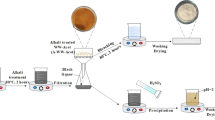
Abstract
Chitin–glucan complex, an essential organic ingredient in the inner layer of the fungal cell wall, was isolated from the stalk and cap of Pleurotus ostreatus mushroom using two methods. In the first, powder of water-insoluble part was treated with NaOH in 3 successive baths, whereas in the second method an additional step was performed using acetic acid for the effective removal of soluble proteins, free chitosan and minerals. The influence of the deproteinization process on the yield and purity of the chitin-glucan complex was assessed by variation of NaOH concentration, reaction time and number of baths.The biopolymer recovered by the first method (yield from stalk 49% of dry weight, Cri 63.19%) presented residual proteins, lipids and minerals (5.52%) whereas the purest copolymer (yield 41.1% and minerals 3.22% of dry weight, Cri 58.43%) was produced using the second method from the stalk; the latter was mostly considered as a valuable waste from an available bio-resource that can easily be cultivated. X-ray diffraction (XRD), thermal analyses, scanning electron microscope, energy-dispersive X-ray spectroscopy, infrared spectroscopy, Kjeldahl method and Biuret test were employed to prove purity and study the physicochemical properties. Furthermore, the isolated chitin–glucan complex was compared with the chitin extracted from Agaricus bisporus mushroom. Fungal chitin could constitute a viable alternative to commercial chitin with better performance in adsorption applications, and in this respect, the results revealed that the isolation of chitinous polysaccharide from P. ostreatus (41.1%) was higher than from most cultivated mushrooms (7–36.72%).
Graphic Abstract








Similar content being viewed by others
Abbreviations
- XRD:
-
X-ray diffraction
- TGA:
-
Thermogravimetric analysis
- DTG:
-
Derivative thermogravimetry
- DSC:
-
Differential scanning calorimetry
- SEM:
-
Scanning electron microscope
- EDS:
-
Energy dispersive spectrometer
- FTIR:
-
Fourier transform infrared spectrometry
- DA:
-
Degree of acetylation
- CrI:
-
Crystallinity index
- ChGCFm :
-
Chitin-glucan complex of the first method
- ChGCSm :
-
Chitin-glucan complex of the second method
- ChA.bisporus :
-
Chitin obtained from A. bisporus
- AIM:
-
Alkali insoluble material
- TN:
-
Total nitrogen
- CN:
-
Nitrogen of ChGCSm
- NPNNP:
-
Nitrogen of protein and non-protein components
- PC:
-
Protein content
References
Roca, C., Chagas, B., Farinha, I., Freitas, F., Mafra, L., Aguiar, F., Oliveira, R., Reis, M.A.M.: Production of yeast chitin–glucan complex from biodiesel industry byproduct. Process Biochem. 47, 1670–1675 (2012). https://doi.org/10.1016/j.procbio.2012.04.004
Bzducha-Wróbel, A., Kieliszek, M., Błażejak, S.: Chemical composition of the cell wall of probiotic and brewer’s yeast in response to cultivation medium with glycerol as a carbon source. Eur. Food Res. Technol. 237, 489–499 (2013). https://doi.org/10.1007/s00217-013-2016-8
Farinha, I., Duarte, P., Pimentel, A., Plotnikova, E., Chagas, B., Mafra, L., Grandfils, C., Freitas, F., Fortunato, E., Reis, M.A.M.: Chitin–glucan complex production by Komagataella pastoris: downstream optimization and product characterization. Carbohydr. Polym. 130, 455–464 (2015). https://doi.org/10.1016/j.carbpol.2015.05.034
Araújo, D., Ferreira, I.C., Torres, C.A., Neves, L., Freitas, F.: Chitinous polymers: extraction from fungal sources, characterization and processing towards value-added applications. J. Chem. Technol. Biotechnol. 95, 1277–1289 (2020). https://doi.org/10.1002/jctb.6325
