Abstract
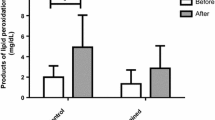
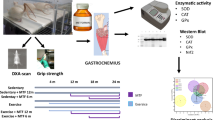
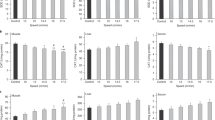
Increase in heart metabolism during severe exercise facilitates production of ROS and result in oxidative stress. Due to shortage of information, the effect of chronic strength exercise on oxidative stress and contractile function of the heart was assessed to explore the threshold for oxidative stress in this kind of exercise training. Male Wistar rats (80) were divided into two test groups exercised 1 and 3 months and two control groups without exercise. Strength exercise was carried by wearing a Canvas Jacket with weights and forced rats to lift the weights. Rats were exercised at 70% of maximum lifted weight 6 days/week, four times/day, and 12 repetitions each time. Finally, the hearts of ten rats/group were homogenized and MDA, SOD, GPX, and catalase (CAT) were determined by ELISA method. In other ten rats/group, left ventricle systolic and end diastolic pressures (LVSP and LVEDP) and contractility indices (LVDP and +dp/dt max) and relaxation velocity (−dp/dt max) were recorded. The coronary outflow was collected. Short- and long-term strength exercise increased heart weight and heart/BW ratio (P < 0.05). In the 3-month exercise group, basal heart rate decreased (P < 0.05). LVEDP did not change but LVDP, +dp/dt max, −dp/dt max, and coronary flow significantly increased in both exercise groups (P < 0.05). None of MDA or SOD, GPX, and CAT significantly changed. The results showed that sub-maximal chronic strength exercise improves heart efficiency without increase in oxidative stress index or decrease in antioxidant defense capacity. These imply that long-time strength exercise up to this intensity is safe for cardiac health.







Similar content being viewed by others
References
Ascensao A, Ferreira R, Magalhaes J (2007) Exercise-induced cardio protection: biochemical, morphological and functional evidence in whole tissue and isolated mitochondria. Int J Cardiol 117(1):16–30
Bank AJ, Shammas RA, Mullen K, Chuang PP (1998) Effects of short-term forearm exercise training on resistance vessel endothelial function in normal subjects and patients with heart failure. J Card Fail 4:193–201
Barauna VG, Rosa KT, Irigoyen MC, de Oliveira EM (2007) Effects of resistance training on ventricular function and hypertrophy in a rat model. Clin Med Res 5(2):114–120
Brown DA, Jew KN, Sparagna GC, Musch TI, Moore RL (2003) Exercise training preserves coronary flow and reduces infarct size after ischemia-reperfusion in rat heart. J Appl Physiol 95(6):2510–2518
Castaneda C, Layne JE, Munoz-Orians L, Gordon PL, Walsmith J, Foldvari M, Roubenoff R, Tucker KL, Nelson ME (2002) A randomized controlled trial of resistance exercise training to improve glycemic control in older adults with type 2 diabetes. Diabetes Care 25:2335–2341
Clanton TL (2007) Hypoxia-induced reactive oxygen species formation in skeletal muscle. J Appl Phsyiol 102:2379–2388
Clarkson PA, Thomson HD (2000) Antioxidants: what role do they play in physical activity and health? Am J Clin Nutr 72:637S–646S
Cooper CE, Vollaard NB, Choueiri T, Wilson MT (2002) Exercise, free radicals and oxidative stress. Biochem Soc Trans 30(2):280–285
Cornelissen VA, Fagard RH (2005) Effect of resistance training on resting blood pressure: a meta-analysis of randomized controlled trials. J Hypertens 23:251–259
Duncker DJ, Bache RJ (2008) Regulation of coronary blood flow during exercise. Physiol Rev 88:1009–1086
Fagard RH (1996) Athlete's heart: a meta-analysis of the echocardiographic experience. Int J Sports Med 17(suppl 3):S140–S144
Hambrecht R, Gielen S, Linke A, Fiehn E et al (2000) Effects of exercise training on left ventricular function and peripheral resistance in patients with chronic heart failure. JAMA 283(23):3095–3101
Lambert JA, Yang CS (2003) Mechanisms of cancer prevention by tea constituents. J Nutr 133(10):3262S–3267S
