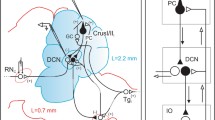
Abstract
Cerebellar Purkinje cells (PCs) encode afferent information in the rate and temporal structure of their spike trains. Both spontaneous firing in these neurons and its modulation by synaptic inputs depend on Ca2+ current carried by Cav2.1 (P/Q) type channels. Previous studies have described how loss-of-function Cav2.1 mutations affect intrinsic excitability and excitatory transmission in PCs. This study examines the effects of the leaner mutation on fast GABAergic transmission and its modulation of spontaneous firing in PCs. The leaner mutation enhances spontaneous synaptic inhibition of PCs, leading to transitory reductions in PC firing rate and increased spike rate variability. Enhanced inhibition is paralleled by an increase in the frequency and amplitude of spontaneous inhibitory postsynaptic currents (sIPSCs) measured under voltage clamp. These differences are abolished by tetrodotoxin, implicating effects of the mutation on spike-induced GABA release. Elevated sIPSC frequency in leaner PCs is not accompanied by increased mean firing rate in molecular layer interneurons, but IPSCs evoked in PCs by direct stimulation of these neurons exhibit larger amplitude, slower decay rate, and a higher burst probability compared to wild-type PCs. Ca2+ release from internal stores appears to be required for enhanced inhibition since differences in sIPSC frequency and amplitude in leaner and wild-type PCs are abolished by thapsigargin, an ER Ca2+ pump inhibitor. These findings represent the first account of the functional consequences of a loss-of-function P/Q channel mutation on PC firing properties through altered GABAergic transmission. Gain in synaptic inhibition shown here would compromise the fidelity of information coding in these neurons and may contribute to impaired cerebellar function resulting from loss-of function mutations in the CaV2.1 channel gene.







Similar content being viewed by others
Abbreviations
- PC:
-
Purkinje cell
- BC:
-
Basket cell
- IPSC:
-
Inhibitory postsynaptic current
- sIPSC:
-
Spontaneous IPSC
- eIPSC:
-
Evoked IPSC
- mIPSC:
-
Miniature IPSC
- TTX:
-
Tetrodotoxin
- SERCA:
-
Sarcoplasmic/endoplasmic reticulum Ca/ATPase
- ISI:
-
Inter-spike interval
- IEI:
-
Inter-event interval
- CV:
-
Coefficient of variation
- CICR:
-
Calcium-induced calcium release
- [Ca2+]i :
-
Cytosolic free calcium concentration
- WT:
-
Wild type
- tgla :
-
Leaner
References
Ito M. The cerebellum and neural control. New York: Raven; 1984.
Jaeger D, Bower JM. Prolonged responses in rat cerebellar Purkinje cells following activation of the granule cell layer: an intracellular in vitro and in vivo investigation. Exp Brain Res. 1994;100(2):200–14.
Hausser M, Clark BA. Tonic synaptic inhibition modulates neuronal output pattern and spatiotemporal synaptic integration. Neuron. 1997;19(3):665–78.
De Schutter E, Steuber V. Patterns and pauses in Purkinje cell simple spike trains: experiments, modeling and theory. Neuroscience. 2009;162(3):816–26.
Llinas R, Sugimori M. Electrophysiological properties of in vitro Purkinje cell somata in mammalian cerebellar slices. J Physiol. 1980;305:171–95.
Llinas R et al. Blocking and isolation of a calcium channel from neurons in mammals and cephalopods utilizing a toxin fraction (FTX) from funnel-web spider poison. Proc Natl Acad Sci USA. 1989;86(5):1689–93.
Jaeger D, De Schutter E, Bower JM. The role of synaptic and voltage-gated currents in the control of Purkinje cell spiking: a modeling study. J Neurosci. 1997;17(1):91–106.
Raman IM, Bean BP. Ionic currents underlying spontaneous action potentials in isolated cerebellar Purkinje neurons. J Neurosci. 1999;19(5):1663–74.
Diener HC et al. Cerebellar dysfunction of movement and perception. Can J Neurol Sci. 1993;20 Suppl 3:S62–9.
Ivry RB, Keele SW, Diener HC. Dissociation of the lateral and medial cerebellum in movement timing and movement execution. Exp Brain Res. 1988;73(1):167–80.
Braitenberg V. Is the cerebellar cortex a biological clock in the millisecond range? Prog Brain Res. 1967;25:334–46.
Zhuchenko O et al. Autosomal dominant cerebellar ataxia (SCA6) associated with small polyglutamine expansions in the alpha 1A-voltage-dependent calcium channel. Nat Genet. 1997;15(1):62–9.
Ophoff RA et al. Familial hemiplegic migraine and episodic ataxia type-2 are caused by mutations in the Ca2+ channel gene CACNL1A4. Cell. 1996;87(3):543–52.