Hong, Y., Ying, T.: Characterization of a chitin-glucan complex from the fruiting body of Termitomyces albuminosus (Berk.) Heim. Int. J. Biol. Macromol. 134, 131–138 (2019). https://doi.org/10.1016/j.ijbiomac.2019.04.198
Zelencova, L., Erdoǧan, S., Baran, T., Kaya, M.: Chitin extraction and chitosan production from Chilopoda (Scolopendra cingulata) with identification of physicochemical properties. Polym. Sci. Ser. A. 57, 437–444 (2015). https://doi.org/10.1134/S0965545X15040161
Abdel-Rahman, R.M., Hrdina, R., Abdel-Mohsen, A.M., Fouda, M.M.G., Soliman, A.Y., Mohamed, F.K., Mohsin, K., Pinto, T.D.: Chitin and chitosan from Brazilian Atlantic Coast: Isolation, characterization and antibacterial activity. Int. J. Biol. Macromol. 80, 107–120 (2015). https://doi.org/10.1016/j.ijbiomac.2015.06.027
Hassainia, A., Satha, H., Boufi, S.: Chitin from Agaricus bisporus: Extraction and characterization. Int. J. Biol. Macromol. 117, 1334–1342 (2018). https://doi.org/10.1016/j.ijbiomac.2017.11.172
Smiderle, F.R., Olsen, L.M., Carbonero, E.R., Baggio, C.H., Freitas, C.S., Marcon, R., Santos, A.R.S., Gorin, P.A.J., Iacomini, M.: Anti-inflammatory and analgesic properties in a rodent model of a (1→3), (1→6)-linked β-glucan isolated from Pleurotus pulmonarius. Eur. J. Pharmacol. (2008). https://doi.org/10.1016/j.ejphar.2008.08.028
Majtán, J., Kumar, P., Koller, J., Dragúńová, J., Gabriž, J.: Induction of Metalloproteinase 9 secretion from human keratinocytes by Pleuran (β-Glucan from Pleurotus ostreatus). Zeitschrift für Naturforschung C. 64, 597–600 (2009). https://doi.org/10.1515/znc-2009-7-820
Bulam, S., Üstün, N.Ş, Pekşen, A.: Evaluation of nutritional and medicinal values of edible wild and cultivated Pleurotus ostreatus. Turkish JAF Sci. Tech. 7, 2054 (2019). https://doi.org/10.24925/turjaf.v7i12.2054-2061.2730
Ospina Álvarez, S.P., Ramírez Cadavid, D.A., Escobar Sierra, D.M., Ossa Orozco, C.P., Rojas Vahos, D.F., Zapata Ocampo, P., Atehortúa, L.: Comparison of extraction methods of chitin from Ganoderma lucidum mushroom obtained in submerged culture. Biomed. Res. Int. 2014, 1–7 (2014). https://doi.org/10.1155/2014/169071
Liao, J., Huang, H.: Extraction of a novel fungal chitin from Hericium erinaceus residue using multistep mild procedures. Int. J. Biol. Macromol. 156, 1279–1286 (2020). https://doi.org/10.1016/j.ijbiomac.2019.11.165
Yen, M.T., Mau, J.L.: Preparation of fungal chitin and chitosan from shiitake stipes. Fungal Sci. 21(1), 2 (2006). https://doi.org/10.7099/FS.200612.0001
Zhou, S., Ma, F., Zhang, X., Zhang, J.: Carbohydrate changes during growth and fruiting in Pleurotus ostreatus. Fungal Biol. 120, 852–861 (2016). https://doi.org/10.1016/j.funbio.2016.03.007
Cherno, N., Osolina, S., Nikitina, A.: Chemical composition of agaricus bisporus and pleurotus ostreatus fruiting bodies and their morphological parts. Food Environ. Saf. J. 12(4) (2016). http://fia-old.usv.ro/fiajournal/index.php/FENS/article/view/180
Baeva, E., Bleha, R., Lavrova, E., Sushytskyi, L., Čopíková, J., Jablonsky, I., Klouček, P., Synytsya, A.: Polysaccharides from basidiocarps of cultivating mushroom Pleurotus ostreatus: isolation and structural characterization. Molecules 24, 2740 (2019). https://doi.org/10.3390/molecules24152740
Synytsya, A., Míčková, K., Synytsya, A., Jablonský, I., Spěváček, J., Erban, V., Kováříková, E., Čopíková, J.: Glucans from fruit bodies of cultivated mushrooms Pleurotus ostreatus and Pleurotus eryngii: structure and potential prebiotic activity. Carbohyd. Polym. 76, 548–556 (2009). https://doi.org/10.1016/j.carbpol.2008.11.021
Sietsma, J.H., Wessels, J.G.H.: Solubility of (1–3)- -d/(1–6)- -D-Glucan in fungal walls: Importance of presumed linkage between glucan and chitin. Microbiology 125, 209–212 (1981). https://doi.org/10.1099/00221287-125-1-209
