Abstract
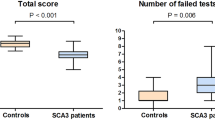
The Cerebellar Cognitive Affective/Schmahmann Syndrome (CCAS) manifests as impaired executive control, linguistic processing, visual spatial function, and affect regulation. The CCAS has been described in the spinocerebellar ataxias (SCAs), but its prevalence is unknown. We analyzed results of the CCAS/Schmahmann Scale (CCAS-S), developed to detect and quantify CCAS, in two natural history studies of 309 individuals Symptomatic for SCA1, SCA2, SCA3, SCA6, SCA7, or SCA8, 26 individuals Pre-symptomatic for SCA1 or SCA3, and 37 Controls. We compared total raw scores, domain scores, and total fail scores between Symptomatic, Pre-symptomatic, and Control cohorts, and between SCA types. We calculated scale sensitivity and selectivity based on CCAS category designation among Symptomatic individuals and Controls, and correlated CCAS-S performance against age and education, and in Symptomatic patients, against genetic repeat length, onset age, disease duration, motor ataxia, depression, and fatigue. Definite CCAS was identified in 46% of the Symptomatic group. False positive rate among Controls was 5.4%. Symptomatic individuals had poorer global CCAS-S performance than Controls, accounting for age and education. The domains of semantic fluency, phonemic fluency, and category switching that tap executive function and linguistic processing consistently separated Symptomatic individuals from Controls. CCAS-S scores correlated most closely with motor ataxia. Controls were similar to Pre-symptomatic individuals whose nearness to symptom onset was unknown. The use of the CCAS-S identifies a high CCAS prevalence in a large cohort of SCA patients, underscoring the utility of the scale and the notion that the CCAS is the third cornerstone of clinical ataxiology.




Similar content being viewed by others
Data Availability
Primary data from the natural history studies (CRC-SCA and READISCA) are housed and curated at the Heath Informatics Institute (HII) at the University of South Florida. Data that serve as the basis for this analysis were downloaded to secure, password-protected Massachusetts General Hospital servers maintained in the Schmahmann Laboratory for Neuroanatomy and Cerebellar Neurobiology. Data are de-identified and available from the senior author upon reasonable request.
References
Schmahmann JD, Sherman JC. The cerebellar cognitive affective syndrome. Brain. 1998;121(4):561–79. https://doi.org/10.1093/brain/121.4.561.
Manto M, Mariën P. Schmahmann’s syndrome - identification of the third cornerstone of clinical ataxiology. Cerebellum Ataxias. 2015;2:2. https://doi.org/10.1186/s40673-015-0023-1.
Levisohn L, Cronin-Golomb A, Schmahmann JD. Neuropsychological consequences of cerebellar tumour resection in children: cerebellar cognitive affective syndrome in a paediatric population. Brain. 2000;123(5):1041–50. https://doi.org/10.1093/brain/123.5.1041.
Albazron FM, Bruss J, Jones RM, et al. Pediatric postoperative cerebellar cognitive affective syndrome follows outflow pathway lesions. Neurology. 2019;93(16):e1561–71. https://doi.org/10.1212/WNL.0000000000008326.
Argyropoulos GPD, van Dun K, Adamaszek M, et al. The cerebellar cognitive affective/Schmahmann syndrome: a task force paper. Cerebellum. 2020;19(1):102–25. https://doi.org/10.1007/s12311-019-01068-8.
Kronemer SI, Slapik MB, Pietrowski JR, et al. Neuropsychiatric symptoms as a reliable phenomenology of cerebellar ataxia. Cerebellum. 2021;20(2):141–50. https://doi.org/10.1007/s12311-020-01195-7.
Moriarty A, Cook A, Hunt H, Adams ME, Cipolotti L, Giunti P. A longitudinal investigation into cognition and disease progression in spinocerebellar ataxia types 1, 2, 3, 6, and 7. Orphanet J Rare Dis. 2016;11(1):82. https://doi.org/10.1186/s13023-016-0447-6.
Olivito G, Cercignani M, Lupo M, et al. Neural substrates of motor and cognitive dysfunctions in SCA2 patients: a network based statistics analysis. NeuroImage: Clinical. 2017;14:719–25. https://doi.org/10.1016/j.nicl.2017.03.009.
