Abstract
Parkinson’s disease (PD) is a common neurodegenerative disease characterized by the death of dopaminergic neurons. Its pathogenesis comprises defects in the physiological pathway of mitophagy and mutations in the genes involved in this process’s regulatory mechanism. PD manifests itself with multiple motor and non-motor symptoms, and currently, there are multiple pharmacological treatments, and unconventional non-drug treatments available. The mainstay of Parkinson’s disease treatment has centered around directly manipulating neural mechanisms to retain high dopamine levels, either by exogenous administration, increasing intrinsic production, or inhibiting the breakdown of dopamine. In this review, we highlight a new potential biochemical modality of treatment, treating PD through glycolysis. We highlight how terazosin (TZ), via PGK1, increases ATP levels and how enhanced glycolysis serves a neuroprotective role in PD, and compensates for damage caused by mitophagy. We also discuss the role of quercetin, a bioactive flavonoid, in preventing the development of PD, and reversing mitochondrial dysfunction but only so in diabetic patients. Thus, further research should be conducted on glycolysis as a protective target in PD that can serve to not just prevent, but also alleviate the non-dopaminergic signs and symptoms of PD.


Similar content being viewed by others
Data Availability
There is no data to provide.
Abbreviations
- PD:
-
Parkinson’s disease
- BPH:
-
Benign prostate hyperplasia
- MPTP:
-
1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine
- 6-OHDA:
-
6-Hydroxydopamine
- PINK1:
-
PTEN-induced kinase 1
- LRRK2:
-
Leucine-rich repeat kinase 2
- WT:
-
Wilms’ tumor protein
- PGK-1:
-
Phosphoglycerate kinase 1
- PARKIN:
-
Parkinson’s juvenile disease protein 2
- PINK1:
-
PTEN-induced kinase 1
- ROS:
-
Reactive oxygen species
- VDAC1:
-
Voltage-dependent anion-selective channel 1
- PARL:
-
Presenilins-associated rhomboid-like protein L-DOPA: Levodopa
- MAO-B:
-
Monoamine oxidase-B
- COMT:
-
Catechol-O-methyltransferase GDNF: Glial cell-derived neurotrophic factor NRTN: Neurturin
- PRF:
-
Proanthocyanidin-rich fraction
- DBS:
-
Deep brain stimulation
- PT:
-
Physiotherapy
- VAS:
-
Visual Analogue Scale
- RCT:
-
Randomized controlled trial
- OT:
-
Occupational therapy
- PGK:
-
Phosphoglycerate kinase
- PK:
-
Pyruvate kinase
- HK:
-
Hexokinase
- PFK:
-
Phosphofructokinase
- PGK-1:
-
Phosphoglycerate kinase 1
- GA3PDH:
-
Glyceraldehyde-3-phosphate dehydrogenase
References
Akhtar MJ, Yar MS, Grover G, Nath R (2020) Neurological and psychiatric management using COMT inhibitors: A review. Bioorg Chem 94:103418. https://doi.org/10.1016/j.bioorg.2019.103418
Application of terazosin in treatment of Parkinsonism - Patent CN-105616417-A - PubChem. https://pubchem.ncbi.nlm.nih.gov/patent/CN-105616417-A. Accessed 28 Jul 2022
Anand David AV, Arulmoli R, Parasuraman S (2016) Overviews of biological importance of quercetin: a bioactive flavoniod. Pharmacogn Rev 10(20):84–89. https://doi.org/10.4103/0973-7847.194044
Armstrong MJ, Okun MS (2020) Diagnosis and treatment of Parkinson disease. JAMA 323(6):548. https://doi.org/10.1001/jama.2019.22360