Elsoud, M.M.A., El Kady, E.M.: Current trends in fungal biosynthesis of chitin and chitosan. Bull. Natl. Res. Centre 43(1), 59 (2019). https://doi.org/10.1186/s42269-019-0105-y
Mol, P.C., Vermeulen, C.A., Wessels, J.G.H.: Glucan-glucosaminoglycan linkages in fungal walls. Acta Bot. Neerlandica. 37, 17–21 (1988). https://doi.org/10.1111/j.1438-8677.1988.tb01577.x
Tolaimate, A., Rhazi, M., Alagui, A., Desbrieres, J., Rinaudo, M.: Valorization of waste products from fishing industry by production of the chitin and chitosan valorisation des dechets des industries de peche par production de la chitine et du chitosane. 9 (2008). https://hal.archives-ouvertes.fr/hal-00396614
Bradstreet, R.B.: Kjeldahl method for organic nitrogen. Anal. Chem. 26, 185–187 (1954). https://doi.org/10.1021/ac60085a028
Manzi, P., Marconi, S., Aguzzi, A., Pizzoferrato, L.: Commercial mushrooms: nutritional quality and effect of cooking. Food Chem. 84, 201–206 (2004). https://doi.org/10.1016/S0308-8146(03)00202-4
Braaksma, A., Schaap, D.J.: Protein analysis of the common mushroom Agaricus bisporus. Postharvest Biol. Technol. 7, 119–127 (1996). https://doi.org/10.1016/0925-5214(95)00034-8
Bautista, J., Jover, M., Gutierrez, J.F., Corpas, R., Cremades, O., Fontiveros, E., Iglesias, F., Vega, J.: Preparation of crayfish chitin by in situ lactic acid production. Process Biochem. 37, 229–234 (2001). https://doi.org/10.1016/S0032-9592(01)00202-3
Bianchi-Bosisio, A.: Proteins | Physiological samples. In: Encyclopedia of Analytical Science. p. 357–375. Elsevier (2005). https://doi.org/https://doi.org/10.1016/B0-12-369397-7/00494-5
Speiser, D.I., DeMartini, D.G., Oakley, T.H.: The shell-eyes of the chiton Acanthopleura granulata (Mollusca, Polyplacophora) use pheomelanin as a screening pigment. J. Nat. Hist. 48, 2899–2911 (2014). https://doi.org/10.1080/00222933.2014.959572
Bonatti, M., Karnopp, P., Soares, H.M., Furlan, S.A.: Evaluation of Pleurotus ostreatus and Pleurotus sajor-caju nutritional characteristics when cultivated in different lignocellulosic wastes. Food Chem. 88, 425–428 (2004). https://doi.org/10.1016/j.foodchem.2004.01.050
Bhatty, R.S., Sosulski, F.W., Wu, K.K.: Protein and nonprotein nitrogen contents of some oilseeds and peas. Can. J. Plant Sci. 53, 651–657 (1973). https://doi.org/10.4141/cjps73-129
Beran, K., Holan, Z., Baldrián, J.: The chitin-glucan complex in Saccharomyces cerevisiae: I IR and X-ray observations. Folia Microbiol. 17, 322–330 (1972). https://doi.org/10.1007/BF02884098
Kaya, M., Akata, I., Baran, T., Menteş, A.: Physicochemical properties of chitin and chitosan produced from medicinal fungus (Fomitopsis pinicola). Food Biophys. 10, 162–168 (2015). https://doi.org/10.1007/s11483-014-9378-8
Ivshin, V.P., Artamonova, S.D., Ivshina, T.N., Sharnina, F.F.: Methods for isolation of chitin-glucan complexes from higher fungi native biomass. Polym. Sci. Ser. B. 49, 305–310 (2007). https://doi.org/10.1134/S1560090407110097
Kaya, M., Halıcı, M.G., Duman, F., Erdoğan, S., Baran, T.: Characterisation of α-chitin extracted from a lichenised fungus species Xanthoria parietina. Nat. Prod. Res. 29, 1280–1284 (2015). https://doi.org/10.1080/14786419.2014.995651
Vetter, J.: Chitin content of cultivated mushrooms Agaricus bisporus, Pleurotus ostreatus and Lentinula edodes. Food Chem. 102, 6–9 (2007). https://doi.org/10.1016/j.foodchem.2006.01.037
Kaya, M., Baublys, V., Šatkauskienė, I., Akyuz, B., Bulut, E., Tubelytė, V.: First chitin extraction from Plumatella repens (Bryozoa) with comparison to chitins of insect and fungal origin. Int. J. Biol. Macromol. 79, 126–132 (2015). https://doi.org/10.1016/j.ijbiomac.2015.04.066
