Abstract
Introduction
Sevelamer hydrochloride (SH) and lanthanum carbonate (LC) are calcium-free phosphate binders used for the management of hyperphosphatemia in patients with end-stage renal disease (ESRD). The objective of this analysis was to evaluate the real-world dose-relativity between SH and LC monotherapy in US patients with ESRD.
Methods
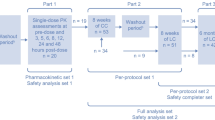
This was a post hoc analysis of a 16-week, real-world study (Vemuri et al. in BMC Nephrol 12:49, 2011) of the efficacy of conversion to LC monotherapy from other phosphate binders. The SH:LC dose-relativity ratio, based on the mean daily dose, was calculated in the subset of patients from the Vemuri study who converted from SH to LC monotherapy and had available SH and LC dose data.
Results
A total of 950 patients converted from SH to LC monotherapy and had recorded dose data. The post hoc analysis population comprised 691 patients with available dose data for both SH at baseline and LC at week 16. The mean (SD) serum phosphate level at baseline was 5.91 (1.66) mg/dL. After conversion to LC monotherapy for 16 weeks, the mean (SD) serum phosphate level was 5.93 (1.85) mg/dL. The mean (SD) daily baseline SH dose was 7,703 (3,642) mg and the mean (SD) daily LC dose at week 16 was 2,800 (939) mg (9.6 versus 2.8 tablets, respectively; P < 0.0001), resulting in a SH:LC dose-relativity ratio of 2.8. The median individual patient SH:LC dose-relativity ratio was 2.6 (95% CI 2.6–2.8). Across baseline SH dose subgroups (2,400–4,800, >4,800–7,200, >7,200–9,600, and >9,600 mg/day), the mean daily SH dose was 4,051, 7,047, 9,253, and 13,150 mg, respectively. In comparison, the mean daily LC dose was 2,445–3,156 mg. Thus, patients requiring baseline SH doses >7,200 mg/day (41% of the analysis population) had higher SH:LC dose-relativity ratios of 3.1–4.2 (median individual patient ratios 3.1–4.0).
Conclusion
In this post hoc analysis of real-world dose-relativity, the overall SH:LC dose-relativity ratio was 2.8 (median individual patient ratio 2.6 (95% CI 2.6–2.8). These findings are consistent with the World Health Organization-defined daily dose and previous studies of the relative phosphate binding capacity of the two drugs. Patients requiring SH doses >7,200 mg/day had higher SH:LC dose-relativities of 3.1–4.2 (median individual patient ratios 3.1–4.0). These findings have implications for the tablet burden and cost-effectiveness of SH and LC in the treatment of hyperphosphatemia.



Similar content being viewed by others
References
Albaaj F, Hutchison A. Hyperphosphatemia in renal failure: causes, consequences and current management. Drugs. 2003;63(6):577–96.
Hruska KA, Mathew S, Lund R, Qiu P, Pratt R. Hyperphosphatemia of chronic kidney disease. Kidney Int. 2008;74(2):148–57.
Block GA, Hulbert-Shearon TE, Levin NW, Port FK. Association of serum phosphorus and calcium x phosphate product with mortality risk in chronic hemodialysis patients: a national study. Am J Kidney Dis. 1998;31(4):607–17.
Block GA, Klassen PS, Lazarus JM, Ofsthun N, Lowrie EG, Chertow GM. Mineral metabolism, mortality, and morbidity in maintenance hemodialysis. J Am Soc Nephrol. 2004;15(8):2208–18.
Menon V, Greene T, Pereira AA, et al. Relationship of phosphorus and calcium–phosphorus product with mortality in CKD. Am J Kidney Dis. 2005;46(3):455–63.
National Kidney Foundation. K/DOQI clinical practice guidelines for bone metabolism and disease in chronic kidney disease. Am J Kidney Dis. 2003;42(4 Suppl 3):S1–201.
Kidney Disease: Improving Global Outcomes (KDIGO) CKD-MBD Work Group. KDIGO clinical practice guideline for the diagnosis, evaluation, prevention, and treatment of Chronic Kidney Disease-Mineral and Bone Disorder (CKD-MBD). Kidney Int Suppl. 2009;113:S1–130.
Navaneethan SD, Palmer SC, Craig JC, Elder GJ, Strippoli GF. Benefits and harms of phosphate binders in CKD: a systematic review of randomized controlled trials. Am J Kidney Dis. 2009;54(4):619–37.
Blacher J, Guerin AP, Pannier B, Marchais SJ, London GM. Arterial calcifications, arterial stiffness, and cardiovascular risk in end-stage renal disease. Hypertension. 2001;38(4):938–42.
Block GA, Raggi P, Bellasi A, Kooienga L, Spiegel DM. Mortality effect of coronary calcification and phosphate binder choice in incident hemodialysis patients. Kidney Int. 2007;71(5):438–41.