Baik D, Yu YM, Jung S-Y, Kang H-Y (2022) Prevalence and patterns of the concurrent use of anticholinergics for the motor symptoms of Parkinson’s disease and acetylcholinesterase inhibitors in Parkinson’s disease patients with dementia: A cross-sectional study using Korea National Health Insurance Claims Data. BMC Geriatrics 22(1). https://doi.org/10.1186/s12877-022-03296-w
Batlevi Y, La Spada AR (2011) Mitochondrial autophagy in neural function, neurodegenerative disease, neuron cell death, and aging. Neurobiol Dis 43(1):46–51. https://doi.org/10.1016/j.nbd.2010.09.009
Bell SM, Burgess T, Lee J, Blackburn DJ, Allen SP, Mortiboys H (2020) Peripheral glycolysis in neurodegenerative diseases. Int J Mol Sci 21(23):8924. Published 2020 Nov 24. https://doi.org/10.3390/ijms21238924
Bloem BR, Okun MS, Klein C (2021) Parkinson’s disease. Lancet 397(10291):2284–2303. https://doi.org/10.1016/S0140-6736(21)00218-X
Bolus H, Crocker K, Boekhoff-Falk G, Chtarbanova S (2020) Modeling neurodegenerative disorders in Drosophila melanogaster. Int J Mol Sci 21(9):3055. Published 2020 Apr 26. https://doi.org/10.3390/ijms21093055
Braun S, Beurskens A, Kleynen M, Schols J, Wade D (2011) Rehabilitation with mental practice has similar effects on mobility as rehabilitation with relaxation in people with Parkinson’s disease: a multicentre randomised trial. J Physiother 57(1):27–34. https://doi.org/10.1016/S1836-9553(11)70004-2
Cai R, Zhang Y, Simmering JE, Schultz JL, Li Y, Fernandez-Carasa I, Consiglio A, Raya A, Polgreen PM, Narayanan NS, Yuan Y (2019) Enhancing glycolysis attenuates Parkinson’s disease progression in models and clinical databases. J Clin Investig 129(10):4539–4549. https://doi.org/10.1172/JCI129987
Celardo I, Martins LM, Gandhi S (2014) Unravelling mitochondrial pathways to Parkinson’s disease. Brit J Pharmacol 171(8):1943–1957. https://doi.org/10.1111/bph.12433
Chan HH, Tse MK, Kumar S, Zhuo L (2018) A novel selective MAO- B inhibitor with neuroprotective and anti- Parkinsonian properties. Eur J Pharmacol 818:254–262. https://doi.org/10.1016/j.ejphar.2017.10.023
Chen X, Zhao C, Li X et al (2015) Terazosin activates Pgk1 and Hsp90 to promote stress resistance. Nat Chem Biol 11:19–25. https://doi.org/10.1038/nchembio.1657
Clarke CE, Patel S, Ives N, Rick CE, Dowling F, Woolley R, Wheatley K , Walker MF, Sackley CM, PD REHAB Collaborative Group (2016) Physiotherapy and occupational therapy vs no therapy in mild to moderate Parkinson disease: A randomized clinical trial. JAMA Neurol 73(3):291–299. https://doi.org/10.1001/jamaneurol.2015.4452
Cormet-Boyoka E, Caution K, Dakhlallah D, Amer AO (2017) Chapter 3- The role of p62 in aggregopathies. Autophagy: cancer, other pathologies, inflammation, immunity, infection and aging 12:139–147. https://doi.org/10.1016/B978-0-12-812146-7.00003-2
Cunnane SC, Trushina E, Morland C et al (2020) Brain energy rescue: an emerging therapeutic concept for neurodegenerative disorders of ageing. Nat Rev Drug Discov 19(9):609–633. https://doi.org/10.1038/s41573-020-0072-x
da Silva SB, Feitosa SG, de L AS, Santos RC, Dos Anjos JV, Araújo AV (2022) A concise and useful guide to understand how alpha1 adrenoceptor antagonists work [published online ahead of print, 2022 May 4]. Mini Rev Med Chem. https://doi.org/10.2174/1389557522666220504141949
