Waters MN (2013) Microphytobenthos and benthic macroalgae determine sediment organic matter composition in shallow photic sediments. Biogeosciences 10:5571–5588. https://doi.org/10.5194/bg-10-5571-2013
Hedges JI, Weliky K (1989) Diagenesis of conifer needles in a coastal marine environment. Geochim Cosmochim Acta 53:2659–2673. https://doi.org/10.1016/0016-7037(89)90137-3
Herman PMJ, Middelburg JJ, Heip CHR (2001) Benthic community structure and sediment processes on an intertidal flat: Results from the ECOFLAT project. Cont Shelf Res 21:2055–2071. https://doi.org/10.1016/S0278-4343(01)00042-5
Hoffman M, Decho AW (1999) Extracellular enzymes within microbial biofilms and the role of the extracellular polymer matrix BT. In: Wingender J, Neu TR, Flemming H-C (eds) Microbial extracellular polymeric substances: characterization, structure and function. Springer, Berlin, pp 217–230
Hume NP, Fleming MS, Horne AJ (2002) Denitrification potential and carbon quality of four aquatic plants in wetland microcosms. Soil Sci Soc Am J 66:1706–1712. https://doi.org/10.2136/sssaj2002.1706
Jeanneau L, Jaffrezic A, Pierson-Wickmann A-C, Gruau G, Lambert T, Petitjean P (2014) Constraints on the sources and production mechanisms of dissolved organic matter in soils from molecular biomarkers. Vadose Zone J 13:2014020015. https://doi.org/10.2136/vzj2014.02.0015
Kawaida S, Nanjo K, Ohtsuchi N, Kohno H, Sano M (2019) Cellulose digestion abilities determine the food utilization of mangrove estuarine crabs. Estuar Coast Shelf Sci 222:43–52. https://doi.org/10.1016/j.ecss.2019.04.004
Kuenen JG (2008) Anammox bacteria: from discovery to application. Nat Rev Microbiol 6:320–326. https://doi.org/10.1038/nrmicro1857
Laverman AM, Van Cappellen P, Van Rotterdam-Los D, Pallud C, Abell J (2006) Potential rates and pathways of microbial nitrate reduction in coastal sediments. FEMS Microbiol Ecol 58:179–192. https://doi.org/10.1111/j.1574-6941.2006.00155.x
Laverman AM, Morelle J, Roose-Amsaleg C, Pannard A (2021) Estuarine benthic nitrate reduction rates: Potential role of microalgae? Estuar Coast Shelf Sci. https://doi.org/10.1016/j.ecss.2021.107394
Laverman AM, Pallud C, Abell J, Van Cappellen P (2012) Comparative survey of potential nitrate and sulfate reduction rates in aquatic sediments. Geochim Cosmochim Acta 77:474–488. https://doi.org/10.1016/j.gca.2011.10.033
Lee NM, Welander T (1996) Use of protozoa and metazoa for decreasing sludge production in aerobic wastewater treatment. Biotechnol Lett 18:429–434. https://doi.org/10.1007/BF00143465
Lemley DA, Snow GC, Human LRD (2014) The decomposition of estuarine macrophytes under different temperature regimes. Water SA 40:117–124
Leng MJ, Lewis JP (2017) C/N ratios and carbon isotope composition of organic matter in estuarine environments. 213–237. https://doi.org/10.1007/978-94-024-0990-1_9
Marinari S, Masciandaro G, Ceccanti B, Grego S (2000) Influence of organic and mineral fertilisers on soil biological and physical properties. Bioresour Technol 72:9–17. https://doi.org/10.1016/S0960-8524(99)00094-2
Marinari S, Moscatelli C, Grego S (2014) Enzymes at plant-soil interface. In: Gianfreda L, Rao MA (eds) Enzymes in agricultural sciences. Foster City, OMICS eBooks Group, pp 94–109
Marschner P, Umar S, Baumann K (2011) The microbial community composition changes rapidly in the early stages of decomposition of wheat residue. Soil Biol Biochem 43:445–451. https://doi.org/10.1016/j.soilbio.2010.11.015
Maxwell TL, Augusto L, Bon L et al (2020) Effect of a tree mixture and water availability on soil nutrients and extracellular enzyme activities along the soil profile in an experimental forest. Soil Biol Biochem 148. https://doi.org/10.1016/j.soilbio.2020.107864
McLusky DS, Elliott M (2004) The estuarine ecosystem: ecology, threats and management. OUP, Oxford
McMahon SK, Williams MA, Bottomley PJ, Myrold DD (2005) Dynamics of microbial communities during decomposition of carbon-13 labeled ryegrass fractions in soil. Soil Sci Soc Am J 69:1238–1247. https://doi.org/10.2136/sssaj2004.0289