Chertow GM, Burke SK, Raggi P. Sevelamer attenuates the progression of coronary and aortic calcification in hemodialysis patients. Kidney Int. 2002;62(1):245–52.
D’Haese PC, Spasovski GB, Sikole A, et al. A multicenter study on the effects of lanthanum carbonate (Fosrenol) and calcium carbonate on renal bone disease in dialysis patients. Kidney Int Suppl. 2003;85:S73–8.
Raggi P, James G, Burke SK, et al. Decrease in thoracic vertebral bone attenuation with calcium-based phosphate binders in hemodialysis. J Bone Miner Res. 2005;20(5):764–72.
Jamal SA, Vandermeer B, Raggi P, et al. Effect of calcium-based versus non-calcium-based phosphate binders on mortality in patients with chronic kidney disease: an updated systematic review and meta-analysis. Lancet. 2013;382:1268–77.
Frazao JM, Adragao T. Non-calcium-containing phosphate binders: comparing efficacy, safety, and other clinical effects. Nephron Clin Pract. 2012;120(2):c108–19.
Hutchison AJ, Smith CP, Brenchley PE. Pharmacology, efficacy and safety of oral phosphate binders. Nat Rev Nephrol. 2011;7(10):578–89.
Iglehart JK. Bundled payment for ESRD—including ESAs in Medicare’s dialysis package. N Engl J Med. 2011;364(7):593–5.
The Senate and House of Representatives of the United States of America. American Taxpayer Relief Act of 2012. Pub.L. 112–240, H.R. 8, 126 Stat. 2313 2013.
Vemuri N, Michelis MF, Matalon A. Conversion to lanthanum carbonate monotherapy effectively controls serum phosphorus with a reduced tablet burden: a multicenter open-label study. BMC Nephrol. 2011;12:49.
Campbell MJ, Gardner MJ. Calculating confidence intervals for some non-parametric analyses. Br Med J (Clin Res Ed). 1988;296(6634):1454–6.
Hochberg Y. A sharper Bonferroni procedure for multiple tests of significance. Biometrika. 1988;75(4):800–2.
WHO Collaborating Centre for Drug Statistics Methodology. Guidelines for ATC classification and DDD assignment. Oslo: Norwegian Institute of Public Health; 2013.
Daugirdas JT, Finn WF, Emmett M, Chertow GM. The phosphate binder equivalent dose. Semin Dial. 2011;24(1):41–9.
Suki WN, Zabaneh R, Cangiano JL, et al. Effects of sevelamer and calcium-based phosphate binders on mortality in hemodialysis patients. Kidney Int. 2007;72(9):1130–7.
Hutchison AJ, Maes B, Vanwalleghem J, et al. Efficacy, tolerability, and safety of lanthanum carbonate in hyperphosphatemia: a 6-month, randomized, comparative trial versus calcium carbonate. Nephron Clin Pract. 2005;100(1):c8–19.
Loghman-Adham M. Medication noncompliance in patients with chronic disease: issues in dialysis and renal transplantation. Am J Manag Care. 2003;9(2):155–71.
Tomasello SDS, Sherman RA. Phosphate binders, K/DOQI guidelines, and compliance: the unfortunate reality. Dial Transplant. 2004;33:236–40.
Chiu YW, Teitelbaum I, Misra M, de Leon EM, Adzize T, Mehrotra R. Pill burden, adherence, hyperphosphatemia, and quality of life in maintenance dialysis patients. Clin J Am Soc Nephrol. 2009;4(6):1089–96.
Curran-Everett D, Milgrom H. Post-hoc data analysis: benefits and limitations. Curr Opin Allergy Clin Immunol. 2013;13(3):223–4.
Acknowledgments
Funding for study design, analysis, and article-processing charges was provided by Shire Development LLC. Writing and editorial support was provided by Fernando Gibson, an employee of PharmaGenesis™ London, with funding from Shire Development LLC. All authors meet the ICMJE criteria for authorship and no deserving authors have been omitted. Rosamund J. Wilson is the guarantor for this article and takes responsibility for the integrity of the work as a whole.
Conflict of interest
Rosamund J. Wilson is a consultant to Shire.
Michael S. Keith is an employee of Shire.
Peter Preston is a consultant to Shire.
J. Brian Copley is an employee of Shire.
Compliance with ethics guidelines
The study (ClinicalTrials.gov Identifier: NCT00160121) was performed in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000 and 2008. All patients provided written, informed consent before being included in the study. The study protocol was approved by an Institutional Review Board at each center.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wilson, R.J., Keith, M.S., Preston, P. et al. The Real-World Dose-Relativity of Sevelamer Hydrochloride and Lanthanum Carbonate Monotherapy in Patients with End-Stage Renal Disease. Adv Ther 30, 1100–1110 (2013). https://doi.org/10.1007/s12325-013-0077-5
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12325-013-0077-5