Abstract
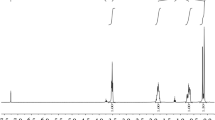
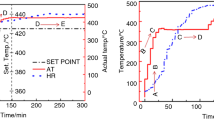
Thermal degradation behavior, exothermic patterns, and kinetic parameters of symmetrically/asymmetrically substituted polysilanes during the kumada thermal rearrangement in the temperature range of 100–450 °C, were investigated applying thermogravimetric (TGA) and differential scanning calorimetric (DSC) techniques. Three kinds of polysilanes, viz., Polydimethylsilane (PDMS), Polydiphenylsilane (PDPS) and Polymethylphenylsilane (PMPS) were synthesized through Wurtz/Wurtz-Fittig polycondensation. As-synthesized polysilanes were then characterized by FT-IR, CHNO, and TGA–DSC techniques. FT-IR spectra confirmed the presence of methyl, phenyl, and a combination of methyl-phenyl functional groups in PDMS, PDPS, and PMPS, respectively. From CHNO analysis, the chemical/empirical formula of PDMS, PDPS, and PMPS was found to be SiC2.01H6.25, SiC12.50H10.26, and SiC6.45H7.76, respectively. TGA results confirmed that PDPS was more thermally stable as compared to PDMS and PMPS. The kinetic parameters of polysilanes were determined by using isoconversional methods such as Ozawa and modified Kissinger–Akahira–Sunose (KAS) methods. The average value of activation energy for PDPS obtained from Ozawa and KAS methods was found to be 1078.49 and 1068.32 kJ/mole, respectively, which is very much higher as compared to that of PDMS and PMPS.
Similar content being viewed by others
Data Availability
No associated data.
References
Miller RD, Michl J (1989) Polysilane high polymers. Chem Rev 89:1359–1410. https://doi.org/10.1021/cr00096a006
Kyushin S, Ichikawa K, Koyama Y, Shiraiwa H, Ichikawa H, Okamura K, Suzuki K (2014) Studies on the detailed structure of poly(dimethylsilylene). Organometallics 33:6298–6304. https://doi.org/10.1021/om500264u
West R (1986) The polysilane high polymers. J Organometal Chem 300:327–346. https://doi.org/10.1016/0022-328X(86)84068-2
Trefonas P, West R, Miller RD (1985) Polysilane high polymers: mechanism of photodegradation. J Am Chem Soc 107:2737–2742. https://doi.org/10.1021/ja00295a028
Tverdokhlebova II, Ronova IA, Men’shov VM, Pertsova NV (1998) Molecular architecture and conformation of macromolecules of novel polysilanes. Russ Chem Bull 4:2348–2351. https://doi.org/10.1007/BF02641532
Grabe N, Michael Rothmann (2017) Method for the synthesis of a chlorine-free, pre-ceramic polymer for the production of ceramic molded bodies. U.S. Patent 9,644,073
Li GY, Li XD, Wang H, Liu L (2010) Long SiC nanowires synthesized from off-gases of the polycarbosilane-derived SiC preparation. Appl Phy 98:293. https://doi.org/10.1007/s00339-009-5426-5
Burkhard AC (1949) Polydimethylsilanes. J Am Chem Soc 71:963–964. https://doi.org/10.1021/ja01171a055
Miller RD, Sooriyakumaran R (1987) The synthesis and characterization of the first soluble, high molecular weight, substituted poly (diphenyl silane) homopolymer. J Polym Sci Part C: Polym Lett 25:321–326. https://doi.org/10.1002/pol.1987.140250803
Lodhe M, Narendra B, Selvam A, Balasubramanian M (2015) Synthesis and characterization of high ceramic yield polycarbosilane precursor for SiC. J Adv Cer 4:307–311. https://doi.org/10.1007/s40145-015-0165-x
Yajima S, Hasegawa Y, Okamura K, Matsuzawa T (1978) Development of high tensile strength silicon carbide fiber using an organosilicon polymer precursor. Nature 273:525–527. https://doi.org/10.1038/273525a0
Noda K, Takehiro S, Issei S, Kouichi M, Dai S, Shun K, Masahiro F, Koji A (2018) Behavior of Si-Si Bond Oxidation by Electron Beam Lithography. J Photopoly Sci Tech 31:581–585. https://doi.org/10.2494/photopolymer.31.581
Cypryk M, Yogendra G, Krzysztof M (1991) Anionic ring-opening polymerization of 1, 2, 3, 4-tetramethyl-1, 2, 3, 4-tetraphenylcyclotetrasilane. J Ame Chem Soc 113:1046–1047. https://doi.org/10.1021/ja00003a050
Jones RG, Ulrich B, Simon JH, William KC (1996) Reappraisal of the origins of the polymodal molecular mass distributions in the formation of poly (methylphenylsilylene) by the Wurtz reductive-coupling reaction. Macromol 29:8036–8046. https://doi.org/10.1021/ma960496y
