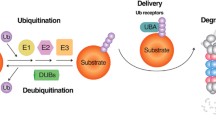
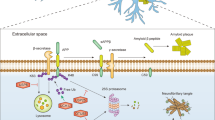
Abstract
Among the most salient features that underpin the development of aging-related neurodegenerative disorders are the accumulation of protein aggregates and the decrease in cellular degradation capacity. Mammalian cells have evolved sophisticated quality control mechanisms to repair or eliminate the otherwise abnormal or misfolded proteins. Chaperones identify unstable or abnormal conformations in proteins and often help them regain their correct conformation. However, if repair is not an option, abnormal proteins are selectively degraded to prevent undesired interactions with other proteins or oligomerization into toxic multimeric complexes. The autophagic-lysosomal system and the ubiquitin–proteasome system mediate the selective and targeted degradation of abnormal or aberrant protein fragments. Despite an increasing understanding regarding the molecular responses that counteract the formation and clearance of dysfunctional protein aggregates, the role of N-degrons in these processes is poorly understood. Previous work demonstrated that the Arg-N-end rule degradation pathway (Arg-N-degron pathway) mediates the degradation of neurodegeneration-associated proteins, thereby regulating crucial signaling hubs that modulate the progression of neurodegenerative diseases. Herein, we discuss the functional interconnection between N-degron pathways and proteins associated with neurodegenerative disorders, including Alzheimer’s disease, amyotrophic lateral sclerosis, and Parkinson’s disease. We also highlight some future prospects related to how the molecular insights gained from these processes will help unveil novel therapeutic approaches.







Similar content being viewed by others
References
Almeida CG, Takahashi RH, Gouras GK (2006) β-Amyloid accumulation impairs multivesicular body sorting by inhibiting the ubiquitin-proteasome system. J Neurosci 26:4277–4288
Altieri DC, Stein GS, Lian JB, Languino LR (2012) TRAP-1, the mitochondrial Hsp90. Biochim Biophys Acta Mol Cell Res 1823:767–773
Andreadis A (2006) Misregulation of tau alternative splicing in neurodegeneration and dementia. In: Jeanteur P (ed) Alternative splicing and disease. Springer, Berlin Heidelberg, Berlin, Heidelberg, pp 89–107
Arai T, Hasegawa M, Akiyama H et al (2006) TDP-43 is a component of ubiquitin-positive tau-negative inclusions in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Biochem Biophys Res Commun 351:602–611. https://doi.org/10.1016/j.bbrc.2006.10.093
Arriagada PV, Growdon JH, Hedley-Whyte ET, Hyman BT (1992) Neurofibrillary tangles but not senile plaques parallel duration and severity of Alzheimer’s disease. Neurology 42:631–639. https://doi.org/10.1212/WNL.42.3.631
Ayala YM, De Conti L, Avendaño-Vázquez SE et al (2011) TDP-43 regulates its mRNA levels through a negative feedback loop. EMBO J 30:277–288. https://doi.org/10.1038/emboj.2010.310
Ayala YM, Zago P, Ambrogio A et al (2008) Structural determinants of the cellular localization and shuttling of TDP-43. J Cell Sci 121:3778–3785. https://doi.org/10.1242/jcs.038950
Baba M, Nakajo S, Tu P-H et al (1998) Aggregation of alpha-synuclein in Lewy bodies of sporadic Parkinson’s disease and dementia with Lewy bodies. Am J Pathol 152:879
Bachmair A, Finley D, Varshavsky A (1986) In vivo half-life of a protein is a function of its amino-terminal residue. Science 234:179–186
Baker JE, Lim YY, Pietrzak RH et al (2017) Cognitive impairment and decline in cognitively normal older adults with high amyloid-β: A meta-analysis. Alzheimers Dement 6:108–121. https://doi.org/10.1016/j.dadm.2016.09.002
Barbier P, Zejneli O, Martinho M et al (2019) Role of Tau as a microtubule-associated protein: structural and functional aspects. Front Aging Neurosci 11:204. https://doi.org/10.3389/fnagi.2019.00204
Barykin EP, Mitkevich VA, Kozin SA, Makarov AA (2017) Amyloid β modification: a key to the sporadic Alzheimer’s disease? Front Genet 8:58
Bendor JT, Logan TP, Edwards RH (2013) The function of α-synuclein. Neuron 79:1044–1066
Berning BA, Walker AK (2019) The pathobiology of TDP-43 C-terminal fragments in ALS and FTLD. Front Neurosci 13:335
Brandt R, Léger J, Lee G (1995) Interaction of tau with the neural plasma membrane mediated by tau’s amino-terminal projection domain. J Cell Biol 131:1327–1340
Brower CS, Piatkov KI, Varshavsky A (2013) Neurodegeneration-associated protein fragments as short-lived substrates of the N-end rule pathway. Mol Cell 50:161–171
Buée L, Bussière T, Buée-Scherrer V et al (2000) Tau protein isoforms, phosphorylation and role in neurodegenerative disorders. Brain Res Rev 33:95–130. https://doi.org/10.1016/S0165-0173(00)00019-9
Buratti E (2018) TDP-43 post-translational modifications in health and disease. Expert Opin Ther Targets 22:279–293. https://doi.org/10.1080/14728222.2018.1439923
Buratti E, Baralle FE (2008) Multiple roles of TDP-43 in gene expression, splicing regulation, and human disease. Front Biosci 13:867–878
Buratti E, Brindisi A, Giombi M et al (2005) TDP-43 binds heterogeneous nuclear ribonucleoprotein A/B through its C-terminal tail an important region for the inhibition of cystic fibrosis transmembrane conductance regulator exon 9 splicing. J Biol Chem 280:37572–37584
Busch A, Hertel KJ (2012) Evolution of SR protein and hnRNP splicing regulatory factors. Wires RNA 3:1–12. https://doi.org/10.1002/wrna.100
Caldwell JH, Klevanski M, Saar M, Müller UC (2013) Roles of the amyloid precursor protein family in the peripheral nervous system. Mech Dev 130:433–446. https://doi.org/10.1016/j.mod.2012.11.001
Carrotta R, Carlo MD, Manno M et al (2006) Toxicity of recombinant β-amyloid prefibrillar oligomers on the morphogenesis of the sea urchin Paracentrotus lividus. FASEB J 20:1916–1917. https://doi.org/10.1096/fj.06-5716fje
Cascella R, Fani G, Capitini C et al (2017) Quantitative assessment of the degradation of aggregated TDP-43 mediated by the ubiquitin proteasome system and macroautophagy. FASEB J 31:5609–5624. https://doi.org/10.1096/fj.201700292RR
Chakraborty G, Ingoglia NA (1993) N-terminal arginylation and ubiquitin-mediated proteolysis in nerve regeneration. Brain Res Bull 30:439–445. https://doi.org/10.1016/0361-9230(93)90276-H
Chen HH, Liu P, Auger P et al (2018) Calpain-mediated tau fragmentation is altered in Alzheimer’s disease progression. Sci Rep 8:16725. https://doi.org/10.1038/s41598-018-35130-y
Chen SJ, Wu X, Wadas B et al (2017) An N-end rule pathway that recognizes proline and destroys gluconeogenic enzymes. Science 355: eaal3655.
