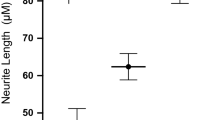
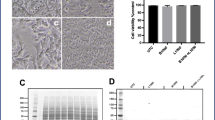
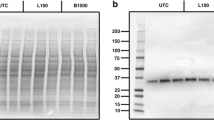
Abstract
l-serine is a naturally occurring dietary amino acid that has recently received renewed attention as a potential therapy for the treatment of amyotrophic lateral sclerosis (ALS), Alzheimer’s disease (AD), hereditary sensory autonomic neuropathy type I (HSAN1), and sleep induction and maintenance. We have previously reported l-serine functions as a competitive inhibitor of l-BMAA toxicity in cell cultures and have since progressed to examine the neuroprotective effects of l-serine independent of l-BMAA-induced neurotoxicity. For example, in a Phase I, FDA-approved human clinical trial of 20 ALS patients, our lab reported 30 g l-serine/day was safe, well-tolerated, and slowed the progression of the disease in a group of 5 patients. Despite increasing evidence for l-serine being useful in the clinic, little is known about the mechanism of action of the observed neuroprotection. We have previously reported, in SH-SY5Y cell cultures, that l-serine alone can dysregulate the unfolded protein response (UPR) and increase the translation of the chaperone protein disulfide isomerase (PDI), and these mechanisms may contribute to the clearance of mis- or unfolded proteins. Here, we further explore the pathways involved in protein clearance when l-serine is present in low and high concentrations in cell culture. We incubated SH-SY5Y cells in the presence and absence of l-serine and measured changes in the activity of proteolytic enzymes from the autophagic-lysosomal system, cathepsin B, cathepsin L, and arylsulfatase and specific activities of the proteasome, peptidylglutamyl-peptide hydrolyzing (PGPH) (also called caspase-like), chymotrypsin, and trypsin-like. Under our conditions, we report that l-serine selectively induced the activity of autophagic-lysosomal enzymes, cathepsins B and L, but not any of the proteasome-hydrolyzing activities. To enable comparison with previous work, we also incubated cells with l-BMAA and report no effect on the activity of the autophagic lysosomes or the proteasomes. We also developed an open-source script for the automation of linear regression calculations of kinetic data. Autophagy impairment or failure is characteristic of many neurodegenerative disease; thus, activation of autophagic-lysosomal proteolysis may contribute to the neuroprotective effect of l-serine, which has been reported in cell culture and human clinical trials.




Similar content being viewed by others
Abbreviations
- AD:
-
Alzheimer’s disease
- ALS:
-
Amyotrophic lateral sclerosis
- ATP:
-
Adenosine 5′-triphosphate
- l-BMAA:
-
Beta-methylamino-l-alanine
- BSA:
-
Bovine serum albumin
- ER:
-
Endoplasmic reticulum
- HSAN1:
-
Hereditary sensory autonomic neuropathy type I
- FBS:
-
Fetal bovine serum
- HD:
-
Huntington’s disease
- LDH:
-
Lactate dehydrogenase
- p-NCS:
-
4-Nitrocatechol sulfate dipotassium salt
- PDI:
-
Protein disulfide isomerase
- PGPH:
-
Peptidylglutamyl-peptide hydrolyzing
- UPR:
-
Unfolded protein response
References
Alfaro IE, Albornoz A, Molina A, Moreno J, Cordero K, Criollo A, Budini M (2018) Chaperone mediated autophagy in the crosstalk of neurodegenerative diseases and metabolic disorders. Front Endocrinol (Lausanne) 9:778. https://doi.org/10.3389/fendo.2018.00778
Alvarez-Erviti L, Rodriguez-Oroz MC, Cooper JM, Caballero C, Ferrer I, Obeso JA, Schapira AHV (2010) Chaperone-mediated autophagy markers in Parkinson disease brains. Arch Neurol 67:1464–1472. https://doi.org/10.1001/archneurol.2010.198
Alves-Rodrigues A, Gregori L, Figueiredo-Pereira ME (1998) Ubiquitin, cellular inclusions and their role in neurodegeneration. Trends Neurosci 21:516–520. https://doi.org/10.1016/S0166-2236(98)01276-4
