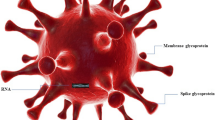
Abstract
The COVID-19 outbreak encouraged the global development of efficient prevention, diagnosis, and treatment while posing a major threat to the global economy and public health. It is discovered that due to the benefits of nanomaterials, including their ease of preparation, low cost, and ease of modification, there is a new possibility to create tactics against SARS-CoV-2. Studies on the economic viability, long-term safety, biocompatibility, and environmental effects of nanomaterials are still scarce, nevertheless. Currently, the primary methods for combating the SARS-CoV-2 virus are the creation of potent disinfectants, quick and precise diagnostic instruments, and nanovaccines. In order to give useful data reference for the development of nanomaterials against SARS-CoV-2 virus, this review first provides a brief introduction to the structure and infection mode of the SARS-CoV-2 virus. It next assesses the use of nanomaterials themselves. In order to establish a comprehensive cooperative battle against SARS-CoV-2 virus, it concludes by focusing on the application of nanomaterials for the prevention, diagnosis, and treatment of SARS-CoV-2 virus as well as their combined application with existing technologies.






Similar content being viewed by others
Data Availability
Not applicable.
Code Availability
Not applicable.
References
Chan, J. F., Yuan, S., Kok, K. H., et al. (2020). A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: A study of a family cluster. Lancet, 395(10223), 514–523.
Paraskevis, D., Kostaki, E. G., Magiorkinis, G., et al. (2020). Full-genome evolutionary analysis of the novel corona virus (2019-nCoV) rejects the hypothesis of emergence as a result of a recent recombination event. Infection, Genetics and Evolution, 79, 104212.
Li, J., Gong, X., Wang, Z., et al. (2020). Clinical features of familial clustering in patients infected with 2019 novel coronavirus in Wuhan, China. Virus Research, 286, 198043.
Zhou, P., Yang, X. L., Wang, X. G., et al. (2020). Addendum: A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature, 588(7836), E6.
Channappanavar, R., & Perlman, S. (2017). Pathogenic human coronavirus infections: Causes and consequences of cytokine storm and immunopathology. Seminars in Immunopathology, 39(5), 529–539.
Decaro, N., Mari, V., Elia, G., et al. (2010). Recombinant canine coronaviruses in dogs, Europe. Emerging Infectious Diseases, 16(1), 41–47.
Chauhan, D. S., Prasad, R., Srivastava, R., et al. (2020). Comprehensive review on current interventions, diagnostics, and nanotechnology perspectives against SARS-CoV-2. Bioconjugate Chemistry, 31(9), 2021–2045.
Talebian, S., Wallace, G. G., Schroeder, A., et al. (2020). Nanotechnology-based disinfectants and sensors for SARS-CoV-2. Nature Nanotechnology, 15(8), 618–621.
Weiss, C., Carriere, M., Fusco, L., et al. (2020). Toward nanotechnology-enabled approaches against the COVID-19 pandemic. ACS Nano, 14(6), 6383–6406.
Kilic, T., Weissleder, R., & Lee, H. (2020). Molecular and immunological diagnostic tests of COVID-19: Current status and challenges. iScience, 23(8), 101406.
Orooji, Y., Sohrabi, H., Hemmat, N., et al. (2021). An overview on SARS-CoV-2 (COVID-19) and other human coronaviruses and their detection capability via amplification assay, chemical sensing, biosensing, immunosensing, and clinical assays. Nano-Micro Letters, 13(1), 18.
Younis, S., Taj, A., Zia, R., et al. (2020). Nanosensors for the detection of viruses. In Nanosensors for Smart Cities (pp. 327–338).
Shetti, N. P., Mishra, A., Bukkitgar, S. D., et al. (2021). Conventional and nanotechnology-based sensing methods for SARS coronavirus (2019-Ncov). ACS Applied Bio Materials, 4, 1178.
Zehbe, I., Hacker, G. W., Su, H., et al. (1997). Sensitive in situ hybridization with catalyzed reporter deposition, streptavidin-nanogold, and silver acetate autometallography: Detection of single-copy human papillomavirus. The American Journal of Pathology, 150(5), 1553–1561.