Chen X, de Silva HR, Pettenati MJ (1995) The human NACP/alpha-synuclein gene: Chromosome assignment to 4q21. 3–q22 and TaqI RFLP analysis. Genomics 26:425–427
Chesser A, Pritchard S, Johnson GV (2013) Tau clearance mechanisms and their possible role in the pathogenesis of Alzheimer disease. Front Neurol 4:122
Chirita CN, Congdon EE, Yin H, Kuret J (2005) Triggers of full-length tau aggregation: a role for partially folded intermediates. Biochemistry 44:5862–5872. https://doi.org/10.1021/bi0500123
Choi D-H, Kim Y-J, Kim Y-G et al (2011) Role of matrix metalloproteinase 3-mediated α-synuclein cleavage in dopaminergic cell death. J Biol Chem 286:14168–14177
Chong FP, Ng KY, Koh RY, Chye SM (2018) Tau proteins and tauopathies in Alzheimer’s disease. Cell Mol Neurobiol 38:965–980. https://doi.org/10.1007/s10571-017-0574-1
Colombrita C, Onesto E, Megiorni F et al (2012) TDP-43 and FUS RNA-binding proteins bind distinct sets of cytoplasmic messenger RNAs and differently regulate their post-transcriptional fate in motoneuron-like cells. J Biol Chem 287:15635–15647. https://doi.org/10.1074/jbc.M111.333450
Conway KA, Rochet J-C, Bieganski RM, Lansbury PT (2001) Kinetic stabilization of the α-synuclein protofibril by a dopamine-α-synuclein adduct. Science 294:1346–1349
De Strooper B, Beullens M, Contreras B et al (1997) Phosphorylation, subcellular localization, and membrane orientation of the Alzheimer’s disease-associated presenilins. J Biol Chem 272:3590–3598. https://doi.org/10.1074/jbc.272.6.3590
Deas E, Plun-Favreau H, Gandhi S et al (2010) PINK1 cleavage at position A103 by the mitochondrial protease PARL. Hum Mol Genet 20:867–879
Delobel P, Leroy O, Hamdane M et al (2005) Proteasome inhibition and Tau proteolysis: an unexpected regulation. FEBS Lett 579:1–5. https://doi.org/10.1016/j.febslet.2004.11.018
Dev KK, Hofele K, Barbieri S et al (2003) Part II: α-synuclein and its molecular pathophysiological role in neurodegenerative disease. Neuropharmacology 45:14–44
Devred F, Barbier P, Lafitte D et al (2010) Chapter 23 - Microtubule and MAPs: thermodynamics of complex formation by AUC, ITC, fluorescence, and NMR. In: Correia JJ (ed) Wilson L. Methods Cell Biol, Academic Press, pp 449–480
Dikic I (2017) Proteasomal and autophagic degradation systems. Annu Rev Biochem 86:193–224
Dissmeyer N, Rivas S, Graciet E (2018) Life and death of proteins after protease cleavage: protein degradation by the N-end rule pathway. New Phytol 218:929–935
Dixit R, Ross JL, Goldman YE, Holzbaur ELF (2008) Differential regulation of dynein and kinesin motor proteins by tau. Science 319:1086–1089. https://doi.org/10.1126/science.1152993
Dreyfuss G, Kim VN, Kataoka N (2002) Messenger-RNA-binding proteins and the messages they carry. Nat Rev Mol Cell Biol 3:195–205. https://doi.org/10.1038/nrm760
Dubey M, Chaudhury P, Kabiru H, Shea TB (2008) Tau inhibits anterograde axonal transport and perturbs stability in growing axonal neurites in part by displacing kinesin cargo: Neurofilaments attenuate tau-mediated neurite instability. Cell Motil 65:89–99. https://doi.org/10.1002/cm.20243
Durcan TM, Fon EA (2015) The three ‘P’s of mitophagy: PARKIN, PINK1, and post-translational modifications. Genes Dev 29:989–999
Eldeeb M, Esmaili M, Fahlman R (2019a) Degradation of proteins with N-terminal glycine. Nat Struct Mol Biol 26:761–763
Eldeeb M, Fahlman R (2016a) The-N-end rule: the beginning determines the end. Protein Pept Lett 23:343–348
Eldeeb MA, Bayne AN, Trempe J-F, Fon EA (2020a) Fine-tuning TOM-mitochondrial import via ubiquitin. Trends Cell Biol 30:425–427. https://doi.org/10.1016/j.tcb.2020.04.007
Eldeeb MA, Fahlman RP (2014) The anti-apoptotic form of tyrosine kinase Lyn that is generated by proteolysis is degraded by the N-end rule pathway. Oncotarget 5:2714–2722
Eldeeb MA, Fahlman RP (2016b) Phosphorylation impacts N-end rule degradation of the proteolytically activated form of BMX kinase. J Biol Chem 291:22757–22768
Eldeeb MA, Fahlman RP (2018) Does too much MAGIC lead to mitophagy? Trends Biochem Sci 43:485–487
Eldeeb MA, Fahlman RP, Esmaili M, Fon EA (2019b) Formylation of eukaryotic cytoplasmic proteins: linking stress to degradation. Trends Biochem Sci 44:181–183. https://doi.org/10.1016/j.tibs.2018.12.008
Eldeeb MA, Fahlman RP, Esmaili M, Ragheb MA (2018a) Regulating apoptosis by degradation: the N-end rule-mediated regulation of apoptotic proteolytic fragments in mammalian cells. Int J Mol Sci 19:3414
Eldeeb MA, Fahlman RP, Ragheb MA, Esmaili M (2019c) Does N-terminal protein acetylation lead to protein degradation? BioEssays 41:1800167
Eldeeb MA, Leitao LC, Fahlman RP (2018b) Emerging branches of the N-end rule pathways are revealing the sequence complexities of N-termini dependent protein degradation. Biochem Cell Biol 96:289–294