Kaya, M., Seyyar, O., Baran, T., Erdoğan, S., Kar, M.: A physicochemical characterization of fully acetylated chitin structure isolated from two spider species: with new surface morphology. Int. J. Biol. Macromol. 65, 553–558 (2014). https://doi.org/10.1016/j.ijbiomac.2014.02.010
Yen, M.-T., Mau, J.-L.: Selected physical properties of chitin prepared from shiitake stipes. LWT Food Sci. Technol. 40, 558–563 (2007). https://doi.org/10.1016/j.lwt.2005.10.008
Jones, M., Weiland, K., Kujundzic, M., Mautner, A., Bismarck, A., John, S.: Sustainable mycelium-derived chitinous thin films. Non-Serials, 4309–4317 (2019. https://search.informit.org/doi/https://doi.org/10.3316/informit.892879054072611
Paulino, A.T., Simionato, J.I., Garcia, J.C., Nozaki, J.: Characterization of chitosan and chitin produced from silkworm crysalides. Carbohydr. Polym. 64, 98–103 (2006). https://doi.org/10.1016/j.carbpol.2005.10.032
Kaya, M., Baran, T., Erdoğan, S., Menteş, A., Aşan Özüsağlam, M., Çakmak, Y.S.: Physicochemical comparison of chitin and chitosan obtained from larvae and adult Colorado potato beetle (Leptinotarsa decemlineata). Mater. Sci. Eng. C 45, 72–81 (2014). https://doi.org/10.1016/j.msec.2014.09.004
Kaya, M., Erdogan, S., Mol, A., Baran, T.: Comparison of chitin structures isolated from seven Orthoptera species. Int. J. Biol. Macromol. 72, 797–805 (2015). https://doi.org/10.1016/j.ijbiomac.2014.09.034
Sagheer, F.A.A., Al-Sughayer, M.A., Muslim, S., Elsabee, M.Z.: Extraction and characterization of chitin and chitosan from marine sources in Arabian Gulf. Carbohydr. Polym. 77, 410–419 (2009). https://doi.org/10.1016/j.carbpol.2009.01.032
Kaya, M., Baran, T., Mentes, A., Asaroglu, M., Sezen, G., Tozak, K.O.: Extraction and characterization of α-chitin and chitosan from six different aquatic invertebrates. Food Biophys. 9, 145–157 (2014). https://doi.org/10.1007/s11483-013-9327-y
Erdogan, S., Kaya, M., Akata, I.: Chitin extraction and chitosan production from cell wall of two mushroom species (Lactarius vellereus and Phyllophora ribis). Introduced to proceedings of the 6th international advances in applied physics and materials science congress & exhibition: (apmas 2016), İstanbul, Turkey (2017). https://doi.org/https://doi.org/10.1063/1.4975427
Kaya, M., Mujtaba, M., Ehrlich, H., Salaberria, A.M., Baran, T., Amemiya, C.T., Galli, R., Akyuz, L., Sargin, I., Labidi, J.: On chemistry of γ-chitin. Carbohydr. Polym. 176, 177–186 (2017). https://doi.org/10.1016/j.carbpol.2017.08.076
Yen, M.-T., Mau, J.-L.: Physico-chemical characterization of fungal chitosan from shiitake stipes. LWT Food Sci. Technol. 40, 472–479 (2007). https://doi.org/10.1016/j.lwt.2006.01.002
Ifuku, S., Nomura, R., Morimoto, M., Saimoto, H.: Preparation of chitin nanofibers from mushrooms. Materials. 4, 1417–1425 (2011). https://doi.org/10.3390/ma4081417
Nasrazadani, S., Hassani, S.: Modern analytical techniques in failure analysis of aerospace, chemical, and oil and gas industries. In: Makhlouf, A., Aliofkhazraei, M. (eds.), Handbook of Materials Failure Analysis with Case Studies from the Oil and Gas Industry. Elsevier, New York, pp. 39–54 (2016). https://doi.org/https://doi.org/10.1016/B978-0-08-100117-2.00010-8
Wolfgong, W.J.: Chemical analysis techniques for failure analysis. In: Makhlouf, A., Aliofkhazraei, M. (eds.), Handbook of Materials Failure Analysis with Case Studies from the Aerospace and Automotive Industries. Elsevier, New York, pp. 309–338 (2016). doi:https://doi.org/10.1016/B978-0-12-800950-5.00014-4