Berger Z, Ravikumar B, Menzies FM, Oroz LG, Underwood BR, Pangalos MN, Schmitt I, Wullner U, Evert BO, O’Kane CJ, Rubinsztein DC (2006) Rapamycin alleviates toxicity of different aggregate-prone proteins. Hum Mol Genet 15:433–442. https://doi.org/10.1093/hmg/ddi458
Caccamo A, De Pinto V, Messina A, Branca C, Oddo S (2014) Genetic reduction of mammalian target of rapamycin ameliorates Alzheimer’s disease-like cognitive and pathological deficits by restoring hippocampal gene expression signature. J Neurosci 34:7988–7998. https://doi.org/10.1523/JNEUROSCI.0777-14.2014
Cox PA, Davis DA, Mash DC, Metcalf JS, Banack SA (2016) Dietary exposure to an environmental toxin triggers neurofibrillary tangles and amyloid deposits in the brain. Proc Biol Sci 283:20152397. https://doi.org/10.1098/rspb.2015.2397
Cuervo AM, Stefanis L, Fredenburg R, Lansbury PT, Sulzer D (2004) Impaired degradation of mutant α-synuclein by chaperone-mediated autophagy. Science 305:1292–1295
Dunlop RA, Guillemin GJ (2019) The cyanotoxin and non-protein amino acid β-methylamino-l-alanine (l-BMAA) in the food chain: incorporation into proteins and its impact on human health. Neurotox Res 36:601–611. https://doi.org/10.1007/s12640-019-00089-9
Dunlop RA, Dean RT, Rodgers KJ (2008) The impact of specific oxidized amino acids on protein turnover in J774 cells. Biochem J 410:131–140. https://doi.org/10.1042/BJ20070161
Dunlop RA, Cox PA, Banack SA, Rodgers KJ (2013) The non-protein amino acid BMAA is incorporated into human proteins in place of l-serine causing protein misfolding and aggregation. PLoS One 8:e75376. https://doi.org/10.1371/journal.pone.0075376
Dunlop RA, Powell J, Guillemin GJ, Cox PA (2018a) Mechanisms of l-serine neuroprotection in vitro include ER proteostasis regulation. Neurotox Res 33:123–132. https://doi.org/10.1007/s12640-017-9829-3
Dunlop RA, Powell JT, Metcalf JS, Guillemin GJ, Cox PA (2018b) l-serine-mediated neuroprotection includes the upregulation of the ER stress chaperone protein disulfide isomerase (PDI). Neurotox Res 33:113–122. https://doi.org/10.1007/s12640-017-9817-7
Fridman V, Suriyanarayanan S, Novak P, David W, Macklin EA, McKenna-Yasek D, Walsh K, Aziz-Bose R, Oaklander AL, Brown R, Hornemann T, Eichler F (2019) Randomized trial of l-serine in patients with hereditary sensory and autonomic neuropathy type 1. Neurology 92:e359–e370. https://doi.org/10.1212/WNL.0000000000006811
Gao J, Perera G, Bhadbhade M, Halliday GM, Dzamko N (2019) Autophagy activation promotes clearance of α-synuclein inclusions in fibril-seeded human neural cells. J Biol Chem 294:14241–14256. https://doi.org/10.1074/jbc.ra119.008733
Garofalo K, Penno A, Schmidt BP, Lee H-J, Frosch MP, von Eckardstein A, Brown RH, Hornemann T, Eichler FS (2011) Oral l-serine supplementation reduces production of neurotoxic deoxysphingolipids in mice and humans with hereditary sensory autonomic neuropathy type 1. J Clin Invest 121:4735–4745. https://doi.org/10.1172/JCI57549
Holm LJ, Buschard K (2019) L-serine: a neglected amino acid with a potential therapeutic role in diabetes. APMIS 127:655–659. https://doi.org/10.1111/apm.12987
Holm LJ, Haupt-Jorgensen M, Larsen J, Giacobini JD, Bilgin M, Buschard K (2018) L-serine supplementation lowers diabetes incidence and improves blood glucose homeostasis in NOD mice. PLoS One 13:e0194414. https://doi.org/10.1371/journal.pone.0194414
Ito Y, Takahashi S, Shen M, Yamaguchi K, Satoh M (2014) Effects of L-serine on human sleep. Springerplus 3:456. https://doi.org/10.1159/000119485
Kaushik S, Cuervo AM (2015) Proteostasis and aging. Nat Med 21:1406–1415. https://doi.org/10.1038/nm.4001
Kiffin R, Kaushik S, Zeng M, Bandyopadhyay U, Zhang C, Massey AC, Martinez-Vicente M, Cuervo AM (2007) Altered dynamics of the lysosomal receptor for chaperone-mediated autophagy with age. J Cell Sci 120:782–791. https://doi.org/10.1242/jcs.001073