Eldeeb MA, Ragheb MA (2020) N-degron-mediated degradation and regulation of mitochondrial PINK1 kinase. Curr Genet 66:693–701. https://doi.org/10.1007/s00294-020-01062-2
Eldeeb MA, Ragheb MA, Esmaili M (2020b) How does protein degradation regulate TOM machinery-dependent mitochondrial import? Curr Genet 66:501–505. https://doi.org/10.1007/s00294-020-01056-0
Eldeeb MA, Ragheb MA, Esmaili M, Hussein F (2020c) Physiological state dictates the proteasomal-mediated purging of misfolded protein fragments. Protein Pept Lett 27:251–255. https://doi.org/10.2174/0929866526666191026111951
Eldeeb MA, Ragheb MA, Fon EA (2019d) Cell death: N-degrons fine-tune pyroptotic cell demise. Curr Biol 29:R588–R591
Eldeeb MA, Siva-Piragasam R, Ragheb MA et al (2019e) A molecular toolbox for studying protein degradation in mammalian cells. J Neurochem 151:520–533. https://doi.org/10.1111/jnc.14838
Eldeeb MA, Zorca CE, Ragheb MA et al (2021) Fine-tuning ER-phagy by post-translational modifications. BioEssays 43:2000212. https://doi.org/10.1002/bies.202000212
Fá M, Puzzo D, Piacentini R et al (2016) Extracellular tau oligomers produce an immediate impairment of LTP and memory. Sci Rep 6:19393. https://doi.org/10.1038/srep19393
Fava VM, Xu YZ, Lettre G et al (2019) Pleiotropic effects for Parkin and LRRK2 in leprosy type-1 reactions and Parkinson’s disease. Proc Natl Acad Sci U S A 116:15616–15624
Ferreira A, Bigio EH (2011) Calpain-mediated Tau cleavage: a mechanism leading to neurodegeneration shared by multiple tauopathies. Mol Med 17:676–685. https://doi.org/10.2119/molmed.2010.00220
Freibaum BD, Chitta RK, High AA, Taylor JP (2010) Global analysis of TDP-43 interacting proteins reveals strong association with RNA splicing and translation machinery. J Proteome Res 9:1104–1120
Gamblin TC, Chen F, Zambrano A et al (2003) Caspase cleavage of tau: linking amyloid and neurofibrillary tangles in Alzheimer’s disease. Proc Natl Acad Sci U S A 100:10032–10037. https://doi.org/10.1073/pnas.1630428100
Garg S, Timm T, Mandelkow E-M et al (2011) Cleavage of Tau by calpain in Alzheimer’s disease: the quest for the toxic 17 kD fragment. Neurobiol Aging 32:1–14. https://doi.org/10.1016/j.neurobiolaging.2010.09.008
Gauthier-Kemper A, Alonso MS, Sündermann F et al (2018) Annexins A2 and A6 interact with the extreme N-terminus of tau and thereby contribute to tau’s axonal localization. J Biol Chem 293:8065–8076
Gendron TF, Petrucelli L (2009) The role of tau in neurodegeneration. Mol Neurodegener 4:13. https://doi.org/10.1186/1750-1326-4-13
Giacobini E, Gold G (2013) Alzheimer disease therapy-moving from amyloid-β to tau. Nat Rev Neurol 9:677–686. https://doi.org/10.1038/nrneurol.2013.223
Gibbs DJ, Bacardit J, Bachmair A, Holdsworth MJ (2014) The eukaryotic N-end rule pathway: conserved mechanisms and diverse functions. Trends Cell Biol 24:603–611
Goate A, Chartier-Harlin M-C, Mullan M et al (1991) Segregation of a missense mutation in the amyloid precursor protein gene with familial Alzheimer’s disease. Nature 349:704–706. https://doi.org/10.1038/349704a0
Goedert M (2018) Tau filaments in neurodegenerative diseases. FEBS Lett 592:2383–2391. https://doi.org/10.1002/1873-3468.13108
Goedert M, Eisenberg DS, Crowther RA (2017) Propagation of tau aggregates and neurodegeneration. Annu Rev Neurosci 40:189–210
Goedert M, Spillantini M, Jakes R et al (1989a) Multiple isoforms of human microtubule-associated protein tau: sequences and localization in neurofibrillary tangles of Alzheimer’s disease. Neuron 3:519–526
Goedert M, Spillantini MG, Potier MC et al (1989b) Cloning and sequencing of the cDNA encoding an isoform of microtubule-associated protein tau containing four tandem repeats: differential expression of tau protein mRNAs in human brain. EMBO J 8:393–399. https://doi.org/10.1002/j.1460-2075.1989.tb03390.x
Gong B, Chen F, Pan Y et al (2010) SCFFbx2-E3-ligase-mediated degradation of BACE1 attenuates Alzheimer’s disease amyloidosis and improves synaptic function. Aging Cell 9:1018–1031
Gouras GK, Tsai J, Naslund J et al (2000) Intraneuronal Aβ42 accumulation in human brain. Am J Pathol 156:15–20. https://doi.org/10.1016/S0002-9440(10)64700-1
Gregori L, Hainfeld JF, Simon MN, Goldgaber D (1997) Binding of amyloid β protein to the 20 S proteasome. J Biol Chem 272:58–62
Grundke-Iqbal I, Iqbal K, Tung YC et al (1986) Abnormal phosphorylation of the microtubule-associated protein tau (tau) in Alzheimer cytoskeletal pathology. Proc Natl Acad Sci U S A 83:4913–4917. https://doi.org/10.1073/pnas.83.13.4913
Guo L, Shorter J (2017) Biology and pathobiology of TDP-43 and emergent therapeutic strategies. Cold Spring Harb Perspect Med 7: a024554.