Mondini C, Cayuela ML, Sanchez-Monedero MA, Roig A, Brookes PC (2006) Soil microbial biomass activation by trace amounts of readily available substrate. Biol Fertil Soils 42:542–549. https://doi.org/10.1007/s00374-005-0049-2
Morelle J, Roose-Amsaleg C, Laverman AM (2022) Microphytobenthos as a source of labile organic matter for denitrifying microbes. Estuar Coast Shelf Sci 275. https://doi.org/10.1016/j.ecss.2022.108006
Nicolardot B, Recous S, Mary B (2001) Simulation of C and N mineralisation during crop residue decomposition : A simple dynamic model based on the C : N ratio of the residues Author ( s ): B. Nicolardot, S. Recous and B. Mary Source : Plant and Soil, Vol. 228, No. 1, THE 10TH INTERNA. Plant Soil 228:83–103
Nielsen SL, Banta GT, Pedersen MF (2004) Decomposition of marine primary producers: consequences for nutrient recycling and retention in coastal ecosystems. In: Nielsen SL, Banta GT, Pedersen MF (eds) Estuarine nutrient cycling: the influence of primary producers. Aquatic Ecology Book Series, vol 2. Springer, Dordrecht. https://doi.org/10.1007/978-1-4020-3021-5_7
Nierop KGJ, Preston CM, Kaal J (2005) Thermally assisted hydrolysis and methylation of purified tannins from plants. Anal Chem 77:5604–5614. https://doi.org/10.1021/ac050564r
Nierop KGJ, Verstraten JM (2004) Rapid molecular assessment of the bioturbation extent in sandy soil horizons under pine using ester-bound lipids by on-line thermally assisted hydrolysis and methylation-gas chromatography/mass spectrometry. Rapid Commun Mass Spectrom 18:1081–1088. https://doi.org/10.1002/rcm.1449
De Nobili M, Contin M, Mondini C, Brookes PC (2001) Soil microbial biomass is triggered into activity by trace amounts of substrate. Soil Biol Biochem 33:1163–1170. https://doi.org/10.1016/S0038-0717(01)00020-7
Obaja D, MacÉ S, Mata-Alvarez J (2005) Biological nutrient removal by a sequencing batch reactor (SBR) using an internal organic carbon source in digested piggery wastewater. Bioresour Technol 96:7–14. https://doi.org/10.1016/j.biortech.2004.03.002
Phillips RL, Zak DR, Holmes WE, White DC (2002) Microbial community composition and function beneath temperate trees exposed to elevated atmospheric carbon dioxide and ozone. Oecologia 131:236–244. https://doi.org/10.1007/s00442-002-0868-x
del Río JC, Hatcher PG (1998) Analysis of aliphatic biopolymers using thermochemolysis with tetramethylammonium hydroxide (TMAH) and gas chromatography-mass spectrometry. Org Geochem 29:1441–1451. https://doi.org/10.1016/S0146-6380(98)00070-9
Sanseverino I, Conduto D, Pozzoli L, Dobricic S, Lettieri T (2016) Algal bloom and its economic impact; EUR 27905 EN. https://doi.org/10.2788/660478
Sardans J, Rivas-Ubach A, Peñuelas J (2012) The C:N: P stoichiometry of organisms and ecosystems in a changing world: A review and perspectives. Perspect Plant Ecol Evol Syst 14:33–47. https://doi.org/10.1016/j.ppees.2011.08.002
Schimel JP, Weintraub MN (2003) The implications of exoenzyme activity on microbial carbon and nitrogen limitation in soil: A theoretical model. Soil Biol Biochem 35:549–563. https://doi.org/10.1016/S0038-0717(03)00015-4
Shao X, Yang W, Wu M (2015) Seasonal dynamics of soil labile organic carbon and enzyme activities in relation to vegetation types in Hangzhou Bay tidal flat wetland. PLoS One 10:1–15. https://doi.org/10.1371/journal.pone.0142677
Sirivedhin T, Gray KA (2006) Factors affecting denitrification rates in experimental wetlands: field and laboratory studies. Ecol Eng 26:167–181. https://doi.org/10.1016/j.ecoleng.2005.09.001
Starr RC, Gillham RW (1993) Denitrification and organic carbon availability in two aquifers. Groundwater 31:934–947. https://doi.org/10.1111/j.1745-6584.1993.tb00867.x
Stelzer RS, Scott JT, Bartsch LA, Parr TB (2014) Particulate organic matter quality influences nitrate retention and denitrification in stream sediments: evidence from a carbon burial experiment. Biogeochemistry 119:387–402. https://doi.org/10.1007/s10533-014-9975-0