Hasöksüz, M., Kilic, S., & Sarac, F. (2020). Coronaviruses and SARS-CoV-2. Turkish. Journal of Medical Sciences, 50(SI-1), 549–556.
Lu, S., Ye, Q., Singh, D., et al. (2021). The SARS-CoV-2 nucleocapsid phosphoprotein forms mutually exclusive condensates with RNA and the membrane-associated M protein. Nature Communications, 12(1), 502.
Coutard, B., Valle, C., de Lamballerie, X., et al. (2020). The spike glycoprotein of the new coronavirus 2019-nCoV contains a furin-like cleavage site absent in CoV of the same clade. Antiviral Research, 176, 104742.
Kang, S., Yang, M., Hong, Z., et al. (2020). Crystal structure of SARS-CoV-2 nucleocapsid protein RNA binding domain reveals potential unique drug targeting sites. Acta Pharmaceutica Sinica B, 10(7), 1228–1238.
Astuti, I., & Ysrafil. (2020). Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2): An overview of viral structure and host response. Diabetes and Metabolic Syndrome: Clinical Research and Reviews, 14(4), 407–412.
Budhraja, A., Pandey, S., Kannan, S., et al. (2021). The polybasic insert, the RBD of the SARS-CoV-2 spike protein, and the feline coronavirus - Evolved or yet to evolve. Biochemistry and Biophysics Reports, 25, 100907.
Schoeman, D., & Fielding, B. C. (2019). Coronavirus envelope protein: Current knowledge. Virology Journal, 16(1), 69.
Scanlon, V. C., & Sanders, T. (2018). Essentials of anatomy and physiology (pp. 80–112). FA Davis.
Al-Halifa, S., Gauthier, L., Arpin, D., et al. (2019). Nanoparticle-based vaccines against respiratory viruses. Frontiers in Immunology, 10, 22.
Heinrich, M. A., Martina, B., & Prakash, J. (2020). Nanomedicine strategies to target coronavirus. Nano Today, 35, 100961.
Izaguirre, G. (2019). The proteolytic regulation of virus cell entry by furin and other proprotein convertases. Viruses, 11(9), 837.
Kuba, K., Imai, Y., Ohto-Nakanishi, T., et al. (2010). Trilogy of Ace2: A peptidase in the renin-angiotensin system, a SARS receptor, and a partner for amino acid transporters. Pharmacology & Therapeutics, 128(1), 119–128.
Zhang, Y., & Tang, L. V. (2020). Overview of targets and potential drugs of SARS-CoV-2 according to the viral replication. Journal of Proteome Research, 20(1), 49–59.
Malik, Y. A. (2020). Properties of coronavirus and SARS-CoV-2. The Malaysian Journal of Pathology, 42, 3–11.
V'kovski, P., Kratzel, A., Steiner, S., et al. (2021). Coronavirus biology and replication: Implications for SARS-CoV-2. National Review, 19, 155–170.
Ueffing, M., Bayyoud, T., Schindler, M., et al. (2020). Basic principles of replication and immunology of SARS-CoV-2. Ophthalmologe, 117(7), 609–614.
Guo, Y. R., Cao, Q. D., Hong, Z. S., et al. (2020). The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak - An update on the status. Military Medical Research, 7(1), 1–10.
Salata, O. V. (2004). Applications of nanoparticles in biology and medicine. Journal of Nanobiotechnology, 2, 3.
Abdelbasir, S. M., McCourt, K. M., Lee, C. M., et al. (2020). Waste-derived nanoparticles: Synthesis approaches, environmental applications, and sustainability considerations. Frontiers in Chemistry, 8, 782.
Kanwar, R., Rathee, J., Salunke, D. B., et al. (2019). Green nanotechnology-driven drug delivery assemblies. ACS Omega, 4, 8804–8815.
Ying, S., Guan, Z., Ofoegbu, P. C., et al. (2022). Green synthesis of nanoparticles: Current developments and limitations. Environmental Technology and Innovation, 26, 102336.