Guo T, Noble W, Hanger DP (2017) Roles of tau protein in health and disease. Acta Neuropathol 133:665–704. https://doi.org/10.1007/s00401-017-1707-9
Haller M, Hock AK, Giampazolias E et al (2014) Ubiquitination and proteasomal degradation of ATG12 regulates its proapoptotic activity. Autophagy 10:2269–2278. https://doi.org/10.4161/15548627.2014.981914
Hansson CA, Frykman S, Farmery MR et al (2004) Nicastrin, presenilin, APH-1, and PEN-2 form active gamma-secretase complexes in mitochondria. J Biol Chem 279:51654–51660. https://doi.org/10.1074/jbc.M404500200
Haque ME, Thomas KJ, D’Souza C et al (2008) Cytoplasmic Pink1 activity protects neurons from dopaminergic neurotoxin MPTP. Proc Natl Acad Sci U S A 105:1716–1721
Hardy J, Selkoe DJ (2002) The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics. Science 297:353–356. https://doi.org/10.1126/science.1072994
Hardy JA, Higgins GA (1992) Alzheimer’s disease: the amyloid cascade hypothesis. Science 256:184–186
He G, Qing H, Cai F et al (2006) Ubiquitin–proteasome pathway mediates degradation of APH-1. J Neurochem 99:1403–1412. https://doi.org/10.1111/j.1471-4159.2006.04184.x
He G, Qing H, Tong Y et al (2007) Degradation of nicastrin involves both proteasome and lysosome. J Neurochem 101:982–992
He Y, Smith R (2009) Nuclear functions of heterogeneous nuclear ribonucleoproteins A/B. Cell Mol Life Sci 66:1239–1256. https://doi.org/10.1007/s00018-008-8532-1
Heo J-M, Ordureau A, Paulo JA et al (2015) The PINK1-PARKIN mitochondrial ubiquitylation pathway drives a program of OPTN/NDP52 recruitment and TBK1 activation to promote mitophagy. Mol Cell 60:7–20
Himmler A, Drechsel D, Kirschner MW, Martin DW (1989) Tau consists of a set of proteins with repeated C-terminal microtubule-binding domains and variable N-terminal domains. Mol Cell Biol 9:1381–1388. https://doi.org/10.1128/mcb.9.4.1381
Hong L, Huang H-C, Jiang Z-F (2014) Relationship between amyloid-beta and the ubiquitin–proteasome system in Alzheimer’s disease. Neurol Res 36:276–282. https://doi.org/10.1179/1743132813Y.0000000288
Hou J, Eldeeb M, Wang X (2017) Beyond deubiquitylation: Usp30-mediated regulation of mitochondrial homeostasis. In: Sun H, Wang X (eds) Mitochondrial DNA and Diseases. Springer, Singapore, pp 133–148
Hu Z-W, Vugmeyster L, Au DF et al (2019) Molecular structure of an N-terminal phosphorylated β-amyloid fibril. Proc Natl Acad Sci U S A 116:11253–11258. https://doi.org/10.1073/pnas.1818530116
Hwang C-S, Shemorry A, Varshavsky A (2010) N-terminal acetylation of cellular proteins creates specific degradation signals. Science 327:973–977
Iijima-Ando K, Sekiya M, Maruko-Otake A et al (2012) Loss of axonal mitochondria promotes Tau-mediated neurodegeneration and Alzheimer’s disease–related Tau phosphorylation Via PAR-1. PLoS Genet 8:e1002918. https://doi.org/10.1371/journal.pgen.1002918
Iqbal K, Grundke-Iqbal I (1991) Ubiquitination and abnormal phosphorylation of paired helical filaments in Alzheimer’s disease. Mol Neurobiol 5:399–410. https://doi.org/10.1007/BF02935561
Ittner LM, Götz J (2011) Amyloid-β and tau - a toxic pas de deux in Alzheimer’s disease. Nat Rev Neurosci 12:67–72. https://doi.org/10.1038/nrn2967
Jarrett JT, Berger EP, Lansbury PT Jr (1993) The carboxy terminus of the b amyloid protein is critical for the seeding of amyloid formation: implications for the pathogenesis of Alzheimer’s disease. Biochemistry 32:4693–4697
Jin SM, Lazarou M, Wang C et al (2010) Mitochondrial membrane potential regulates PINK1 import and proteolytic destabilization by PARL. J Cell Biol 191:933–942
Kageyama S, Sou Y-s, Uemura T et al (2014) Proteasome dysfunction activates autophagy and the Keap1-Nrf2 pathway. J Biol Chem 289:24944–24955
Kasu YAT, Alemu S, Lamari A et al (2018) The N termini of TAR DNA-binding protein 43 (TDP43) C-terminal fragments influence degradation, aggregation propensity, and morphology. Mol Cell Biol 38:e00243-e1218. https://doi.org/10.1128/MCB.00243-18
Kawahara Y, Mieda-Sato A (2012) TDP-43 promotes microRNA biogenesis as a component of the Drosha and Dicer complexes. Proc Natl Acad Sci U S A 109:3347–3352. https://doi.org/10.1073/pnas.1112427109
Kechko OI, Petrushanko IY, Brower CS et al (2019) Beta-amyloid induces apoptosis of neuronal cells by inhibition of the Arg/N-end rule pathway proteolytic activity. Aging (albany NY) 11:6134–6152
Keck S, Nitsch R, Grune T, Ullrich O (2003) Proteasome inhibition by paired helical filament-tau in brains of patients with Alzheimer’s disease. J Neurochem 85:115–122. https://doi.org/10.1046/j.1471-4159.2003.01642.x
Keller JN, Hanni KB, Markesbery WR (2000) Impaired proteasome function in Alzheimer’s disease. J Neurochem 75:436–439. https://doi.org/10.1046/j.1471-4159.2000.0750436.x
Kempf M, Clement A, Faissner A et al (1996) Tau binds to the distal axon early in development of polarity in a microtubule-and microfilament-dependent manner. J Neurosci 16:5583–5592
Kim J-M, Seok O-H, Ju S et al (2018) Formyl-methionine as an N-degron of a eukaryotic N-end rule pathway. Science 362: eaat0174. https://doi.org/10.1126/science.aat0174.
Kim JM (2019) N-terminal formylmethionine as a novel initiator and N-degron of eukaryotic proteins. BMB Rep 52:163–164. https://doi.org/10.5483/BMBRep.2019.52.3.069
Kim S-H, Lah JJ, Thinakaran G et al (2000) Subcellular localization of presenilins: association with a unique membrane pool in cultured cells. Neurobiol Dis 7:99–117
Klionsky DJ, Emr SD (2000) Autophagy as a regulated pathway of cellular degradation. Science 290:1717–1721. https://doi.org/10.1126/science.290.5497.1717
Kondapalli C, Kazlauskaite A, Zhang N et al (2012) PINK1 is activated by mitochondrial membrane potential depolarization and stimulates Parkin E3 ligase activity by phosphorylating Serine 65. Open Biol 2: 120080.