Kim KY, Hwang S-K, Park SY, Kim MJ, Jun DY, Kim YH (2019) l-serine protects mouse hippocampal neuronal HT22 cells against oxidative stress-mediated mitochondrial damage and apoptotic cell death. Free Radic Biol Med 141:447–460. https://doi.org/10.1016/j.freeradbiomed.2019.07.018
Kisselev AF, Goldberg AL (2005) Monitoring activity and inhibition of 26S proteasomes with fluorogenic peptide substrates. Methods Enzymol 398:364–378
Levine TD, Miller RG, Bradley WG, Moore DH, Saperstein DS, Flynn LE, Katz JS, Forshew DA, Metcalf JS, Banack SA, Cox PA (2016) Phase I clinical trial of safety of L-serine for ALS patients. Amyotroph Lateral Scler Front Degener:1–5. https://doi.org/10.1080/21678421.2016.1221971
Lipinski MM, Zheng B, Lu T, Yan Z, Py BF, Ng A, Xavier RJ, Li C, Yankner BA, Scherzer CR, Yuan J (2010) Genome-wide analysis reveals mechanisms modulating autophagy in normal brain aging and in Alzheimer’s disease. Proc Natl Acad Sci U S A 107:14164–14169. https://doi.org/10.1073/pnas.1009485107
Meng L, Mohan R, Kwok BH, Elofsson M, Sin N, Crews CM (1999) Epoxomicin, a potent and selective proteasome inhibitor, exhibits in vivo antiinflammatory activity. Proc Natl Acad Sci U S A 96:10403–10408. https://doi.org/10.1073/pnas.96.18.10403
Metcalf JS, Dunlop RA, Powell JT, Banack SA, Cox PA (2018) L-serine: a naturally-occurring amino acid with therapeutic potential. Neurotox Res 33:213–221. https://doi.org/10.1007/s12640-017-9814-x
Nixon RA, Wegiel J, Kumar A, Yu WH, Peterhoff C, Cataldo A, Cuervo AM (2005) Extensive involvement of autophagy in Alzheimer disease: an immuno-electron microscopy study. J Neuropathol Exp Neurol 64:113–122. https://doi.org/10.1093/jnen/64.2.113
Orange RP, Moore EG (1976) Functional characterization of rat mast cell arylsulfatase activity. J Immunol 117:2191–2196
Perri ER, Thomas CJ, Parakh S, Spencer DM, Atkin JD (2016) The unfolded protein response and the role of protein disulfide isomerase in neurodegeneration. Front Cell Dev Biol 3:80. https://doi.org/10.3389/fcell.2015.00080
Pickford F, Masliah E, Britschgi M, Lucin K, Narasimhan R, Jaeger PA, Small S, Spencer B, Rockenstein E, Levine B, Wyss-Coray T (2008) The autophagy-related protein beclin 1 shows reduced expression in early Alzheimer disease and regulates amyloid β accumulation in mice. J Clin Invest 118:2190–2199. https://doi.org/10.1172/JCI33585
Ravikumar B, Vacher C, Berger Z, Davies JE, Luo S, Oroz LG, Scaravilli F, Easton DF, Duden R, O’Kane CJ, Rubinsztein DC (2004) Inhibition of mTOR induces autophagy and reduces toxicity of polyglutamine expansions in fly and mouse models of Huntington disease. Nat Genet 36:585–595. https://doi.org/10.1038/ng1362
Schneider JL, Cuervo AM (2014) Autophagy and human disease: emerging themes. Curr Opin Genet Dev 26:16–23. https://doi.org/10.1016/j.gde.2014.04.003
Tanaka K (2009) The proteasome: overview of structure and functions. Proc Jpn Acad Ser B Phys Biol Sci 85:12–36. https://doi.org/10.2183/pjab.85.12
Wang Y, Martinez-Vicente M, Krüger U, Kaushik S, Wong E, Mandelkow EM, Cuervo AM, Mandelkow E (2009) Tau fragmentation, aggregation and clearance: the dual role of lysosomal processing. Hum Mol Genet 18:4153–4170. https://doi.org/10.1093/hmg/ddp367
Webb JL, Ravikumar B, Atkins J, Skepper JN, Rubinsztein DC (2003) α-Synuclein is degraded by both autophagy and the proteasome. J Biol Chem 278:25009–25013. https://doi.org/10.1074/jbc.M300227200
Funding
This research was supported by The William Stamps Farish Fund.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Dunlop, R.A., Carney, J.M. Mechanisms of l-Serine-Mediated Neuroprotection Include Selective Activation of Lysosomal Cathepsins B and L. Neurotox Res 39, 17–26 (2021). https://doi.org/10.1007/s12640-020-00168-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-020-00168-2