Strous M, Kuenen JG, Jetten MSM (1999) Key physiology of anaerobic ammonium oxidation. Appl Environ Microbiol 65:3248–3250. https://doi.org/10.1128/aem.65.7.3248-3250.1999
Tiedje JM (1988) Ecology of denitrification and dissimilatory nitrate reduction to ammonium. In: Zehnder AJB (ed) Environ Microbiol Anaerobes. John Wiley and Sons, NY, pp 179–244
Tiedje JM, Sexstone AJ, Myrold DD, Robinson JA (1983) Denitrification: ecological niches, competition and survival. Antonie Van Leeuwenhoek 48:569–583. https://doi.org/10.1007/BF00399542
Vichkovitten T, Holmer M (2004) Contribution of plant carbohydrates to sedimentary carbon mineralization. Org Geochem 35:1053–1066. https://doi.org/10.1016/j.orggeochem.2004.04.007
Waldrop MP, Firestone MK (2004) Microbial community utilization of recalcitrant and simple carbon compounds: Impact of oak-woodland plant communities. Oecologia 138:275–284. https://doi.org/10.1007/s00442-003-1419-9
Wall DH, Moore JC (1999) Interactions underground: Soil biodiversity, mutualism, and ecosystem processes. Bioscience 49:109–117. https://doi.org/10.2307/1313536
Wang Q, Wang S, He T, Liu L, Wu J (2014) Response of organic carbon mineralization and microbial community to leaf litter and nutrient additions in subtropical forest soils. Soil Biol Biochem 71:13–20. https://doi.org/10.1016/j.soilbio.2014.01.004
Windham L, Ehrenfeld JG (2003) Net impact of a plant invasion on nitrogen-cycling processes within a brackish tidal marsh. Ecol Appl 13:883–896. https://doi.org/10.1890/02-5005
Yin R, Deng H, Wang HL, Zhang B (2014) Vegetation type affects soil enzyme activities and microbial functional diversity following re-vegetation of a severely eroded red soil in sub-tropical China. Catena 115:96–103. https://doi.org/10.1016/j.catena.2013.11.015
Zhou S, Borjigin S, Riya S, Terada A, Hosomi M (2014) The relationship between anammox and denitrification in the sediment of an inland river. Sci Total Environ 490:1029–1036. https://doi.org/10.1016/j.scitotenv.2014.05.096
Acknowledgements
This work was supported by the GIP Seine-Aval project “FEREE” (Program SA6) and the CESAM (UIDP/50017/2020 + UIDB/50017/2020 + LA/P/0094/2020). The authors acknowledge Estelle Langlois (PI of the project), Samuel Hermant for its implication in this experimental study, as well as Garance Regimbeau, Guillaume Bouger, and Sarah Coffinet for their help during sampling. The ECOCHIM platform of Rennes University is acknowledged for help with the Gallery device. The authors finally acknowledge the anonymous reviewers that significantly helped to improve the manuscript.
Funding
This work was funded by the GIP Seine-Aval through the SA6 – FEREE project (https://www.seine37aval.fr/projet/feree/) and supported by the CESAM (UIDP/50017/2020 + UIDB/50017/2020 + LA/P/0094/2020).
Author information
Authors and Affiliations
Contributions
JM performed conception and design of this study, material preparation, sample collection, analyzes, interpretation of data, and wrote the first draft of the manuscript. LJ performed analyzes, interpretation of data, and contributed to the manuscript. AML performed conception and design of this study, material preparation, analysis, interpretation of data and contributed to the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
The prefectural decree n ° ME/2020/09 was established to authorize the samplings within the national reserve carried out for this study (Project FEREE). No further authorization was required, and this field study did not involve endangered or protected species.
Consent to Participate
All authors agreed to participate in this research study
Consent for Publication
All authors agreed to published this research study.
Conflicts of Interest/Competing Interests
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Morelle, J., Jeanneau, L. & Laverman, A.M. Organic Carbon Quality Impacts Benthic Microbial Nitrate Reduction. Wetlands 43, 99 (2023). https://doi.org/10.1007/s13157-023-01747-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13157-023-01747-7