Dawson T, Golde T, Lagier-Tourenne C (2018) Animal models of neurodegenerative diseases. Nat Neurosci 21(10):1370–1379. https://doi.org/10.1016/j.bbr.2017.10.021
Deas E, Wood NW, Plun-Favreau H (2011) Mitophagy and Parkinson’s disease: The PINK1–parkin link. Biochem Biophys Acta 1813(4):623–633. https://doi.org/10.1016/j.bbamcr.2010.08.007
Dezsi L, Vecsei L (2017) Monoamine oxidase B inhibitors in Parkinson’s disease. CNS Neurol Disord Drug Targets 16(4):425–439. https://doi.org/10.2174/1871527316666170124165222
Deeb W, Nozile-Firth K, Okun MS (2019) Parkinson’s disease: diagnosis and appreciation of comorbidities. In: Handb Clin Neurol. Elsevier B.V. p 257–77. https://doi.org/10.1016/B978-0-12-804766-8.00014-5
de Vries RLA, Przedborski S (2013) Mitophagy and Parkinson's disease: Be eaten to stay healthy. Mol Cell Neurosci 55:37–43. https://doi.org/10.1016/j.mcn.2012.07.008
Dexter DT, Statton SA, Whitmore C, Freinbichler W, Weinberger P, Tipton KF, Della Corte L, Ward RJ, Crichton RR (2011) Clinically available iron chelators induce neuroprotection in the 6-OHDA model of Parkinson’s disease after peripheral administration. J Neural Transm (Vienna) 118(2):223–231. https://doi.org/10.1007/s00702-010-0531-3
Di Lazzaro G, Magrinelli F, Estevez-Fraga C, Valente EM, Pisani A, Bhatia KP (2021) X-Linked parkinsonism: phenotypic and genetic heterogeneity. Mov Disord 36(7):1511–1525. https://doi.org/10.1002/mds.28565
Dolhun R (2018) Ask the MD: the language of Parkinson’s, ‘off’ time [Internet]. The Michael J. Fox Foundation for Parkinson's Research [cited 2021 Mar 5]. Available from: https://www.michaeljfox.org/news/ask-md-language-parkinsons-time#:~:text=%22Off%22%20is%20time%20when%20Parkinson%27s,taking%20levodopa%20for%20longer%20periods
Dong Z, Wang Z, Yang K, Liu Y, Gao W, Chen W (2009) Tamsulosin versus terazosin for benign prostatic hyperplasia: a systematic review. Systems Biology in Reproductive Medicine 55(4):129–136. https://doi.org/10.3109/19396360902833235
Erro R, Stamelou M (2017) The motor syndrome of Parkinson’s disease. Int Rev Neurobiol 132:25–32. https://doi.org/10.1016/bs.irn.2017.01.004
Firbank MJ, Yarnall AJ, Lawson RA et al (2017) Cerebral glucose metabolism and cognition in newly diagnosed Parkinson’s disease: ICICLE-PD study. J Neurol Neurosurg Psychiatry 88(4):310–316. https://doi.org/10.1136/jnnp-2016-313918
Foltynie T (2019) Glycolysis as a therapeutic target for Parkinson’s disease. Lancet Neurol 18(12):1072–1074. https://doi.org/10.1016/S1474-4422(19)30404-1
Gaballah HH, Zakaria SS, Mwafy SE, Tahoon NM, Ebeid AM (2017) Mechanistic insights into the effects of quercetin and/or GLP-1 analogue liraglutide on high-fat diet/streptozotocin-induced type 2 diabetes in rats. Biomed Pharmacother 92:331–339. https://doi.org/10.1016/j.biopha.2017.05.086
Goiran T, Eldeeb MA, Zorca CE, Fon EA (2022) Hallmarks and molecular tools for the study of mitophagy in Parkinson’s disease. Cells 11(13):2097. https://doi.org/10.3389/fnagi.2022.890823
Goyal V, Radhakrishnan D (2018) Parkinson’s disease: a review. Neurol India 66(7):26. https://doi.org/10.4103/0028-3886.226451
