Abstract
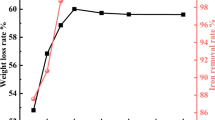
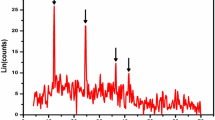
For the gold locked within hematite in the two-stage calcination, which leads to a low gold leaching rate, the present work is aiming at pretreating the calcination to selectively dissolve hematite. The calcination was pretreated by sulfuric acid with cosolvent B. The factors influencing the hematite dissolution rate were studied, and the pretreatment parameters considered were H2SO4 content, B dosage and added time, leaching temperature and time. Simultaneously, mineralogy analysis of the calcination, H2SO4 pretreatment residue and cyanide leaching residue were also carried out. Results indicate that the hematite dissolution rate is quicker, the dissolution temperature is lower and the dissolution time is shorter in the role of cosolvent B than without B. At the same time, the gold locked within hematite is effectively released, so the leaching rate of gold cyanide increases about 10 % than that in actual factory production. The results have an actual significance in guiding industrial production.










Similar content being viewed by others
References
Zhong SP, Wu Z, Huang ZS, Ruan RM. Oxidation kinetics reaction of gold-bearing pyrite in sulphuric acid. Chin J Rare Met. 2013;37(2):295.
Chapman JT, Marchant PB, Lawrence RW, Knopp R. Bio-oxidation of a refractory gold bearing high arsenic sulphide concentrate: a pilot study. FEMS Microbiol Rev. 1993;11(1–3):243.
Amankwah RK, Yen WT, Ramsay JA. A two-stage bacterial pretreatment process for double refractory gold ores. Miner Eng. 2005;18(1):103.
Nanthakumar B, Pickles CA, Kelebek S. Microwave pretreatment of a double refractory gold ore. Miner Eng. 2007;20(11):1109.
Gudyanga FP, Mahlangu T, Roman RJ, Mungoshi J, Mbeve K. An acidic pressure oxidation pretreatment of refractory gold concentrates from the KweKwe Roasting Plant. Miner Eng. 1999;12(8):863.
Lehmann MN, Leary SO, Dunn JG. An evaluation of pretreatments to increase gold recovery from a refractory ore containing arsenopyrite and pyrrhotite. Miner Eng. 2000;13(1):1.
Curreli L, Garbarino C, Ghiani M, Orru G. Arsenic leaching from a gold bearing enargite flotation concentrate. Hydrometallurgy. 2009;96(2):258.
Awe Samuel A, Sandstrom Ake. Selective leaching of arsenic and antimony from a tetrahedrite rich complex sulphide concentrate using alkaline sulphide solution. Miner Eng. 2010;23(15):1227.
Zhang YF, Wu X. Application of two-stage roasting process in gold production. Nonferrous Metall China. 2010;5:37.
Shen KB. Discussion on pretreatment of leaching difficult gold concentrate containing high content of sulfur and arsenic by two stages roasting. Yunnan Chem Technol. 2007;34(5):26.
Zhang YL, Li HM, Yu XJ. Recovery of iron from cyanide tailings with reduction roasting–water leaching followed by magnetic separation. J Hazard Mater. 2012;213–214(30):167.
Syed S. Recovery of gold from secondary sources—a review. Hydrometallurgy. 2012;115–116:30.
Liu BL, Zhang ZH, Li LB, Wang YJ. Recovery of gold and iron from the cyanide tailings by magnetic roasting. Rare Met Mater Eng. 2013;42(9):1805.
Ojeda MW, Perino E, Ruiz MD. Gold extraction by chlorination using a pyrometallurgical process. Miner Eng. 2009;22(4):409.
Zhang FY, Zhang YH, Zu XF. Experimental research on extracting gold and silver from cyanide residue. Gold. 2007;28(9):37.
Acknowledgments
This study was financially supported by the Key Laboratory Project of Education Department in Shaanxi Province (No. 14JS055). The authors would like to appreciate the help from the Key Laboratory for Gold and Resources of Shaanxi, China, during the experimental work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dang, XE., Ke, WS., Tang, C. et al. Increasing leaching rate of gold cyanide of two-stage calcination generated from refractory ore containing arsenopyrite and pyrrhotite. Rare Met. 35, 804–810 (2016). https://doi.org/10.1007/s12598-015-0470-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12598-015-0470-0