Olivito G, Lupo M, Iacobacci C, et al. Structural cerebellar correlates of cognitive functions in spinocerebellar ataxia type 2. J Neurol. 2018;265(3):597–606. https://doi.org/10.1007/s00415-018-8738-6.
Stoodley CJ, MacMore JP, Makris N, Sherman JC, Schmahmann JD. Location of lesion determines motor vs. cognitive consequences in patients with cerebellar stroke. NeuroImage: Clinical. 2016;12:765–75. https://doi.org/10.1016/j.nicl.2016.10.013.
Tedesco AM, Chiricozzi FR, Clausi S, Lupo M, Molinari M, Leggio MG. The cerebellar cognitive profile. Brain. 2011;134(12):3672–86. https://doi.org/10.1093/brain/awr266.
Schmahmann JD. An emerging concept: the cerebellar contribution to higher function. Arch Neurol. 1991;48(11):1178. https://doi.org/10.1001/archneur.1991.00530230086029.
Schmahmann JD. Dysmetria of thought: clinical consequences of cerebellar dysfunction on cognition and affect. Trends Cogn Sci. 1998;2(9):362–71. https://doi.org/10.1016/S1364-6613(98)01218-2.
Schmahmann JD. The role of the cerebellum in affect and psychosis. J Neurolinguistics. 2000;13(2-3):189–214. https://doi.org/10.1016/S0911-6044(00)00011-7.
Schmahmann JD, Guell X, Stoodley CJ, Halko MA. The theory and neuroscience of cerebellar cognition. Annu Rev Neurosci. 2019;42(1):337–64. https://doi.org/10.1146/annurev-neuro-070918-050258.
Shakkottai VG, Fogel BL. Autosomal dominant spinocerebellar ataxia. Neurol Clin. 2013;31(4):987–1007. https://doi.org/10.1016/j.ncl.2013.04.006.
Giocondo F, Curcio G. Spinocerebellar ataxia: a critical review of cognitive and socio-cognitive deficits. Int J Neurosci. 2018;128(2):182–91. https://doi.org/10.1080/00207454.2017.1377198.
Lindsay E, Storey E. Cognitive changes in the spinocerebellar ataxias due to expanded polyglutamine tracts: a survey of the literature. Brain Sci. 2017;7(7):83. https://doi.org/10.3390/brainsci7070083.
Manto M, Lorivel T. Cognitive repercussions of hereditary cerebellar disorders. Cortex. 2011;47(1):81–100. https://doi.org/10.1016/j.cortex.2009.04.012.
Hoche F, Guell X, Vangel MG, Sherman JC, Schmahmann JD. The cerebellar cognitive affective/Schmahmann syndrome scale. Brain. 2018;141(1):248–70. https://doi.org/10.1093/brain/awx317.
Thieme A, Faber J, Sulzer P, et al. The CCAS-scale in hereditary ataxias: helpful on the group level, particularly in SCA3, but limited in individual patients. J Neurol. 2022;1 https://doi.org/10.1007/s00415-022-11071-5.
Maas RPPWM, Killaars S, van de Warrenburg BPC, Schutter DJLG. The cerebellar cognitive affective syndrome scale reveals early neuropsychological deficits in SCA3 patients. J Neurol. 2021;268(9):3456–66. https://doi.org/10.1007/s00415-021-10516-7.
Rodríguez-Labrada R, Batista-Izquierdo A, González-Melix Z, Reynado-Cejas L, Vázquez-Mojena Y, Sanz YA, Canales-Ochoa N, González-Zaldívar Y, Dogan I, Reetz K, Velázquez-Pérez L. Cognitive decline is closely associated with ataxia severity in spinocerebellar ataxia type 2: a validation study of the schmahmann syndrome scale. Cerebellum. 2022;21(3):391–403. https://doi.org/10.1007/s12311-021-01305-z.
Thieme A, Röske S, Faber J, et al. Reference values for the Cerebellar Cognitive Affective Syndrome Scale: age and education matter. Brain. 2021;144(2):e20. https://doi.org/10.1093/brain/awaa417.
Kim DH, Kim R, Lee JY, Lee KM. Clinical, imaging, and laboratory markers of premanifest spinocerebellar ataxia 1, 2, 3, and 6: a systematic review. J Clin Neurol. 2021;17(2):187. https://doi.org/10.3988/jcn.2021.17.2.187.