Guzmán R, Fernández JC, Pedroso M, Fernández L, Illnait J, Mendoza S, Quiala AT, Rodríguez Z, Mena J, Rodíguez A, Campos M, Sánchez C, Alvarez Y, Jiménez G (2019) Efficacy and tolerability of Roystonea regia lipid extract (D-004) and terazosin in men with symptomatic benign prostatic hyperplasia: a 6-month study. Ther Adv Urol 11(11):1756287219854923. https://doi.org/10.1177/1756287219854923
Han J-H, Kim S-E, Ko I-G, Kim J, Kim KH (2017) Afferent pathway-mediated effect of α1 adrenergic antagonist, tamsulosin, on the neurogenic bladder after SPIANL Cord Injury. Int Neurourol J 21(3):178–188. https://doi.org/10.5213/inj.1734984.492
Hayes MT (2019) Parkinson’s disease and parkinsonism. Am J Med Elsevier Inc 132:802–807. https://doi.org/10.1016/j.amjmed.2019.03.001
Hauser RA (2011) Future treatments for Parkinson’s disease: surfing the PD pipeline. Int J Neurosci 121(Suppl 2):53–62. https://doi.org/10.3109/00207454.2011.620195
Heisters D (2011) Parkinson's: Symptoms, treatments and research. Br J Nurs 13–26;20(9):548–54. https://doi.org/10.12968/bjon.2011.20.9.548
Hipkiss AR (2019) Aging, Alzheimer’s disease and dysfunctional glycolysis; similar effects of too much and too little. Aging Dis10:1328–1331. https://doi.org/10.14336/AD.2019.0611
Houghton MJ, Kerimi A, Tumova S, Boyle JP, Williamson G (2018) Quercetin preserves redox status and stimulates mitochondrial function in metabolically-stressed HepG2 cells. Free Radic Biol Med 129:296–309. https://doi.org/10.1016/j.freeradbiomed.2018.09.037
Huang J, Qin X, Cai X, Huang Y (2020) Effectiveness of acupuncture in the treatment of Parkinson’s disease: an overview of systematic reviews. Front Neurol 11. https://doi.org/10.3389/fneur.2020.00917
Huot P (2018) 5-HT1A agonists and dyskinesia in Parkinson’s disease: a pharmacological perspective. Neurodegener Dis Manag 8(4):207–209. https://doi.org/10.2217/nmt-2018-0017
Hurelbrink CB, Lewis SJ (2011) Pathological considerations in the treatment of Parkinson’s disease: More than just a wiring diagram. Clin Neurol Neurosurg 113(1):1–6. https://doi.org/10.1016/j.clineuro.2010.08.010
Jagadeesan AJ, Murugesan R, Vimala Devi S, Meera M, Madhumala G, Vishwanathan Padmaja M et al (2017) Current trends in etiology, prognosis and therapeutic aspects of Parkinson’s disease: A review. Acta Bio-Medica: Atenei Parmensis 88:249–62. https://doi.org/10.23750/abm.v88i3.6063
Jankovic J (2008) Parkinson’s disease: Clinical features and diagnosis. J Neurol Neurosurg Psychia. BMJ Publishing Group 79:368–76. https://doi.org/10.1136/jnnp.2007.131045
Jia L, Huang S, Yin X, Zan Y, Guo Y, Han L (2018) Quercetin suppresses the mobility of breast cancer by suppressing glycolysis through Akt-mTOR pathway mediated autophagy induction. Life Sci 208:123–130. https://doi.org/10.1016/j.lfs.2018.07.027
Kanda T, Jenner P (2020) Can adenosine A2A receptor antagonists modify motor behavior and Dyskinesia in experimental models of Parkinson’s disease?. Parkinson Rel Disord 80. https://doi.org/10.1016/j.parkreldis.2020.09.026
Kincses Z, Vecsei L (2010) Pharmacological therapy in Parkinson’s disease: Focus on Neuroprotection. CNS Neurosci Ther 17(5):345–367. https://doi.org/10.1111/j.1755-5949.2010.00150.x
Kwon SH, Lee SR, Park YJ et al (2019) Suppression of 6-hydroxydopamine-induced oxidative stress by hyperoside via activation of Nrf2/HO-1 signaling in dopaminergic neurons. Int J Mol Sci 20(23):5832. Published 2019 Nov 20. https://doi.org/10.3390/ijms20235832