Miao, Z., Shastri, Y., Grift, T. E., et al. (2012). Lignocellulosic biomass feedstock transportation alternatives, logistics, equipment configurations, and modeling. Biofuels, Bioproducts and Biorefining, 6(3), 351–362.
Zhang, S., Malik, S., Ali, N., et al. (2022). Covalent and Non-covalent Functionalized Nanomaterials for Environmental Restoration. Top Curr Chem (Cham), 380(5), 44.
Karlsson, H. L., Gustafsson, J., Cronholm, P., et al. (2009). Size-dependent toxicity of metal oxide particles--a comparison between nano- and micrometer size. Toxicology Letters, 188(2), 112–118.
Farnoud, A. M., & Nazemidashtarjandi, S. (2019). Emerging investigator series: Interactions of engineered nanomaterials with the cell plasma membrane; what have we learned from membrane models? Environmental Science: Nano, 6, 13–40.
Sravan Bollu, V., & Soren, G. (2016). Genotoxic and histopathological evaluation of zinc oxide nanorods in vivo in Swiss albino mice. Journal of Evolution of Medical and Dental Sciences, 5, 6186–6192.
Donia, D. T., & Carbone, M. (2018). Fate of the Nanoparticles in environmental cycles. International journal of Environmental Science and Technology, 16, 583–600.
Wamucho, A., Heffley, A., & Tsyusko, O. V. (2020). Epigenetic effects induced by silver nanoparticles in caenorhabditis elegans after multigenerational exposure. Science of the Total Environment, 725, 138523.
Starnes, D. L., Lichtenberg, S. S., Unrine, J. M., et al. (2016). Distinct transcriptomic responses of caenorhabditis elegans to pristine and sulfidized silver nanoparticles. Environmental Pollution, 213, 314–321.
Zhang, Y., Gu, A. Z., Xie, S., et al. (2018). Nano-metal oxides induce antimicrobial resistance via radical-mediated mutagenesis. Environment International, 121, 1162–1171.
Subhan, M. A., & Subhan, T. (2022). Safety and global regulations for application of nanomaterials. In Nanomaterials Recycling; Micro and Nano Technologies (pp. 83–107). Elsevier.
Food and Drug Administration; Office of the Commissioner; Office of Policy, L.I.A.; Office of Policy. (2014). Considering whether an FDA-regulated product involves the application of nanotechnology. Food and Drug Administration: Silver Spring.
Yang, Y., & Westerhoff, P. (2014). Presence in, and release of, nanomaterials from consumer products. Advances in Experimental Medicine and Biology, 811, 1–17.
Malakar, A., Kanel, S. R., Ray, C., et al. (2021). Nanomaterials in the environment, human exposure pathway, and health effects: A review. Science of the Total Environment, 759, 143470.
Donaldson, K., Tran, L., Jimenez, L. A., et al. (2005). Combustion-derived nanoparticles: A review of their toxicology following inhalation exposure. Particle and Fibre Toxicology, 2, 10.
Solanki, R., Shankar, A., Modi, U., et al. (2023). New insights from nanotechnology in SARS-CoV-2 detection, treatment strategy, and prevention. Materials Today Chemistry, 29, 101478.
El-Atab, N., Mishra, R. B., & Hussain, M. M. (2021). Toward nanotechnology-enabled face masks against SARS-CoV-2 and pandemic respiratory diseases. Nanotechnology, 33(6).
Valenzuela-Fernández, A., Cabrera-Rodriguez, R., Ciuffreda, L., et al. (2022). Nanomaterials to combat SARS-CoV-2: Strategies to prevent, diagnose and treat COVID-19. Frontiers in Bioengineering and Biotechnology, 10, 1052436.
Moabelo, K. L., Martin, D. R., Fadaka, A. O., et al. (2021). Nanotechnology-based strategies for effective and rapid detection of SARS-CoV-2. Materials (Basel), 14(24), 7851.