Kipping FS, James ES (1921) XCIII.—Organic derivatives of silicon. Part XXV. Saturated and unsaturated silicohydrocarbons Si4Ph8. J Chem Soc Trans 119:830–847. https://doi.org/10.1039/CT9211900830
Kipping FS (1924) CCCVIII. —Organic derivatives of silicon. Part XXX. Complex silicohydrocarbons [SiPh2]n. J Chem Soc Trans 125:2291–2297. https://doi.org/10.1039/CT9242502291
Mavric A, Artem B, Mattia F, Matjaz V (2016) Molecular size and solubility conditions of polysilane macromolecules with different topology. Sci Rep 6:1–8. https://doi.org/10.1038/srep35450
Abu-eid MA, King RB, Kotliar AM (1992) Synthesis of polysilane polymer precursors and their pyrolysis to silicon carbides. Eur Polym J 28:315–320. https://doi.org/10.1016/0014-3057(92)90196-9
Lee YJ, Lee JH, Kim SR, Kwon WT, Oh H, Klepeis JH, Teat SJ, Kim YH (2010) Synthesis and characterization of novel pre-ceramic polymer for SiC. J Mater Sci 45:1025–1031. https://doi.org/10.1007/s10853-009-4034-2
Watanabe A, Komatsubara T, Matsuda M, Yoshida Y, Tagawa S (1995) Radical ions of poly(phenylsilyne) and poly(phenylgermyne). Macromol Chem Phys 1101:1229–1240. https://doi.org/10.1002/macp.1995.021960422
Yoshikawa M, Goshi Y, Yamada S, Koga N (2014) Multistep kinetic behavior in the thermal degradation of poly (L-lactic acid): a physico-geometrical kinetic interpretation. J Phys Chem B 118:11397–11405. https://doi.org/10.1021/jp507247x
Dong F, Sun X, Feng S (2016) Thermal degradation kinetics of functional polysiloxanes containing chloromethyl groups. Thermochim Acta 639:14–19. https://doi.org/10.1016/j.tca.2016.07.007
Li L, Wei G, Bin L, Jia-Jia J, Jun-Cheng J, Chi-Min S (2021) Effects of 1-butyl-3-metylimidazolium tetrafluoroborate on the thermal hazard of triacetone triperoxide. Process Saf Environ Prot 149:518–525. https://doi.org/10.1016/j.psep.2020.11.008
Santhana Krishnan G, Naveen S, Shahnawaz M, Ramcharan T (2022) Pyrolysis and thermal degradation studies of silane-carbosilane transformation using hyphenated thermal analysis. J Anal Appl Pyrolysis 164:105535. https://doi.org/10.1016/j.jaap.2022.105535
Shukla SK, Tiwari RK, Ranjan A, Saxena AK, Mathur GN (2004) Some thermal studies on Polysilanes and Polycarbosilanes. Thermochim Acta 424:209–217. https://doi.org/10.1016/j.tca.2004.06.003
Vyazovkin S, Burnham AK, Criado JM, Pérez-Maqueda LA, Popescu C, Sbirrazzuoli N (2011) ICTAC Kinetics Committee recommendations for performing kinetic computations on thermal analysis data. Thermochim Acta 520:1–19. https://doi.org/10.1016/j.tca.2011.03.034
Acknowledgements
The research work was carried out as part of the project under the development of functional ceramic polymer precursors (Grant No M-8-116). Financial support by CSIR-National Aerospace Laboratories, Bangalore, India, is gratefully acknowledged. Authors thank the Director and Head, Materials Science Division, CSIR-National Aerospace Laboratories, Bangalore, for their support and permission to publish this work.
Funding
The research work was carried out as part of the project under the development of functional ceramic polymer precursors (Grant No M-8–116).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design of experiment, polymer synthesis experiments, sample preparation for thermal and spectral characterization, data collection [Shahnawaz, M. and Ramcharan,T] The first draft of the manuscript was written by G Santhana Krishnan and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics Approval
All authors granted ethics approval.
Consent to Participate
We give our consent participate.
Consent for Publication
All the authors are giving consent to publish.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Santhana Krishnan, G., Shahnawaz, M. & Ramcharan, T. Thermal Aspects of Kumada Rearrangement in Symmetrical and Asymmetrical Polysilanes-Interdependence of Molecular Structure, Thermal Degradation, and Stability. Silicon 15, 1499–1510 (2023). https://doi.org/10.1007/s12633-022-02126-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12633-022-02126-3