Malek N (2019) Deep brain stimulation in Parkinson’s disease. Neurol India Jul-Aug 67(4):968–978. https://doi.org/10.4103/0028-3886.266268
Manta K, Papakyriakopoulou P, Chountoulesi M et al (2020) Preparation and biophysical characterization of quercetin inclusion complexes with β-cyclodextrin derivatives to be formulated as possible nose-to-brain quercetin delivery systems. Mol Pharm 17(11):4241–4255. https://doi.org/10.1021/acs.molpharmaceut.0c00672
Maraki MI, Yannakoulia M, Stamelou M et al (2018) Mediterranean diet adherence is related to reduced probability of prodromal Parkinson’s disease. Mov Disord 34(1):48–57. https://doi.org/10.1002/mds.27489
McQuibban GA, Bulman DE (2011) The PARLance of Parkinson disease. Autophagy 7(7):790–792. https://doi.org/10.4161/auto.7.7.15614
Milanese C, Payán-Gómez C, Galvani M et al (2019) Peripheral mitochondrial function correlates with clinical severity in idiopathic Parkinson’s disease. Mov Disord 34(8):1192–1202. https://doi.org/10.1002/mds.27723
Mohammad ME, Vizcarra JA, Garcia X et al (2021) Impact of behavioral side effects on the management of parkinson patients treated with dopamine agonists. Clinical Parkinsonism & Related Disorders 4:100091. https://doi.org/10.1016/j.prdoa.2021.100091
Morales-Briceño H, Ha AD, London K, Farlow D, Chang FCF, Fung VSC (2019) Parkinsonism in PGK1 deficiency implicates the glycolytic pathway in nigrostriatal dysfunction. Parkinsonism Relat Disord 64:319–323. https://doi.org/10.1016/j.parkreldis.2019.04.004
Niu C, Zhang J, Mei J, Xianming Fu (2011) Does neurotrophic factor benefit to PD therapy via co- function with ubiquitin-proteasome system? Med Hypotheses 76(4):589–592. https://doi.org/10.1016/j.mehy.2011.01.007
Ohno Y (2011) Therapeutic role of 5-HT1A receptors in the treatment of schizophrenia and Parkinson’s disease. CNS Neurosci Ther 17(1):58–65. https://doi.org/10.1111/j.1755-5949.2010.00211.x
O’Sullivan JD (2020) Too much of a good thing: hedonistic homoeostatic dysregulation and other behavioural consequences of excessive dopamine replacement therapy in Parkinson’s disease. J Neurol Neurosurg Psychiatry 91(6):566–567. https://doi.org/10.1136/jnnp-2019-322378
Panche AN, Diwan AD, Chandra SR (2016) Flavonoids: An overview. J Nutr Sci 5:e47. Published 2016 Dec 29. https://doi.org/10.1017/jns.2016.41
Perez NE, Godbole NP, Amin K, Syan R, Gater DR Jr (2022) Neurogenic bladder physiology, pathogenesis, and management after spinal cord injury. J Pers Med 12(6):968. Published 2022 Jun 14. https://doi.org/10.3390/jpm12060968
Poewe W, Seppi K, Tanner CM et al (2017) Parkinson disease. Nat Rev Dis Primers 3:17013. Published 2017 Mar 23. https://doi.org/10.1038/nrdp.2017.1367
Prediger RD, Aguiar AS, Matheus FC, Walz R, Antoury L, Raisman-Vozari R, Doty RL (2011) The intranasal administration of 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP): A new rodent model to test palliative and neuroprotective agents for Parkinson's disease. Curr Pharm Des 17(5):489–507. https://doi.org/10.2174/138161211795164095
Qiu L, Luo Y, Chen X (2018) Quercetin attenuates mitochondrial dysfunction and biogenesis via upregulated AMPK/SIRT1 signaling pathway in OA rats. Biomed Pharmacother 103:1585–1591. https://doi.org/10.1016/j.biopha.2018.05.003