Korolchuk VI, Menzies FM, Rubinsztein DC (2010) Mechanisms of cross-talk between the ubiquitin-proteasome and autophagy-lysosome systems. FEBS Lett 584:1393–1398. https://doi.org/10.1016/j.febslet.2009.12.047
Koyano F, Okatsu K, Kosako H et al (2014) Ubiquitin is phosphorylated by PINK1 to activate parkin. Nature 510:162–166. https://doi.org/10.1038/nature13392
Kramer DA, Eldeeb MA, Wuest M et al (2017) Proteomic characterization of EL4 lymphoma-derived tumors upon chemotherapy treatment reveals potential roles for lysosomes and caspase-6 during tumor cell death in vivo. Proteomics 17:1700060. https://doi.org/10.1002/pmic.201700060
Krecic AM, Swanson MS (1999) hnRNP complexes: composition, structure, and function. Curr Opin Cell Biol 11:363–371. https://doi.org/10.1016/S0955-0674(99)80051-9
Krüger U, Wang Y, Kumar S, Mandelkow E-M (2012) Autophagic degradation of tau in primary neurons and its enhancement by trehalose. Neurobiol Aging 33:2291–2305. https://doi.org/10.1016/j.neurobiolaging.2011.11.009
Kummer MP, Heneka MT (2014) Truncated and modified amyloid-beta species. Alz Res Therapy 6:28. https://doi.org/10.1186/alzrt258
Lambert MP, Barlow AK, Chromy BA et al (1998) Diffusible, nonfibrillar ligands derived from Aβ1–42 are potent central nervous system neurotoxins. Proc Natl Acad Sci U S A 95:6448–6453. https://doi.org/10.1073/pnas.95.11.6448
Lammich S, Kojro E, Postina R et al (1999) Constitutive and regulated α-secretase cleavage of Alzheimer’s amyloid precursor protein by a disintegrin metalloprotease. Proc Natl Acad Sci U S A 96:3922–3927. https://doi.org/10.1073/pnas.96.7.3922
Lasagna-Reeves CA, Castillo-Carranza DL, Guerrero-Muñoz MJ et al (2010) Preparation and characterization of neurotoxic tau oligomers. Biochemistry 49:10039–10041. https://doi.org/10.1021/bi1016233
Lazarou M, Jin SM, Kane LA, Youle RJ (2012) Role of PINK1 binding to the TOM complex and alternate intracellular membranes in recruitment and activation of the E3 ligase Parkin. Dev Cell 22:320–333
Lee EB, Lee VMY, Trojanowski JQ (2012) Gains or losses: molecular mechanisms of TDP43-mediated neurodegeneration. Nat Rev Neurosci 13:38–50. https://doi.org/10.1038/nrn3121
Lee G, Newman ST, Gard DL et al (1998) Tau interacts with src-family non-receptor tyrosine kinases. J Cell Sci 111:3167–3177
Lee KJ, Moussa CEH, Lee Y et al (2010) Beta amyloid-independent role of amyloid precursor protein in generation and maintenance of dendritic spines. Neuroscience 169:344–356. https://doi.org/10.1016/j.neuroscience.2010.04.078
Lee M-S, Kao S-C, Lemere CA et al (2003) APP processing is regulated by cytoplasmic phosphorylation. J Cell Biol 163:83–95. https://doi.org/10.1083/jcb.200301115
Lee VM, Goedert M, Trojanowski JQ (2001) Neurodegenerative tauopathies. Annu Rev Neurosci 24:1121–1159
Leyssen M, Ayaz D, Hébert SS et al (2005) Amyloid precursor protein promotes post-developmental neurite arborization in the Drosophila brain. EMBO J 24:2944–2955. https://doi.org/10.1038/sj.emboj.7600757
Li YR, King OD, Shorter J, Gitler AD (2013) Stress granules as crucibles of ALS pathogenesis. J Cell Biol 201:361–372
Lindwall G, Cole RD (1984) Phosphorylation affects the ability of tau protein to promote microtubule assembly. J Biol Chem 259:5301–5305
Ling S-C, Polymenidou M, Cleveland DW (2013) Converging mechanisms in ALS and FTD: disrupted RNA and protein homeostasis. Neuron 79:416–438
Liu F, Iqbal K, Grundke-Iqbal I et al (2004) O-GlcNAcylation regulates phosphorylation of tau: a mechanism involved in Alzheimer’s disease. Proc Natl Acad Sci U S A 101:10804–10809. https://doi.org/10.1073/pnas.0400348101
López Salon M, Morelli L, Castaño EM et al (2000) Defective ubiquitination of cerebral proteins in Alzheimer’s disease. J Neurosci Res 62:302–310. https://doi.org/10.1002/1097-4547(20001015)62:2%3c302::aid-jnr15%3e3.0.co;2-l
Lopez Salon M, Pasquini L, Besio Moreno M et al (2003) Relationship between β-amyloid degradation and the 26S proteasome in neural cells. Exp Neurol 180:131–143. https://doi.org/10.1016/S0014-4886(02)00060-2
Maas T, Eidenmüller J, Brandt R (2000) Interaction of tau with the neural membrane cortex is regulated by phosphorylation at sites that are modified in paired helical filaments. J Biol Chem 275:15733–15740
Maïo ILD, Barbier P, Allegro D et al (2014) Quantitative analysis of tau-microtubule interaction using FRET. Int J Mol Sci 15:14697–14714
Mandelkow EM, Biernat J, Drewes G et al (1995) Tau domains, phosphorylation, and interactions with microtubules. Neurobiol Aging 16:355–362. https://doi.org/10.1016/0197-4580(95)00025-A
Maraganore DM, De Andrade M, Elbaz A et al (2006) Collaborative analysis of α-synuclein gene promoter variability and Parkinson disease. JAMA 296:661–670
Maroteaux L, Campanelli JT, Scheller RH (1988) Synuclein: a neuron-specific protein localized to the nucleus and presynaptic nerve terminal. J Neurosci 8:2804–2815
Martinez-Contreras R, Cloutier P, Shkreta L et al (2007) hnRNP proteins and splicing control. Adv Exp Med Biol 623:123–147. https://doi.org/10.1007/978-0-387-77374-2_8