Lin CC, Ashizawa T, Kuo SH. Collaborative efforts for spinocerebellar ataxia research in the United States: CRC-SCA and READISCA. Front Neurol. 2020;11:902. https://doi.org/10.3389/fneur.2020.00902.
Schmitz-Hubsch T, du Montcel ST, Baliko L, et al. Scale for the assessment and rating of ataxia: development of a new clinical scale. Neurology. 2006;66(11):1717–20. https://doi.org/10.1212/01.wnl.0000219042.60538.92.
Schmahmann JD, Gardner R, MacMore J, Vangel MG. Development of a brief ataxia rating scale (BARS) based on a modified form of the ICARS: Brief Ataxia Rating Scale. Mov Disord. 2009;24(12):1820–8. https://doi.org/10.1002/mds.22681.
Subramony SH, May W, Lynch D, et al. Measuring Friedreich ataxia: interrater reliability of a neurologic rating scale. Neurology. 2005;64(7):1261–2. https://doi.org/10.1212/01.WNL.0000156802.15466.79.
Krupp LB, LaRocca NG, Muir-Nash J, Steinberg AD. The Fatigue Severity Scale: application to patients with multiple sclerosis and systemic lupus erythematosus. Arch Neurol. 1989;46(10):1121. https://doi.org/10.1001/archneur.1989.00520460115022.
Kroenke K, Spitzer RL, Williams JBW. The PHQ-9: validity of a brief depression severity measure. J Gen Intern Med. 2001;16(9):606–13. https://doi.org/10.1046/j.1525-1497.2001.016009606.x.
Selvadurai LP, Perlman SL, Wilmot GR, et al. The S-Factor, a new measure of disease severity in spinocerebellar ataxia: findings and implications. Cerebellum. 2023;22(5):790–809. https://doi.org/10.1007/s12311-022-01424-1.
Mair P, Wilcox R. Robust statistical methods in R using the WRS2 package. Behav Res. 2020;52(2):464–88. https://doi.org/10.3758/s13428-019-01246-w.
Schmahmann JD, Vangel MG, Hoche F, Guell X, Sherman JC. Reply: reference values for the Cerebellar Cognitive Affective Syndrome Scale: age and education matter. Brain. 2021;144(2):e21. https://doi.org/10.1093/brain/awaa419.
Leggio MG. Phonological grouping is specifically affected in cerebellar patients: a verbal fluency study. J Neurol Neurosurg Psychiatry. 2000;69(1):102–6. https://doi.org/10.1136/jnnp.69.1.102.
Richter S, Gerwig M, Aslan B, et al. Cognitive functions in patients with MR-defined chronic focal cerebellar lesions. J Neurol. 2007;254(9):1193–203. https://doi.org/10.1007/s00415-006-0500-9.
Stoodley CJ, Schmahmann JD. The cerebellum and language: evidence from patients with cerebellar degeneration. Brain Lang. 2009;110(3):149–53. https://doi.org/10.1016/j.bandl.2009.07.006.
Baldarçara L, Currie S, Hadjivassiliou M, et al. Consensus paper: radiological biomarkers of cerebellar diseases. Cerebellum. 2015;14(2):175–96. https://doi.org/10.1007/s12311-014-0610-3.
Rüb U, Schöls L, Paulson H, et al. Clinical features, neurogenetics and neuropathology of the polyglutamine spinocerebellar ataxias type 1, 2, 3, 6 and 7. Prog Neurobiol. 2013;104:38–66. https://doi.org/10.1016/j.pneurobio.2013.01.001.
Bürk K, Globas C, Bösch S, et al. Cognitive deficits in spinocerebellar ataxia type 1, 2, and 3. J Neurol. 2003;250(2):207–11. https://doi.org/10.1007/s00415-003-0976-5.
Sokolovsky N, Cook A, Hunt H, Giunti P, Cipolotti L. A preliminary characterisation of cognition and social cognition in spinocerebellar ataxia types 2, 1, and 7. Behav Neurol. 2010;23(1-2):17–29. https://doi.org/10.1155/2010/395045.