Leeuwenburgh C, Hollander J, Leichtweis S, Griffiths M, Gore M, Ji LL (1997) Adaptations of glutathione antioxidant system to endurance training are tissue and muscle fibre specific. Am J Physiol 272:R363–R369
Levick JR (2010) An introduction to cardiovascular physiology. In: Initiation and nervous control of heart beat, vol Chapter 4, 5th edn. Hodder Arnold Co, London, pp 51–65
Michailidis Y, Jamurtas AZ, Kickolaidis MG, Fatouros IG, Koutedakis Y, Papssotiriou I, Kouretas D (2007) Sampling time is crucial for measurement of aerobic exercise-induced oxidative stress. Med Sci Sports Exer 39(7):1107–1113
Moran M, Delgsdo J, Gonzalez B, Manso R, Megias A (2004) Responses of rat myocardial antioxidant defenses and heat shock protein HSP72 induced by 12 and 24-week treadmill training. Acta Physiol Scand 180:157–166
Pluim B, Zwinderman AH, Van der Laarse A, Vander Wall EE (2000) The athlete's heart: meta-analysis of cardiac structure and function. Circulation 101:336–344
Powers SK, Demirel HA, Vincent HK, Coombes JS (1998) Exercise training improves myocardial tolerance to in vivo ischemia-reperfusion in the rat. Am Physiol Soc 44:R1468–R1477
Powers SK, Quindry JC, Kavazis AN (2008) Exercise-induced cardio protection against myocardial ischemia-reperfusion injury. Free Radic Biol Med 44(2):193–201
Rakobowchuk M, McGowan CL, de Groot PC, Hartman JW (2005) Endothelial function of young healthy males following whole body resistance training. J Appl Physiol 98:2185–2190
Siu PM, Bryner RW, Martyn JK, Always E (2004) Apoptotic adaptations from exercise training in skeletal and cardiac muscles. FASEB J 18(10):1150–1152
Skrzypiec-Spring M, Grotthus B, Szelag A, Schulz R (2007) Isolated heart perfusion according to Langendorff–Still viable in the new millennium. J Pharmacol Toxicol Meth 55:113–126
Somani SM, Frank S, Rybak LP (1995) Responses of antioxidant system to acute and trained exercise in rat heart subcellular fraction. Pharmacol Biochem Behav 51(4):627–634
Spirito P, Pelliccia A, Proschan MA, Granata M, Spataro A, Bellone P, Caselli G, Biffi A, Vecchio C, Maron BJ (1994) Morphology of the “athlete’s heart” assessed by echocardiography in 947 elite athletes representing 27 sports. Am J Cardiol 74:802–806
Steinberg JG, Delliaux S, Jammes Y (2006) Reliability of different blood indices to explore the oxidative stress in response to maximal cycling and static exercises. Clin Phyiol Func Imaging 26(2):106–112
Suman OE, Hasten D, Turner MJ, Rinder MR, Spina RJ, Ehsani AA (2000) Enhanced inotropic response to dobutamine in strength-trained subjects with left ventricular hypertrophy. J Appl Physiol 88:534–539
Trueblood N, Ramasamy R, Wang LF, Schaefer A (2000) Niacin protects the isolated heart from ischemia-reperfusion injury. Am J Physiol Heart Circ Physiol 279(2):H767–H771
Williams MA, Haskell WL, Ades PA, Amsterdam EA, Bittner V, Franklin BA, Gulanick M, Laing ST, Stewart KJ (2007) Resistance exercise in individuals with and without cardiovascular disease: 2007 update: a scientific statement from the American Heart Association Council on Clinical Cardiology and Council on Nutrition, Physical Activity and Metabolism. Circulation 116:572–584
Zembron-Lacny L, Ostapiuk J, Slowinska L, Witkowski K, Szyska K (2008) Pro-antioxidant ratio in healthy men exposed to muscle-damaging resistance exercise. J Physiol Biochem 64(1):27–35
Acknowledgment
This work was supported by grants from Physiology Research Centers of Kerman University of Medical Sciences, Kerman, Iran and Drug Applied Research center of Tabriz University of Medical Sciences, Tabriz, Iran, under a joint research project.
Conflict of interest
The authors declare that there is no conflict of interest in this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ahmadiasl, N., Najafipour, H., Soufi, F.G. et al. Effect of short- and long-term strength exercise on cardiac oxidative stress and performance in rat. J Physiol Biochem 68, 121–128 (2012). https://doi.org/10.1007/s13105-011-0125-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13105-011-0125-z