Matsuda N, Sato S, Shiba K et al (2010) PINK1 stabilized by mitochondrial depolarization recruits Parkin to damaged mitochondria and activates latent Parkin for mitophagy. J Cell Biol 189:211–221
Mattson MP (2004) Pathways towards and away from Alzheimer’s disease. Nature 430:631–639. https://doi.org/10.1038/nature02621
Menon PK, Koistinen NA, Iverfeldt K, Strom AL (2019) Phosphorylation of the amyloid precursor protein (APP) at Ser-675 promotes APP processing involving meprin b. J Biol Chem 294:17768–17776. https://doi.org/10.1074/jbc.RA119.008310
Mercado PA, Ayala YM, Romano M et al (2005) Depletion of TDP 43 overrides the need for exonic and intronic splicing enhancers in the human apoA-II gene. Nucleic Acids Res 33:6000–6010. https://doi.org/10.1093/nar/gki897
Mietelska-Porowska A, Wasik U, Goras M et al (2014) Tau protein modifications and interactions: their role in function and dysfunction. Int J Mol Sci 15:4671–4713
Mondragón-Rodríguez S, Basurto-Islas G, Santa-Maria I et al (2008) Cleavage and conformational changes of tau protein follow phosphorylation during Alzheimer’s disease. Int J Exp Pathol 89:81–90. https://doi.org/10.1111/j.1365-2613.2007.00568.x
Morais VA, Haddad D, Craessaerts K et al (2014) PINK1 loss-of-function mutations affect mitochondrial complex I activity via NdufA10 ubiquinone uncoupling. Science 344:203–207
Morel E, Chamoun Z, Lasiecka ZM et al (2013) Phosphatidylinositol-3-phosphate regulates sorting and processing of amyloid precursor protein through the endosomal system. Nat Commun 4:2250. https://doi.org/10.1038/ncomms3250
Mueller JC, Fuchs J, Hofer A et al (2005) Multiple regions of α-synuclein are associated with Parkinson’s disease. Ann Neurol 57:535–541
Müller UC, Deller T, Korte M (2017) Not just amyloid: physiological functions of the amyloid precursor protein family. Nat Rev Neurosci 18:281–298. https://doi.org/10.1038/nrn.2017.29
Musiek ES, Holtzman DM (2015) Three dimensions of the amyloid hypothesis: time, space and ‘wingmen.’ Nat Neurosci 18:800–806. https://doi.org/10.1038/nn.4018
Nakielny S, Dreyfuss G (1997) Nuclear export of proteins and RNAs. Curr Opin Cell Biol 9:420–429. https://doi.org/10.1016/S0955-0674(97)80016-6
Nalivaeva NN, Turner AJ (2013) The amyloid precursor protein: a biochemical enigma in brain development, function and disease. FEBS Lett 587:2046–2054. https://doi.org/10.1016/j.febslet.2013.05.010
Narayanan RK, Mangelsdorf M, Panwar A et al (2013) Identification of RNA bound to the TDP-43 ribonucleoprotein complex in the adult mouse brain. Amyotroph Lateral Scler Frontotemporal Degener 14:252–260. https://doi.org/10.3109/21678421.2012.734520
Narendra DP, Jin SM, Tanaka A et al (2010) PINK1 is selectively stabilized on impaired mitochondria to activate Parkin. PLoS Biol 8: e1000298.
Neumann M, Sampathu DM, Kwong LK et al (2006) Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science 314:130–133. https://doi.org/10.1126/science.1134108
Nixon RA (2013) The role of autophagy in neurodegenerative disease. Nat Med 19:983–997. https://doi.org/10.1038/nm.3232
Nouar R, Devred F, Breuzard G, Peyrot V (2013) FRET and FRAP imaging: approaches to characterise tau and stathmin interactions with microtubules in cells. Biol Cell 105:149–161. https://doi.org/10.1111/boc.201200060
Novak M, Kabat J, Wischik CM (1993) Molecular characterization of the minimal protease resistant tau unit of the Alzheimer’s disease paired helical filament. EMBO J 12:365–370. https://doi.org/10.1002/j.1460-2075.1993.tb05665.x
Paravastu AK, Leapman RD, Yau W-M, Tycko R (2008) Molecular structural basis for polymorphism in Alzheimer’s β-amyloid fibrils. Proc Natl Acad Sci U S A 105:18349–18354. https://doi.org/10.1073/pnas.0806270105
Park C, Cuervo AM (2013) Selective autophagy: talking with the UPS. Cell Biochem Biophys 67:3–13. https://doi.org/10.1007/s12013-013-9623-7
Pasternak SH, Bagshaw RD, Guiral M et al (2003) Presenilin-1, nicastrin, amyloid precursor protein, and γ-secretase activity are co-localized in the lysosomal membrane. J Biol Chem 278:26687–26694
Patterson KR, Remmers C, Fu Y et al (2011) Characterization of prefibrillar Tau oligomers in vitro and in Alzheimer disease. J Biol Chem 286:23063–23076
Payton JE, Perrin RJ, Clayton DF, George JM (2001) Protein–protein interactions of alpha-synuclein in brain homogenates and transfected cells. Mol Brain Res 95:138–145
Pearson HA, Peers C (2006) Physiological roles for amyloid β peptides. J Physiol 575:5–10. https://doi.org/10.1113/jphysiol.2006.111203
Picone P, Carrotta R, Montana G et al (2009) Aβ oligomers and fibrillar aggregates induce different apoptotic pathways in LAN5 neuroblastoma cell cultures. Biophys J 96:4200–4211. https://doi.org/10.1016/j.bpj.2008.11.056
Picone P, Nuzzo D, Giacomazza D, Di Carlo M (2020) β-Amyloid peptide: the cell compartment multi-faceted interaction in Alzheimer’s disease. Neurotox Res 37:250–263. https://doi.org/10.1007/s12640-019-00116-9