Patra, S., Kerry, R. G., Maurya, G. K., et al. (2020). Emerging molecular prospective of SARS-CoV-2: Feasible nanotechnology based detection and inhibition. Frontiers in Microbiology, 11, 2098.
Moitra, P., Alafeef, M., Dighe, K., et al. (2020). Selective naked-eye detection of SARS-CoV-2 mediated by N gene targeted antisense oligonucleotide capped plasmonic nanoparticles. ACS Nano, 14(6), 7617–7627.
Zhu, X., Wang, X., Han, L., et al. (2020). Multiplex reverse transcription loop-mediated isothermal amplification combined with nanoparticle-based lateral flow biosensor for the diagnosis of COVID-19. Biosensors & Bioelectronics, 166, 112437.
Qiu, G., Gai, Z., Tao, Y., et al. (2020). Dual-functional plasmonic photothermal biosensors for highly accurate severe acute respiratory syndrome coronavirus 2 detection. ACS Nano, 14(5), 5268–5277.
Huang, X., Kon, E., Han, X., et al. (2022). Nanotechnology-based strategies against SARS-CoV-2 variants. Nature Nanotechnology, 17(10), 1027–1037.
Li, S., Liu, X., Liu, G., et al. (2023). Biomimetic nanotechnology for SARS-CoV-2 treatment. Viruses, 15(3), 596.
Tang, Z., Zhang, X., Shu, Y., et al. (2021). Insights from nanotechnology in COVID-19 treatment. Nano Today, 36, 101019.
Albaz, A. A., Rafeeq, M. M., Sain, Z. M., et al. (2021). Nanotechnology-based approaches in the fight against SARS-CoV-2. AIMS Microbiology, 7(4), 368–398.
Khan, M. M., Goh, Y. W., Ahmad, N., et al. (2022). Understanding and combating COVID-19 using the biology and chemistry of SARS-CoV-2. Bioprocess and Biosystems Engineering, 45(11), 1753–1769.
Huang, L., Rong, Y., Pan, Q., et al. (2021). SARS-CoV-2 vaccine research and development: Conventional vaccines and biomimetic nanotechnology strategies. Asian Journal of Pharmaceutical Sciences, 16(2), 136–146.
Chauhan, G., Madou, M. J., Kalra, S., et al. (2020). Nanotechnology for COVID-19: Therapeutics and vaccine research. ACS Nano, 14(7), 7760–7782.
Milane, L., & Amiji, M. (2021). Clinical approval of nanotechnology-based SARS-CoV-2 mRNA vaccines: Impact on translational nanomedicine. Drug Delivery and Translational Research, 11(4), 1309–1315.
Khurana, A., Allawadhi, P., Khurana, I., et al. (2021). Role of nanotechnology behind the success of mRNA vaccines for COVID-19. Nano Today, 38, 101142.
Xu, C., Lei, C., Hosseinpour, S., et al. (2022). Nanotechnology for the management of COVID-19 during the pandemic and in the post-pandemic era. National Science Review, 9(10), nwac124.
Chen, Y. N., Hsueh, Y. H., Hsieh, C. T., et al. (2016). Antiviral activity of graphene-silver nanocomposites against non-enveloped and enveloped viruses. International Journal of Environmental Research and Public Health, 13(4), 430.
Ye, S., Shao, K., Li, Z., et al. (2015). Antiviral activity of graphene oxide: How sharp edged structure and charge matter. ACS Applied Materials & Interfaces, 7(38), 21571–21579.
Hosseini, M., Behzadinasab, S., Benmamoun, Z., et al. (2021). The viability of SARS-CoV-2 on solid surfaces. Current Opinion in Colloid & Interface Science, 55, 101481.
Doremalen, N. V., Bushmaker, T., Morris, D. H., et al. (2020). Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. The New England Journal of Medicine, 382(16), 1564–1567.
Nikaeen, G., Abbaszadeh, S., & Yousefinejad, S. (2020). Application of nanomaterials in treatment, anti-infection and detection of coronaviruses. Nanomedicine(Lond), 15(15), 1501–1512.
