Abstract
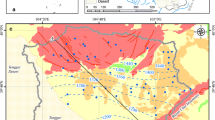
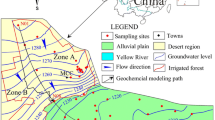
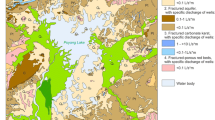
The forest ecosystem in the Maolan karst forest, southwest China is the only concentrated, intact, and relatively stable karst forest ecosystem which has survived in the area at the same latitude in the world, and is a valuable karst forest plant resource as well. Groundwater samples from Maolan karst forest were collected from wells and springs during summer; and concentrations of major ions and dissolved inorganic carbon (DIC) isotopic compositions were measured. The pH values range from 7.2 to 8.3 results from the dissolution of carbonate, HCO3 − is the dominant species of DIC in groundwater. Calcium and HCO3 −, followed by Mg2+ and SO4 2− dominate the chemical composition of major ions in the groundwaters. Groundwater samples have δ13C values in the range from −8.1‰ to −16.6‰, which are lower than that of the other karst city groundwaters in the southwest China. Combining δ13CDIC ratios with measurements of HCO3 − and pH clearly distinguishes the principal processes underlying the geochemical evolution of groundwater in Carboniferous carbonate aquifers, where processes can be both degradation of organic matters in the soil and the carbonate dissolution.



Similar content being viewed by others
References
Atekwana EA, Krishnamurthy RV (1998) Seasonal variations of dissolved inorganic carbon and δ13C of surface waters: application of a modified gas evolution technique. J Hydrol 205:265–278. doi:10.1016/S0022-1694(98)00080-8
Aucour AM, Sheppard SMF, Guyomar O, Wattelet J (1999) Use of δ13C to trace origin and cycling of inorganic carbon in the Rhône river system. Chem Geol 159:87–105. doi:10.1016/S0009-2541(99)00035-2
Cane G, Clark ID (1999) Tracing ground water recharge in an agricultural watershed with isotopes. Ground Water 37:133–139. doi:10.1111/j.1745-6584.1999.tb00966.x
Cerling TE, Solomon DK, Quade J (1991) On the isotopic composition of carbon in soil carbon dioxide. Geochim Cosmochim Acta 55:3403–3405. doi:10.1016/0016-7037(91)90498-T
Chapelle FH, Knobel LL (1985) Stable carbon isotopes of HCO3 − in the aquia aquifer, Maryland: evidence for an isotopically heavy source of CO2. Ground Water 23:592–599. doi:10.1111/j.1745-6584.1985.tb01507.x
Chou L, Garrels RM, Wollast R (1989) Comparative study of the kinetics an mechanisms of dissolution of carbonate minerals. Chem Geol 78:269–282. doi:10.1016/0009-2541(89)90063-6
Clark ID, Fritz P (1997) Environmental isotopes in hydrogeology. LEWIS Publishers, New York 328
Eisenlohr L, Meteva K, Gabrovsek F, Dreybrodt W (1999) The inhibiting action of intrinsic impurities in natural calcium carbonate minerals to their dissolution kinetics in aqueous H2O–CO2 solutions. Geochim Cosmochem Acta 63:989–1002. doi:10.1016/S0016-7037(98)00301-9
Fang JS, Barcelona MJ, Krishnamurthy RV, Atekwana EA (2000) Stable carbon isotope biogeochemistry of shallow sand aquifer contaminated with fuel hydrocarbons. Appl Geochem 15:157–169. doi:10.1016/S0883-2927(99)00037-2
Fritz P, Fontes JC, Frape SK, Louvat D, Michelot JM, Balderer W (1989) The isotope geochemistry of carbon in groundwater at Stripa. Geochim Cosmochem Acta 53:1765–1775. doi:10.1016/0016-7037(89)90297-4
Gonfiantini R, Zuppi GM (2003) Carbon isotope exchange rate of DIC in karst groundwater. Chem Geol 197:319–336. doi:10.1016/S0009-2541(02)00402-3
Grossman EI (1997) Stable carbon isotopes as indicators of microbial activity in aquifers. In: Hurst CJ, Knudson GR, McInerney MJ, Stetzenbach LD (eds) Manual of environmental microbiology. American Society for Microbiology, Washington, DC, pp 565–567
Han G, Liu C-Q (2004) Water geochemistry controlled by carbonate dissolution: a study of the river waters draining karst-dominated terrain, Guizhou Province, China. Chem Geol 204:1–21. doi:10.1016/j.chemgeo.2003.09.009
Landmeyer JE, Stone PA (1995) Radiocarbon and δ13C values related to ground-water recharge and mixing. Ground Water 33:227–234. doi:10.1111/j.1745-6584.1995.tb00277.x
Li S-L, Liu C-Q, Tao F-X, Lang Y-C, Han G (2005) Carbon biogeochemistry of ground water, Guiyang, southwest China. Ground Water 43:494–499
Li S-L, Liu C-Q, Lang Y-C, Tao F, Zhao Z, Zhou Z (2008) Stable carbon isotope biogeochemistry and anthropogenic impacts on karst ground water, Zunyi, southwest China. Aquat Geochem 14:211–221. doi:10.1007/s10498-008-9033-4
Liu Z, Li Q, Sun H, Wang J (2007) Seasonal, diurnal and storm-scale hydrochemical variations of typical epikarst spring in subtropical karst areas of SW China: soil CO2 and dilution effects. J Hydrol 337:207–223. doi:10.1016/j.jhydrol.2007.01.034
Murad AA, Krishnamurthy RV (2008) Factors controlling stable oxygen, hydrogen and carbon isotope ratios in regional groundwater of eastern United Arab Emirates (UAE). Hydrol Process 22:1922–1931. doi:10.1002/hyp.6776
Plummer LN, Wigley TML, Parkhurst DL (1978) The kinetics of calcite dissolution in CO2-water systems at 5 to 60°C and 0.0 to 1 atm CO2. Am J Sci 278:179–216
Rueedi J, Cronin AA, Taylor RG, Morris BL (2007) Tracing sources of carbon in urban groundwater using δ13CTDIC ratios. Environ Geol 52:541–557. doi:10.1007/s00254-006-0486-8
Stumm W, Morgan JJ (1981) Aquatic chemistry, 2nd edn. Wiley, New York
Telmer K, Veizer J (1999) Carbon fluxes, \( {\text{p}}_{{{\text{CO}}_{ 2}}} \) and substrate weathering in a large northern river basin, Canada: carbon isotope perspectives. Chem Geol 159:61–86. doi:10.1016/S0009-2541(99)00034-0
Zhou Z (1987) Scientific survey of the Maolan karst forest. Guihzou People’s Publishing House, Guiyang
Acknowledgments
This study was supported jointly by the Innovation Program of Chinese Academy of Sciences (No. KZCX-YW-306) and Ministry of Science and Technology of China (Grant No. 2006CB403206) as well as the Chinese National Natural Science Foundation (Nos. 40673010, 40721002).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Han, G., Tang, Y. & Wu, Q. Hydrogeochemistry and dissolved inorganic carbon isotopic composition on karst groundwater in Maolan, southwest China. Environ Earth Sci 60, 893–899 (2010). https://doi.org/10.1007/s12665-009-0226-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12665-009-0226-y