Plun-Favreau H, Klupsch K, Moisoi N et al (2007) The mitochondrial protease HtrA2 is regulated by Parkinson’s disease-associated kinase PINK1. Nat Cell Biol 9:1243–1252
Polymenidou M, Lagier-Tourenne C, Hutt KR et al (2011) Long pre-mRNA depletion and RNA missplicing contribute to neuronal vulnerability from loss of TDP-43. Nat Neurosci 14:459–468. https://doi.org/10.1038/nn.2779
Prasanth KV, Prasanth SG, Xuan Z et al (2005) Regulating gene expression through RNA nuclear retention. Cell 123:249–263
Rajasekhar K, Chakrabarti M, Govindaraju T (2015) Function and toxicity of amyloid beta and recent therapeutic interventions targeting amyloid beta in Alzheimer’s disease. Chem Commun 51:13434–13450. https://doi.org/10.1039/C5CC05264E
Ramkumar A, Jong BY, Ori-McKenney KM (2018) ReMAPping the microtubule landscape: how phosphorylation dictates the activities of microtubule-associated proteins. Dev Dyn 247:138–155. https://doi.org/10.1002/dvdy.24599
Reddy PH (2011) Abnormal tau, mitochondrial dysfunction, impaired axonal transport of mitochondria, and synaptic deprivation in Alzheimer’s disease. Brain Res 1415:136–148. https://doi.org/10.1016/j.brainres.2011.07.052
Reinhard C, Hébert SS, De Strooper B (2005) The amyloid-β precursor protein: integrating structure with biological function. EMBO J 24:3996–4006. https://doi.org/10.1038/sj.emboj.7600860
Roch JM, Masliah E, Roch-Levecq AC et al (1994) Increase of synaptic density and memory retention by a peptide representing the trophic domain of the amyloid beta/A4 protein precursor. Proc Natl Acad Sci U S A 91:7450–7454. https://doi.org/10.1073/pnas.91.16.7450
Ruan L, Zhou C, Jin E et al (2017) Cytosolic proteostasis through importing of misfolded proteins into mitochondria. Nature 543:443–446. https://doi.org/10.1038/nature21695
Sahara N, Maeda S, Murayama M et al (2007) Assembly of two distinct dimers and higher-order oligomers from full-length tau. Eur J Neurosci 25:3020–3029. https://doi.org/10.1111/j.1460-9568.2007.05555.x
Sambataro F, Pennuto M (2017) Post-translational modifications and protein quality control in motor neuron and polyglutamine diseases. Front Mol Neurosci 10:82. https://doi.org/10.3389/fnmol.2017.00082
Santamaria N, Alhothali M, Alfonso MH et al (2017) Intrinsic disorder in proteins involved in amyotrophic lateral sclerosis. Cell Mol Life Sci 74:1297–1318. https://doi.org/10.1007/s00018-016-2416-6
Schedin-Weiss S, Winblad B, Tjernberg LO (2014) The role of protein glycosylation in Alzheimer disease. FEBS J 281:46–62. https://doi.org/10.1111/febs.12590
Scotter EL, Vance C, Nishimura AL et al (2014) Differential roles of the ubiquitin proteasome system and autophagy in the clearance of soluble and aggregated TDP-43 species. J Cell Sci 127:1263–1278. https://doi.org/10.1242/jcs.140087
Selkoe DJ (1998) The cell biology of β-amyloid precursor protein and presenilin in Alzheimer’s disease. Trends Cell Biol 8:447–453
Shiba K, Arai T, Sato S et al (2009) Parkin stabilizes PINK1 through direct interaction. Biochem Biophys Res Commun 383:331–335
Spillantini MG, Schmidt ML, Lee VM-Y et al (1997) α-Synuclein in Lewy bodies. Nature 388:839
Stefanis L (2012) α-Synuclein in Parkinson’s disease. Cold Spring Harb Perspect Med 2: a009399.
Stefanoska K, Volkerling A, Bertz J et al (2018) An N-terminal motif unique to primate tau enables differential protein–protein interactions. J Biol Chem 293:3710–3719
Stelzmann RA, Norman Schnitzlein H, Reed Murtagh F (1995) An english translation of alzheimer’s 1907 paper, “über eine eigenartige erkankung der hirnrinde.” Clin Anat 8:429–431. https://doi.org/10.1002/ca.980080612
Sung JY, Park SM, Lee C-H et al (2005) Proteolytic cleavage of extracellular secreted α-synuclein via matrix metalloproteinases. J Biol Chem 280:25216–25224
Tai H-C, Serrano-Pozo A, Hashimoto T et al (2012) The synaptic accumulation of hyperphosphorylated tau oligomers in Alzheimer disease is associated with dysfunction of the ubiquitin-proteasome system. Am J Pathol 181:1426–1435
Tanaka S, Shiojiri S, Takahashi Y et al (1989) Tissue-specific expression of three types of β-protein precursor mRNA: enhancement of protease inhibitor-harboring types in Alzheimer’s disease brain. Biochem Biophys Res Commun 165:1406–1414. https://doi.org/10.1016/0006-291X(89)92760-5
Tasaki T, Mulder LCF, Iwamatsu A et al (2005) A Family of mammalian E3 ubiquitin ligases that contain the UBR box motif and recognize N-degrons. Mol Cell Biol 25:7120–7136
Tasaki T, Sriram SM, Park KS, Kwon YT (2012) The N-end rule pathway. Annu Rev Biochem 81:261–289
Tashiro Y, Urushitani M, Inoue H et al (2012) Motor neuron-specific disruption of proteasomes, but not autophagy, replicates amyotrophic lateral sclerosis. J Biol Chem 287:42984–42994. https://doi.org/10.1074/jbc.M112.417600
Thinakaran G, Koo EH (2008) Amyloid precursor protein trafficking, processing, and function. J Biol Chem 283:29615–29619
Timms RT, Zhang Z, Rhee DY et al (2019) A glycine-specific N-degron pathway mediates the quality control of protein N-myristoylation. Science 365: eaaw4912. https://doi.org/10.1126/science.aaw4912.