Jones, G. W., Monopoli, M. P., Campagnolo, L., et al. (2020). No small matter: A perspective on nanotechnology-enabled solutions to fight COVID-19. Nanomedicine (London, England), 15(24), 2411–2427.
Abo-Zeid, Y., Ismail, N. S. M., McLean, G. R., et al. (2020). A molecular docking study repurposes FDA approved iron oxide nanoparticles to treat and control COVID-19 infection. European Journal of Pharmaceutical Sciences, 153, 105465.
Chaudhary, V., Royal, A., Chavali, M., et al. (2021). Advancements in research and development to combat COVID-19 using nanotechnology. Nanotechnology for Environmental Engineering, 6(1), 1–15.
Anand, U., Jakhmola, S., Indari, O., et al. (2021). Potential therapeutic targets and vaccine development for SARS-CoV-2/COVID-19 pandemic management: A review on the recent update. Front Immunol, 12, 658519.
Nakamura, S., Sato, M., Sato, Y., et al. (2019). Synthesis and application of silver nanoparticles (Ag NPs) for the prevention of infection in healthcare workers. International Journal of Molecular Sciences, 20, 3620.
Kalantar-Zadeh, K., Ward, S. A., Kalantar-Zadeh, K., et al. (2020). Considering the effects of microbiome and diet on SARS-CoV-2 infection: Nanotechnology roles. ACS Nano, 14(5), 5179–5182.
Sportelli, M. C., Izzi, M., Kukushkina, E. A., et al. (2020). Can nanotechnology and materials science help the fight against SARS-CoV-2? Nanomaterials, 10, 802.
Purwar, T., Dey, S., Al-Kayyali, O. Z. A., et al. (2022). Electrostatic spray disinfection using nano-engineered solution on frequently touched surfaces in indoor and outdoor environments. Int J Environ Res Public Health, 19(12), 7241.
Makvandi, P., Wang, C., Zare, E. N., et al. (2020). Metal-based nanomaterials in biomedical applications: Antimicrobial activity and cytotoxicity aspects. Advanced Functional Materials, 30(12), 1910021.1–1910021.40.
Rajendran, S., Mukherjee, A., Nguyen, T. A., et al. (2020). Nanotoxicity: Prevention and antibacterial applications of nanomaterials (pp. 107–141). Elsevier.
Wang, Y., Jin, Y., Chen, W., et al. (2019). Construction of nanomaterials with targeting phototherapy properties to inhibit resistant bacteria and biofilm infections. Chemical Engineering Journal, 358, 74–90.
Guelph researchers: Self-sterilizing nano-coating and no more coronavirus infected surfaces. In: Statnano. NBIC+. 2020.
Nickels, L. (2020). Antiviral boost for nanoparticles. Metal Powder Report, 75, 330–333.
Mahapatra, P. S., Chatterjee, S., Tiwari, M. K., et al. (2020). Surface treatments to enhance the functionality of PPEs. Transactions of the Indian National Academy of Engineering, 5(2), 333–336.
(2021). Personalized reusable face masks with smart nano‐assisted destruction of pathogens for covid‐19: a visionary road. Chemistry – A European Journal.
Hasanzadeh, A., Alamdaran, M., Ahmadi, S., et al. (2021). Nanotechnology against COVID-19: Immunization, diagnostic and therapeutic studies. Journal of Controlled Release, 336, 354–374.
Mallakpour, S., Azadi, E., & Hussain, C. M. (2021). The latest strategies in the fight against the COVID-19 pandemic: The role of metal and metal oxide nanoparticles. New Journal of Chemistry, 45(14), 6167–6179.
Ju, J. T. J., Boisvert, L. N., & Zuo, Y. Y. (2021). Face masks against COVID-19: Standards, efficacy, testing and decontamination methods. Advances in Colloid and Interface Science, 292, 102435.
Rasmi, Y., Saloua, K. S., Nemati, M., et al. (2021). Recent progress in nanotechnology for COVID-19 prevention, diagnostics and treatment. Nanomaterials, 11, 1788.