Klinke I, Minnerop M, Schmitz-Hübsch T, et al. Neuropsychological features of patients with spinocerebellar ataxia (SCA) types 1, 2, 3, and 6. Cerebellum. 2010;9(3):433–42. https://doi.org/10.1007/s12311-010-0183-8.
Chirino-Pérez A, Marrufo-Meléndez OR, Muñoz-López JI, et al. Mapping the cerebellar cognitive affective syndrome in patients with chronic cerebellar strokes. Cerebellum. 2022;21(2):208–18. https://doi.org/10.1007/s12311-021-01290-3.
Naeije G, Rai M, Allaerts N, Sjogard M, De Tiège X, Pandolfo M. Cerebellar cognitive disorder parallels cerebellar motor symptoms in Friedreich ataxia. Ann Clin Transl Neurol. 2020;7(6):1050–4. https://doi.org/10.1002/acn3.51079.
Brusse E, Brusse-Keizer MGJ, Duivenvoorden HJ, van Swieten JC. Fatigue in spinocerebellar ataxia: patient self-assessment of an early and disabling symptom. Neurology. 2011;76(11):953–9. https://doi.org/10.1212/WNL.0b013e31821043a4.
Martinez ARM, Nunes MB, Faber I, D’Abreu A, Lopes-Cendes Í, França MC. Fatigue and its associated factors in spinocerebellar ataxia type 3/Machado-Joseph disease. Cerebellum. 2017;16(1):118–21. https://doi.org/10.1007/s12311-016-0775-z.
Mastammanavar VS, Kamble N, Yadav R, et al. Non-motor symptoms in patients with autosomal dominant spinocerebellar ataxia. Acta Neurol Scand. 2020;142(4):368–76. https://doi.org/10.1111/ane.13318.
Fancellu R, Paridi D, Tomasello C, et al. Longitudinal study of cognitive and psychiatric functions in spinocerebellar ataxia types 1 and 2. J Neurol. 2013;260(12):3134–43. https://doi.org/10.1007/s00415-013-7138-1.
Acknowledgements
We are grateful to the patients and families who participate in the natural history studies and the coordinators at the participating sites who administer these studies together with their site principal investigators. The assistance of Jason MacMore in this project is gratefully acknowledged.
Funding
Supported in part by the National Ataxia Foundation, the Gordon and Marilyn Macklin Foundation, the MINDlink Foundation, and the National Institute of Neurological Disorders and Stroke (NINDS) grant U01 NS104326 for the READISCA project (NCT03487367). L.P.S. was funded by an Australian-US Fulbright Commission scholarship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics Approval
Approval was obtained for study procedures at all sites via the local IRB at each institution, and all participants gave informed consent. The central sites (UCLA for CRC-SCA and Houston Methodist for READISCA) prepared the original IRB documents that were used by all participating sites during their local reviews.
Conflict of Interest
L.P.S. was funded by an Australian-American Fulbright Commission scholarship.
C.U.O. receives research support from Alector Inc. and Transposon Inc., and consults for Acadia Pharmaceuticals. He is also supported by the Robert and Nancy Hall for Brain Research, and the Joseph Trovato Fund.
L.S.R. is site PI for Biohaven Pharma and has consulted for Bial Biotech and Reata Pharmaceuticals. She receives research support from NINDS, Pfizer, the National Ataxia Foundation, Gordon and Marilyn Macklin Foundation, and the Daniel B and Florence E. Green Foundation.
M.D.G is site PI for Biohaven Pharma, receives funding from the NIH/NIA (R01 AG AG031189; R01AG062562; R56 AG055619) and is supported by the Michael J. Homer Family Fund.
J.D.S. is site PI for Biohaven Pharma, consults for Biohaven, and holds the copyright with the General Hospital Corporation to the Brief Ataxia Rating scale, Patient Reported outcome Measure of Ataxia, the Cerebellar Cognitive Affective / Schmahmann Syndrome Scale, and the Cerebellar Neuropsychiatric Rating Scale.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Selvadurai, L.P., Perlman, S.L., Ashizawa, T. et al. The Cerebellar Cognitive Affective/Schmahmann Syndrome Scale in Spinocerebellar Ataxias. Cerebellum (2024). https://doi.org/10.1007/s12311-023-01651-0
Accepted:
Published:
DOI: https://doi.org/10.1007/s12311-023-01651-0