Fletcher CF et al. Absence epilepsy in tottering mutant mice is associated with calcium channel defects. Cell. 1996;87(4):607–17.
Harrington DL et al. Does the representation of time depend on the cerebellum? Effect of cerebellar stroke. Brain. 2004;127(Pt 3):561–74.
Timmann D, Watts S, Hore J. Failure of cerebellar patients to time finger opening precisely causes ball high-low inaccuracy in overarm throws. J Neurophysiol. 1999;82(1):103–14.
Welsh JP, Llinas R. Some organizing principles for the control of movement based on olivocerebellar physiology. Prog Brain Res. 1997;114:449–61.
Marr D. A theory of cerebellar cortex. J Physiol. 1969;202(2):437–70.
Braitenberg V, Atwood RP. Morphological observations on the cerebellar cortex. J Comp Neurol. 1958;109(1):1–33.
Mittmann W, Koch U, Hausser M. Feed-forward inhibition shapes the spike output of cerebellar Purkinje cells. J Physiol. 2005;563(Pt 2):369–78.
Jaeger D, Bower JM. Synaptic control of spiking in cerebellar Purkinje cells: dynamic current clamp based on model conductances. J Neurosci. 1999;19(14):6090–101.
Hoebeek FE et al. Increased noise level of Purkinje cell activities minimizes impact of their modulation during sensorimotor control. Neuron. 2005;45(6):953–65.
Walter JT et al. Decreases in the precision of Purkinje cell pacemaking cause cerebellar dysfunction and ataxia. Nat Neurosci. 2006;9(3):389–97.
Pietrobon D. Ca(V) 2.1 channnelopathies. Pflungers Archiv–European Journal of Physiology. 2010;460(2):375–93.
Mori Y et al. Reduced voltage sensitivity of activation of P/Q-type Ca2+ channels is associated with the ataxic mouse mutation rolling Nagoya (tg(rol)). J Neurosci. 2000;20(15):5654–62.
Matsushita K et al. Bidirectional alterations in cerebellar synaptic transmission of tottering and rolling Ca2+ channel mutant mice. J Neurosci. 2002;22(11):4388–98.
Liu S, Friel DD. Impact of the leaner P/Q-type Ca2+ channel mutation on excitatory synaptic transmission in cerebellar Purkinje cells. J Physiol. 2008;586(Pt 18):4501–15.
Ovsepian SV, Friel DD. The leaner P/Q-type calcium channel mutation renders cerebellar Purkinje neurons hyper-excitable and eliminates Ca2+–Na+ spike bursts. Eur J Neurosci. 2008;27(1):93–103.
Zhou YD, Turner TJ, Dunlap K. Enhanced G protein-dependent modulation of excitatory synaptic transmission in the cerebellum of the Ca2+ channel-mutant mouse, tottering. J Physiol. 2003;547(Pt 2):497–507.
Dove LS, Abbott LC, Griffith WH. Whole-cell and single-channel analysis of P-type calcium currents in cerebellar Purkinje cells of leaner mutant mice. J Neurosci. 1998;18(19):7687–99.
Lorenzon NM et al. Altered calcium channel currents in Purkinje cells of the neurological mutant mouse leaner. J Neurosci. 1998;18(12):4482–9.
Wakamori M et al. Single tottering mutations responsible for the neuropathic phenotype of the P-type calcium channel. J Biol Chem. 1998;273(52):34857–67.
Andersen P, Eccles JC, Voorhoeve PE. Postsynaptic inhibition of cerebellar Purkinje cells. J Neurophysiol. 1964;27:1138–53.
Chan-Palay V. The recurrent collaterals of Purkinje cell axons: a correlated study of the rat’s cerebellar cortex with electron microscopy and the Golgi method. Z Anat Entwicklungsgesch. 1971;134(2):200–34.
Eccles JC, Llinas R, Sasaki K. The inhibitory interneurones within the cerebellar cortex. Exp Brain Res. 1966;1(1):1–16.
Midtgaard J. Stellate cell inhibition of Purkinje cells in the turtle cerebellum in vitro. J Physiol. 1992;457:355–67.
Orduz D, Llano I. Recurrent axon collaterals underlie facilitating synapses between cerebellar Purkinje cells. Proc Natl Acad Sci USA. 2007;104(45):17831–6.
Forti L, Pouzat C, Llano I. Action potential-evoked Ca2+ signals and calcium channels in axons of developing rat cerebellar interneurones. J Physiol. 2000;527(Pt 1):33–48.
Stephens GJ et al. The Cav2.1/alpha1A (P/Q-type) voltage-dependent calcium channel mediates inhibitory neurotransmission onto mouse cerebellar Purkinje cells. Eur J Neurosci. 2001;13(10):1902–12.
Herrup K, Wilczynski SL. Cerebellar cell degeneration in the leaner mutant mouse. Neuroscience. 1982;7(9):2185–96.