Ngonghala, C. N., Iboi, E. A., & Gumel A. B. (2020). Could masks curtail the post-lockdown resurgence of COVID-19 in the US? Math Biosci, 329, 108452.
Bueckert, M., Gupta, R., Gupta, A., et al. (2020). Infectivity of SARS-CoV-2 and other Coronaviruses on dry surfaces: Potential for Indirect Transmission. Materials (Basel), 13(22), 5211.
Behzadinasab, S., Chin, A., Hosseini, M., et al. (2020). A surface coating that rapidly inactivates SARS-CoV-2. ACS Applied Materials & Interfaces, 12(31), 34723–34727.
Matsuura, R., Lo, C. W., Wada, S., et al. (2021). SARS-CoV-2 Disinfection of Air and surface contamination by TiO2 photocatalyst-mediated damage to viral morphology, RNA, and protein. Viruses, 13(5), 942.
Micochova, P., Chadha, A., Hesseloj, T., et al. (2021). Rapid inactivation of SARS-CoV-2 by titanium dioxide surface coating. Wellcome Open Research, 6, 56.
Li, Y. H., Li, J., Liu, X. E., et al. (2005). Detection of the nucleocapsid protein of severe acute respiratory syndrome coronavirus in serum: Comparison with results of other viral markers. Journal of Virological Methods, 130(1-2), 45–50.
Yu-Cheng, S., Wei-En, H., & Lin, C. T. (2018). Review-field-effect transistor biosensing: Devices and clinical applications. ECS Journal of Solid State Science and Technology, 7(7), Q3196–Q3207.
Seo, G., Lee, G., Kim, M. J., et al. (2020). Rapid detection of COVID-19 causative virus (SARS-CoV-2) in human nasopharyngeal swab specimens using field-effect transistor-based biosensor. ACS Nano, 14(4), 5135–5142.
Mahari, S., Roberts, A., Shahdeo, D., et al. (2020). eCovSens-Ultrasensitive Novel In-House Built Printed Circuit Board Based Electrochemical Device for Rapid Detection of nCovid-19 antigen, a spike protein domain 1 of SARS-CoV-2. Cold Spring Harbor Laboratory.
Laderman, E. I., Whitworth, E., Dumaual, E., et al. (2008). Rapid, sensitive, and specific lateral-flow immunochromatographic point-of-care device for detection of herpes simplex virus type 2-specific immunoglobulin G antibodies in serum and whole blood. Clinical and Vaccine Immunology, 15(1), 159–163.
Nikbakht, H., Gill, P., Tabarraei, A., et al. (2014). Nanomolecular detection of human influenza virus type A using reverse transcription loop-mediated isothermal amplification assisted with rod-shaped gold nanoparticles. RSC Advances, 4(26), 13575–13580.
Rabiee, N., Bagherzadeh, M., Ghasemi, A., et al. (2020). Point-of-use rapid detection of SARS-CoV-2: Nanotechnology-enabled solutions for the COVID-19 pandemic. International Journal of Molecular Sciences, 21(14), 5126.
Somvanshi, S. B., Kharat, P. B., Saraf, T. S., et al. (2021). Multifunctional nano-magnetic particles assisted viral RNA-extraction protocol for potential detection of COVID-19. Materials Research Innovations, 25, 169–174.
Algar, W. R., Susumu, K., Delehanty, J. B., et al. (2011). Semiconductor quantum dots in bioanalysis: Crossing the valley of death. Analytical Chemistry, 83(23), 8826–8837.
Ashiba, H., Sugiyama, Y., Wang, X., et al. (2017). Detection of norovirus virus-like particles using a surface plasmon resonance-assisted fluoroimmunosensor optimized for quantum dot fluorescent labels. Biosensors & Bioelectronics, 93, 260–266.
Roh, C., & Jo, S. K. (2011). Quantitative and sensitive detection of SARS coronavirus nucleocapsid protein using quantum dots-conjugated RNA aptamer on chip. Journal of Chemical Technology and Biotechnology, 86(12), 1475–1479.