Tollervey JR, Curk T, Rogelj B et al (2011) Characterizing the RNA targets and position-dependent splicing regulation by TDP-43. Nat Neurosci 14:452–458. https://doi.org/10.1038/nn.2778
Tsvetkov PO, La Rocca R, Malesinski S, Devred F (2019) Characterization of microtubule-associated proteins (MAPs) and tubulin interactions by isothermal titration calorimetry (ITC). In: Ennifar E (ed) Microcalorimetry of biological molecules. Humana Press, New York, NY, Methods in molecular biology, pp 151–165
Turner PR, O’Connor K, Tate WP, Abraham WC (2003) Roles of amyloid precursor protein and its fragments in regulating neural activity, plasticity and memory. Prog Neurobiol 70:1–32. https://doi.org/10.1016/S0301-0082(03)00089-3
Uéda K, Fukushima H, Masliah E et al (1993) Molecular cloning of cDNA encoding an unrecognized component of amyloid in Alzheimer disease. Proc Natl Acad Sci U S A 90:11282–11286
Uversky VN (2015) Intrinsically disordered proteins and their (disordered) proteomes in neurodegenerative disorders. Front Aging Neurosci 7:18. https://doi.org/10.3389/fnagi.2015.00018
Valente EM, Abou-Sleiman PM, Caputo V et al (2004) Hereditary early-onset Parkinson’s disease caused by mutations in PINK1. Science 304:1158–1160
Varshavsky A (2008) Discovery of cellular regulation by protein degradation. J Biol Chem 283:34469–34489
Varshavsky A (2011) The N-end rule pathway and regulation by proteolysis. Protein Sci 20:1298–1345
Varshavsky A (2017) The ubiquitin system, autophagy, and regulated protein degradation. Annu Rev Biochem 86:123–128
Varshavsky A (2019) N-degron and C-degron pathways of protein degradation. Proc Natl Acad Sci U S A 116:358–366
Verma M, Vats A, Taneja V (2015) Toxic species in amyloid disorders: oligomers or mature fibrils. Ann Indian Acad Neurol 18:138–145. https://doi.org/10.4103/0972-2327.144284
Vershinin M, Carter BC, Razafsky DS et al (2007) Multiple-motor based transport and its regulation by Tau. Proc Natl Acad Sci U S A 104:87–92. https://doi.org/10.1073/pnas.0607919104
Viswanathan J, Haapasalo A, Böttcher C et al (2011) Alzheimer’s disease-associated ubiquilin-1 regulates presenilin-1 accumulation and aggresome formation. Traffic 12:330–348. https://doi.org/10.1111/j.1600-0854.2010.01149.x
Voigt A, Berlemann LA, Winklhofer KF (2016) The mitochondrial kinase PINK 1: functions beyond mitophagy. J Neurochem 139:232–239
Walker AK, Tripathy K, Restrepo CR et al (2015) An insoluble frontotemporal lobar degeneration-associated TDP-43 C-terminal fragment causes neurodegeneration and hippocampus pathology in transgenic mice. Hum Mol Genet 24:7241–7254. https://doi.org/10.1093/hmg/ddv424
Wang J-Z, Grundke-Iqbal I, Iqbal K (1996) Glycosylation of microtubule–associated protein tau: an abnormal posttranslational modification in Alzheimer’s disease. Nat Med 2:871–875. https://doi.org/10.1038/nm0896-871
Wang X, Fan H, Ying Z et al (2010) Degradation of TDP-43 and its pathogenic form by autophagy and the ubiquitin-proteasome system. Neurosci Lett 469:112–116. https://doi.org/10.1016/j.neulet.2009.11.055
Wang X, Zhou X, Li G et al (2017) Modifications and trafficking of APP in the pathogenesis of Alzheimer’s disease. Front Mol Neurosci 10:294
Wang Y, Mandelkow E (2012) Degradation of tau protein by autophagy and proteasomal pathways. Biochem Soc Trans 40:644–652. https://doi.org/10.1042/bst20120071
Wang Y, Mandelkow E (2016) Tau in physiology and pathology. Nat Rev Neurosci 17:22–35. https://doi.org/10.1038/nrn.2015.1
Weihofen A, Thomas KJ, Ostaszewski BL et al (2009) Pink1 forms a multiprotein complex with Miro and Milton, linking Pink1 function to mitochondrial trafficking. Biochemistry 48:2045–2052
Weingarten MD, Lockwood AH, Hwo SY, Kirschner MW (1975) A protein factor essential for microtubule assembly. Proc Natl Acad Sci U S A 72:1858–1862. https://doi.org/10.1073/pnas.72.5.1858
Whyte LS, Lau AA, Hemsley KM et al (2017) Endo-lysosomal and autophagic dysfunction: a driving factor in Alzheimer’s disease? J Neurochem 140:703–717. https://doi.org/10.1111/jnc.13935
Winton MJ, Igaz LM, Wong MM et al (2008) Disturbance of nuclear and cytoplasmic TAR DNA-binding protein (TDP-43) induces disease-like redistribution, sequestration, and aggregate formation. J Biol Chem 283:13302–13309
Wischik CM, Novak M, Edwards PC et al (1988) Structural characterization of the core of the paired helical filament of Alzheimer disease. Proc Natl Acad Sci U S A 85:4884–4888. https://doi.org/10.1073/pnas.85.13.4884
Witman GB, Cleveland DW, Weingarten MD, Kirschner MW (1976) Tubulin requires tau for growth onto microtubule initiating sites. Proc Natl Acad Sci U S A 73:4070–4074. https://doi.org/10.1073/pnas.73.11.4070
Wu K-P, Kim S, Fela DA, Baum J (2008) Characterization of conformational and dynamic properties of natively unfolded human and mouse α-synuclein ensembles by NMR: implication for aggregation. J Mol Biol 378:1104–1115
Xiao S, Sanelli T, Dib S et al (2011) RNA targets of TDP-43 identified by UV-CLIP are deregulated in ALS. Mol Cell Neurosci 47:167–180. https://doi.org/10.1016/j.mcn.2011.02.013
Yamano K, Youle RJ (2013) PINK1 is degraded through the N-end rule pathway. Autophagy 9:1758–1769. https://doi.org/10.4161/auto.24633
Yan SD, Chen X, Schmidt AM et al (1994) Glycated tau protein in Alzheimer disease: a mechanism for induction of oxidant stress. Proc Natl Acad Sci U S A 91:7787–7791. https://doi.org/10.1073/pnas.91.16.7787
Yan SD, Yan SF, Chen X et al (1995) Non-enzymatically glycated tau in Alzheimer’s disease induces neuronal oxidant stress resulting in cytokine gene expression and release of amyloid β-peptide. Nat Med 1:693–699. https://doi.org/10.1038/nm0795-693
Yin Y, Wang Y, Gao D et al (2016) Accumulation of human full-length tau induces degradation of nicotinic acetylcholine receptor α4 via activating calpain-2. Sci Rep 6:27283. https://doi.org/10.1038/srep27283
Zare-Shahabadi A, Masliah E, Johnson GV, Rezaei N (2015) Autophagy in Alzheimer’s disease. Rev Neurosci 26:385–395
Zhang YJ, Xu YF, Chen XQ et al (2005) Nitration and oligomerization of tau induced by peroxynitrite inhibit its microtubule-binding activity. FEBS Lett 579:2421–2427. https://doi.org/10.1016/j.febslet.2005.03.041
Zhang Z, Song M, Liu X et al (2014) Cleavage of tau by asparagine endopeptidase mediates the neurofibrillary pathology in Alzheimer’s disease. Nat Med 20:1254–1262. https://doi.org/10.1038/nm.3700
Zhao M, Kim JR, van Bruggen R, Park J (2018) RNA-binding proteins in amyotrophic lateral sclerosis. Mol Cells 41: 818–829. https://doi.org/10.14348/molcells.2018.0243.
Zhao X, Yang J (2010) Amyloid-β peptide is a substrate of the human 20S proteasome. ACS Chem Neurosci 1:655–660
Acknowledgements
MAE is Parkinson Canada Scholar. ‘
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Eldeeb, M.A., Ragheb, M.A., Soliman, M.H. et al. Regulation of Neurodegeneration-associated Protein Fragments by the N-degron Pathways. Neurotox Res 40, 298–318 (2022). https://doi.org/10.1007/s12640-021-00396-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-021-00396-0