Radhakrishnan DM, Goyal V (2018) Parkinson’s disease: a review. Neurol India 66(Supplement):S26–S35. https://doi.org/10.4103/0028-3886.226451
Radhakrishnan DM, Goyal V (2018) Parkinson’s disease: a review. Neurol India 66(7):26. https://doi.org/10.4103/0028-3886.226451
Rajesh P, Kelly EL (2011) Current issues in the diagnosis and treatment of Parkinson’s disease. Introduction Int J Neurosci 121(Suppl 2):1–2. https://doi.org/10.3109/00207454.2011.622454
Rojas-Pirela M, Andrade-Alviárez D, Rojas V, Kemmerling U, Cáceres AJ, Michels PA, Concepción JL, Quiñones W (2020) Phosphoglycerate kinase: structural aspects and functions, with special emphasis on the enzyme from Kinetoplastea. Open Biol 10(11):200302. https://doi.org/10.1098/rsob.200302
Sakaue S, Kasai T, Mizuta I, Suematsu M, Osone S, Azuma Y, Imamura T, Tokuda T, Kanno H, El-Agnaf O, Morimoto M (2017) Early-onset parkinsonism in a pedigree with phosphoglycerate kinase deficiency and a heterozygous carrier: do PGK-1 mutations contribute to vulnerability to parkinsonism?. NPJ Parkinsons Dis 3:13. https://doi.org/10.1038/s41531-017-0014-4
Sarkar S, Raymick J, Imam S (2016) Neuroprotective and therapeutic strategies against Parkinson’s disease: recent perspectives. Int J Mol Sci 17(6):904. https://doi.org/10.3390/ijms17060904
Segur-Bailach E, Ugarteburu O, Tort F et al (2022) Over-mutated mitochondrial, lysosomal and TFEB-regulated genes in Parkinson’s disease. J Clin Med 11(6):1749. Published 2022 Mar 21. https://doi.org/10.3390/jcm11061749
Shi G (2011) Functional alteration of PARL contributes to mitochondrial dysregulation in Parkinson’s disease. Hum Mol Gen 20(10):1966–1974. https://doi.org/10.1093/hmg/ddr077
Shi GJ, Li Y, Cao QH et al (2019) In vitro and in vivo evidence that quercetin protects against diabetes and its complications: A systematic review of the literature. Biomed Pharmacother 109:1085–1099. https://doi.org/10.1016/j.biopha.2018.10.130
Silvian LF (2022) PINK1/Parkin pathway activation for mitochondrial quality control – which is the best molecular target for therapy? Frontiers in Aging Neuroscience 14. https://doi.org/10.3389/fnagi.2022.890823
Simmering JE, Welsh MJ, Liu L, Narayanan NS, Pottegård A (2021) Association of glycolysis-enhancing α-1 blockers with risk of developing Parkinson disease. JAMA Neurol e205157. https://doi.org/10.1001/jamaneurol.2020.5157
Song Z, Zhang J, Xue T et al (2021) Different catechol-O-methyltransferase inhibitors in parkinson's disease: A bayesian network meta-analysis. Front Neurol 12. https://doi.org/10.3389/fneur.2021.707723
Sotiriou E, Greene P, Krishna S, Hirano M, DiMauro S (2010) Myopathy and parkinsonism in phosphoglycerate kinase deficiency. Muscle Nerve 41(5):707–710. https://doi.org/10.1002/mus.21612
Stoker TB, Barker RA (2020) Recent developments in the treatment of Parkinson's Disease. F1000Res 9:F1000 Faculty Rev-862. Published 2020 Jul 31. https://doi.org/10.12688/f1000research.25634.1
Sun X, Xue L, Wang Z, Xie A (2022) Update to the treatment of Parkinson’s disease based on the gut-brain axis mechanism. Front Neurosci 16. https://doi.org/10.3389/fnins.2022.878239