Georgakilas, V., Perman, J. A., Tucek, J., et al. (2015). Broad family of carbon nanoallotropes: Classification, chemistry, and applications of fullerenes, carbon dots, nanotubes, graphene, nanodiamonds, and combined superstructures. Chemical Reviews, 115(11), 4744–4822.
Lim, S. Y., Shen, W., & Gao, Z. (2015). Carbon quantum dots and their applications. Chemical Society Reviews, 44, 362–381.
Yeh, Y. T., Tang, Y., Sebastian, A., et al. (2016). Tunable and label-free virus enrichment for ultrasensitive virus detection using carbon nanotube arrays. Science Advances, 2(10), e1601026.
Carvalho, A. P. A., & Conte-Junior, C. A. (2021). Recent advances on nanomaterials to COVID-19 management: A systematic review on antiviral/virucidal agents and mechanisms of SARS-CoV-2 inhibition/inactivation. Global Challenges, 5(5), 2000115.
Galdiero, S., Falanga, A., Cantisani, M., et al. (2014). Silver nanoparticles as novel antibacterial and antiviral agents. In Handbook of Nanobiomedical Research: Fundamentals, Applications and Recent Developments: Vol. 1. Materials for Nanomedicine (pp. 565–594). World Scientific.
Cagno, V., Andreozzi, P., D’Alicarnasso, M., et al. (2018). Broad-spectrum non-toxic antiviral nanoparticles with a virucidal inhibition mechanism. Nature Materials, 17(2), 195–203.
Kim, J., Yeom, M., Lee, T., et al. (2020). Porous gold nanoparticles for attenuating infectivity of influenza A virus. Journal of Nanobiotechnology, 18(1), 54.
El-Gaffary, M., Bashandy, M. M., Ahmed, A. R., et al. (2019). Self-assembled gold nanoparticles for in-vitro inhibition of bovine viral diarrhea virus as surrogate model for HCV. Materials Research Express, 6(7), 075075.
Mehranfar, A., & Izadyar, M. (2020). Theoretical Design of functionalized gold nanoparticles as antiviral agents against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Journal of Physical Chemistry Letters, 11(24), 10284–10289.
Mahajan, S. D., Aalinkeel, R., Law, W. C., et al. (2012). Anti-HIV-1 nanotherapeutics: Promises and challenges for the future. International Journal of Nanomedicine, 7, 5301–5314.
Yazdi, M. H., Mahdavi, M., Faghfuri, E., et al. (2015). Th1 immune response induction by biogenic selenium nanoparticles in mice with breast cancer: Preliminary vaccine model. Iranian Journal of Biotechnology, 13(2), 1–9.
Christiansen, D., Earnest-Silveira, L., Grubor-Bauk, B., et al. (2019). Pre-clinical evaluation of a quadrivalent HCV VLP vaccine in pigs following microneedle delivery. Scientific Reports, 9(1), 9251.
Hodgins, B., Pillet, S., Landry, N., et al. (2019). A plant-derived VLP influenza vaccine elicits a balanced immune response even in very old mice with co-morbidities. PLoS One, 14(1), e0210009.
Mohsen, M. O., Speiser, D. E., Knuth, A., et al. (2020). Virus-like particles for vaccination against cancer. Wiley Interdisciplinary Reviews. Nanomedicine and Nanobiotechnology, 12(1), e1579.
Quan, F. S., Basak, S., Chu, K. B., et al. (2020). Progress in the development of virus-like particle vaccines against respiratory viruses. Expert Review of Vaccines, 19(1), 11–24.
Xu, R., Shi, M., Li, J., et al. (2020). Construction of SARS-CoV-2 virus-like particles by mammalian expression system. Frontiers in Bioengineering and Biotechnology, 8, 862.
LaTourette, P. C., II, Awasthi, S., Desmond, A., et al. (2020). Protection against herpes simplex virus type 2 infection in a neonatal murine model using a trivalent nucleoside-modified mRNA in lipid nanoparticle vaccine. Vaccine, 38(47), 7409–7413.