Mehranian, M., Pourabad, R.F., Bashir, N.S., Taieban, S.: Physicochemical characterization of chitin from the Mediterranean flour moth, Ephestia kuehniella Zeller (Lepidoptera: Pyralidae). J. Macromol. Sci. Part A 54, 720–726 (2017). https://doi.org/10.1080/10601325.2017.1332461
Ablouh, E.-H., Jalal, R., Rhazi, M., Taourirte, M.: Surface modification of α-chitin using an acidic treatment followed by ultrasonication: measurements of their sorption properties. Int. J. Biol. Macromol. 151, 492–498 (2020). https://doi.org/10.1016/j.ijbiomac.2020.02.204
Knidri, E.L., Dahmani, J., Addaou, A., Laajeb, A., Lahsini, A.: Rapid and efficient extraction of chitin and chitosan for scale-up production: effect of process parameters on deacetylation degree and molecular weight. Int. J. Biol. Macromol. 139, 1092–1102 (2019). https://doi.org/10.1016/j.ijbiomac.2019.08.079
El Knidri, H., El Khalfaouy, R., Laajeb, A., Addaou, A., Lahsini, A.: Eco-friendly extraction and characterization of chitin and chitosan from the shrimp shell waste via microwave irradiation. Process Saf. Environ. Prot. 104, 395–405 (2016). https://doi.org/10.1016/j.psep.2016.09.020
Pestov, A.V., Drachuk, S.V., Koryakova, O.V., Yatluk, Y.G.: Isolation and characterization of chitin-glucan complexes from the mycothallus of fungi belonging to Russula genus. Chem. Sustain. Dev. 17, 281–287 (2009)
Draczynski, Z.: Honeybee corpses as an available source of chitin. J. Appl. Polym. Sci. 109, 1974–1981 (2008). https://doi.org/10.1002/app.28356
Chaussard, G., Domard, A.: New aspects of the extraction of chitin from squid pens. Biomacromol 5, 559–564 (2004). https://doi.org/10.1021/bm034401t
Focher, B., Naggi, A., Torri, G., Cosani, A., Terbojevich, M.: Structural differences between chitin polymorphs and their precipitates from solutions—evidence from CP-MAS 13C-NMR FT-IR and FT-Raman spectroscopy. Carbohydr. Polym. 17(2), 97–102 (1992). https://doi.org/10.1016/0144-8617(92)90101-U
Di Mario, F., Rapanà, P., Tomati, U., Galli, E.: Chitin and chitosan from Basidiomycetes. Int. J. Biol. Macromol. 43, 8–12 (2008). https://doi.org/10.1016/j.ijbiomac.2007.10.005
Wu, T., Zivanovic, S., Draughon, F.A., Sams, C.E.: Chitin and chitosan value-added products from mushroom waste. J. Agric. Food Chem. 52, 7905–7910 (2004). https://doi.org/10.1021/jf0492565
Cárdenas, G., Cabrera, G., Taboada, E., Miranda, S.P.: Chitin characterization by SEM, FTIR, XRD, and 13 C cross polarization/mass angle spinning NMR: Chitin characterization. J. Appl. Polym. Sci. 93, 1876–1885 (2004). https://doi.org/10.1002/app.20647
Mesa Ospina, N., Ospina Alvarez, S.P., Escobar Sierra, D.M., Rojas Vahos, D.F., Zapata Ocampo, P.A., Ossa Orozco, C.P.: Isolation of chitosan from Ganoderma lucidum mushroom for biomedical applications. J. Mater. Sci. 26, 135 (2015). https://doi.org/10.1007/s10856-015-5461-z
Synowiecki, J., Al-Khateeb, N.A.: Production, properties, and some new applications of chitin and its derivatives. Crit. Rev. Food Sci. Nutr. 43, 145–171 (2003). https://doi.org/10.1080/10408690390826473
Acknowledgements
The authors gratefully acknowledge (la Direction Générale de la Recherche Scientifque et du Développement Technologique, Algerie) DGRSDT for their support in this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors disclose that there are no financial and personal relationships with other parties.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Boureghda, Y., Satha, H. & Bendebane, F. Chitin–Glucan Complex from Pleurotus ostreatus Mushroom: Physicochemical Characterization and Comparison of Extraction Methods. Waste Biomass Valor 12, 6139–6153 (2021). https://doi.org/10.1007/s12649-021-01449-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12649-021-01449-3