Tambasco N, Romoli M, Calabresi P (2018) Levodopa in Parkinson’s disease: current status and future developments. Curr Neuropharmacol 16(8):1239–1252. https://doi.org/10.2174/1570159x15666170510143821
Tang BL (2020) Glucose, glycolysis, and neurodegenerative diseases. J Cell Physiol 235(11):7653–7662. https://doi.org/10.1002/jcp.29682
Tysnes OB, Storstein A (2017) Epidemiology of Parkinson’s disease. J Neural Transm 124(8):901–905. https://doi.org/10.1007/s00702-017-1686-y
Vingill S, Connor-Robson N, Wade-Martins R (2018) Are rodent models of Parkinson’s disease behaving as they should? Behav Brain Res 352:133–141. https://doi.org/10.1016/j.bbr.2017.10.021
Vos M, Klein C (2021) The importance of Drosophila melanogaster research to uncover cellular pathways underlying Parkinson’s disease. Cells 10(3):579. Published 2021 Mar 6. https://doi.org/10.3390/cells10030579
Warren N, O’Gorman C, Lehn A, Siskind D (2017) Dopamine dysregulation syndrome in Parkinson’s disease: a systematic review of published cases. J Neurol Neurosurg Psychiatry 88(12):1060–1064. https://doi.org/10.1136/jnnp-2017-315985
Wu H, Pan L, Gao C et al (2019) Quercetin inhibits the proliferation of glycolysis-addicted HCC cells by reducing hexokinase 2 and Akt-mTOR pathway. Molecules 24(10):1993. Published 2019 May 24. https://doi.org/10.3390/molecules24101993
Xilouri M, Stefanis L (2011) Autophagic pathways in Parkinson disease and related disorders. Expert Rev Mol Med 21(13):e8. https://doi.org/10.1017/S1462399411001803
Xiang C, Teng Y, Yao C, Li X, Cao M, Li X, Pan G, Lu K, Galons H, Yu P (2018) Antioxidant properties of flavonoid derivatives and their hepatoprotective effects on CCL4induced acute liver injury in mice. RSC Adv 8(28):15366–15371. https://doi.org/10.1039/c8ra02523a
Yang CH, Raja A (2022) Terazosin. In: StatPearls. Treasure Island (FL): StatPearls Publishing
Yin W, Löf M, Pedersen NL, Sandin S, Fang F (2020) Mediterranean dietary pattern at middle age and risk of Parkinson’s disease: a Swedish cohort study. Mov Disord 36(1):255–260. https://doi.org/10.1002/mds.28314
Zhang PL, Chen Y, Zhang CH, Wang YX, Fernandez-Funez P (2018) Genetics of Parkinson’s disease and related disorders. J Med Gen. BMJ Publishing Group 55:73–80. https://doi.org/10.1136/jmedgenet-2017-105047
Acknowledgements
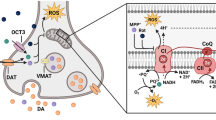
We would like to thank Yumna Waseem for helping us with Fig. 1 in this paper.
Funding
None. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Authors Unaiza Naeem, Abdul Rehman Arshad, Areesha Jawed, Farea Eqbal, Laiba Imran, and Zayeema Khan performed the literature search for the review. Additionally. Unaiza Naeem did the conceptualizing of the paper, Farea Eqbal sorted the references in chronological order, and Laiba Imran did the correspondence. All authors were involved in writing the manuscript and the manuscript was reviewed by the senior author, Farhat Ijaz.
Corresponding author
Ethics declarations
Ethical Approval
Not applicable.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Naeem, U., Arshad, A.R., Jawed, A. et al. Glycolysis: The Next Big Breakthrough in Parkinson’s Disease. Neurotox Res 40, 1707–1717 (2022). https://doi.org/10.1007/s12640-022-00579-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-022-00579-3