Mucker, E. M., Karmali, P. P., Vega, J., et al. (2020). Lipid nanoparticle formulation increases efficiency of DNA-vectored vaccines/immunoprophylaxis in animals including transchromosomic bovines. Scientific Reports, 10(1), 8764.
Miao, L., Li, L., Huang, Y., et al. (2019). Delivery of Mrna vaccines with heterocyclic lipids increases anti-tumor efficacy by sting-mediated immune cell activation. Nature Biotechnology, 37(10), 1174–1185.
Liu, L., Liu, Z., Chen, H., et al. (2020). Subunit nanovaccine with potent cellular and mucosal immunity for COVID-19. ACS Applied Bio Materials, 3(9), 5633–5638.
Pardi, N., Hogan, M. J., Porter, F. W., et al. (2018). mRNA vaccines - a new era in vaccinology. Nature Reviews Drug Discovery, 17(4), 261–279.
Smith, T. R., Patel, A., Ramos, S., et al. (2020). Immunogenicity of a DNA vaccine candidate for COVID-19. Nature Communications, 11(1), 1–13.
Zhang, N. N., Li, X. F., Deng, Y. Q., et al. (2020). A thermostable mRNA vaccine against COVID-19. Cell, 182(5), 1271–1283.
Tiram, G., Scomparin, A., Ofek, P., et al. (2014). Interfering cancer with polymeric siRNA nanomedicines. Journal of Biomedical Nanotechnology, 10(1), 50.
Cojocaru, F. D., Doru, B., Gardikiotis, I., et al. (2020). Nanomaterials designed for antiviral drug delivery transport across biological barriers. Pharmaceutics, 12(2), 171.
Amir, G., Navid, R., Sepideh, A., et al. (2018). Optical assays based on colloidal inorganic nanoparticles. Analyst, 143, 10.1039.
Avery, J. C., & Hoffmann, P. R. (2018). Selenium, selenoproteins, and immunity. Nutrients, 10(9), 1203.
Zhong, J., Xia, Y., Hua, L., et al. (2019). Functionalized selenium nanoparticles enhance the anti-EV71 activity of oseltamivir in human astrocytoma cell model. Artificial Cells, 47(1), 3485–3491.
Ahmadi, S., Rabiee, N., Bagherzadeh, M., et al. (2020). Stimulus-responsive sequential release systems for drug and gene delivery. Nano Today, 34, 100914.
Shakiba, S., Astete, C. E., Paudel, S., et al. (2020). Emerging investigator series: Polymeric nanocarriers for agricultural applications: Synthesis, characterization, and environmental and biological interactions. Environmental Science: Nano, 7(1), 37–67.
LaBauve, A. E., Rinker, T. E., Noureddine, A., et al. (2018). Lipid-coated mesoporous silica nanoparticles for the delivery of the ML336 antiviral to inhibit encephalitic alphavirus infection. Scientific Reports, 8(1), 13990.
Funding
This study was supported by National Natural Science Foundation of China (no. 81760750); Natural Science Foundation of Hebei Province (no. H2020208018, C2020208023); the Graduate Student Innovation Ability Training Project of Hebei University of Science and Technology (XJCXZZSS202307); Doctoral research fund project of Hebei University of Science and Technology (no. QD2023004).
Author information
Authors and Affiliations
Contributions
Yachan Feng draft the article, conduct literature retrieval, and provide the overall structure of the article. Haojie Zhang, Jiangtao Shao, Xiaolei Zhou, Yu Fu, and Chao Du modified the article and added some content. Yingze Wang and Xueling Guo revised the arrangement of the article to help improve the accuracy of the language. All authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics Approval
Not applicable.
Consent for Publication
Not applicable.
Informed Consent
All authors are aware of and agree to any action taken regarding the article.
Conflict of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Research Involving Humans and Animals Statement
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Feng, Y., Zhang, H., Shao, J. et al. Research Progress of Nanomaterials for Prevention, Diagnosis, and Treatment of SARS-CoV-2. BioNanoSci. (2024). https://doi.org/10.1007/s12668-024-01310-6
Accepted:
Published:
DOI: https://doi.org/10.1007/s12668-024-01310-6