Sidman RL, Green MC, Appel SH. Catalog of the neurological mutants of the mouse. Cambridge: Harvard University Press; 1965.
Vincent P, Marty A. Fluctuations of inhibitory postsynaptic currents in Purkinje cells from rat cerebellar slices. J Physiol. 1996;494(Pt 1):183–99.
Stone TW. Neuropharmacology of quinolinic and kynurenic acids. Pharmacol Rev. 1993;45(3):309–79.
Allen C, Stevens CF. An evaluation of causes for unreliability of synaptic transmission. Proc Natl Acad Sci USA. 1994;91(22):10380–3.
McKay BE, Turner RW. Physiological and morphological development of the rat cerebellar Purkinje cell. J Physiol. 2005;567(Pt 3):829–50.
Womack M, Khodakhah K. Active contribution of dendrites to the tonic and trimodal patterns of activity in cerebellar Purkinje neurons. J Neurosci. 2002;22(24):10603–12.
Rokni D et al. Regularity, variability and bi-stability in the activity of cerebellar purkinje cells. Front Cell Neurosci. 2009;3:12.
Galante M, Marty A. Presynaptic ryanodine-sensitive calcium stores contribute to evoked neurotransmitter release at the basket cell–Purkinje cell synapse. J Neurosci. 2003;23(35):11229–34.
Conti R, Tan YP, Llano I. Action potential-evoked and ryanodine-sensitive spontaneous Ca2+ transients at the presynaptic terminal of a developing CNS inhibitory synapse. J Neurosci. 2004;24(31):6946–57.
Bouchard R, Pattarini R, Geiger JD. Presence and functional significance of presynaptic ryanodine receptors. Prog Neurobiol. 2003;69(6):391–418.
Collin T, Marty A, Llano I. Presynaptic calcium stores and synaptic transmission. Curr Opin Neurobiol. 2005;15(3):275–81.
Fierro L, DiPolo R, Llano I. Intracellular calcium clearance in Purkinje cell somata from rat cerebellar slices. J Physiol. 1998;510(Pt 2):499–512.
Carter AG et al. Assessing the role of calcium-induced calcium release in short-term presynaptic plasticity at excitatory central synapses. J Neurosci. 2002;22(1):21–8.
Hongpaisan J et al. Multiple modes of calcium-induced calcium release in sympathetic neurons II: a [Ca2+](i)- and location-dependent transition from endoplasmic reticulum Ca accumulation to net Ca release. J Gen Physiol. 2001;118(1):101–12.
Thastrup O et al. Thapsigargin, a tumor promoter, discharges intracellular Ca2+ stores by specific inhibition of the endoplasmic reticulum Ca2(+)-ATPase. Proc Natl Acad Sci USA. 1990;87(7):2466–70.
Gillard SE et al. Identification of pore-forming subunit of P-type calcium channels: an antisense study on rat cerebellar Purkinje cells in culture. Neuropharmacology. 1997;36(3):405–9.
Walter JT, Khodakhah K. The linear computational algorithm of cerebellar Purkinje cells. J Neurosci. 2006;26(50):12861–72.
Sakaba T. Two Ca(2+)-dependent steps controlling synaptic vesicle fusion and replenishment at the cerebellar basket cell terminal. Neuron. 2008;57(3):406–19.
Caddick SJ et al. Excitatory but not inhibitory synaptic transmission is reduced in lethargic (Cacnb4(lh)) and tottering (Cacna1atg) mouse thalami. J Neurophysiol. 1999;81(5):2066–74.
Jun K et al. Ablation of P/Q-type Ca(2+) channel currents, altered synaptic transmission, and progressive ataxia in mice lacking the alpha(1A)-subunit. Proc Natl Acad Sci USA. 1999;96(26):15245–50.
Qian J, Noebels JL. Presynaptic Ca(2+) influx at a mouse central synapse with Ca(2+) channel subunit mutations. J Neurosci. 2000;20(1):163–70.
Dove LS et al. Altered calcium homeostasis in cerebellar Purkinje cells of leaner mutant mice. J Neurophysiol. 2000;84(1):513–24.
Murchison D et al. Homeostatic compensation maintains Ca2+ signaling functions in Purkinje neurons in the leaner mutant mouse. Cerebellum. 2002;1(2):119–27.
Collin T et al. Developmental changes in parvalbumin regulate presynaptic Ca2+ signaling. J Neurosci. 2005;25(1):96–107.
Shmigol A, Kostyuk P, Verkhratsky A. Dual action of thapsigargin on calcium mobilization in sensory neurons: inhibition of Ca2+ uptake by caffeine-sensitive pools and blockade of plasmalemmal Ca2+ channels. Neuroscience. 1995;65(4):1109–18.
Llano I et al. Presynaptic calcium stores underlie large-amplitude miniature IPSCs and spontaneous calcium transients. Nat Neurosci. 2000;3(12):1256–65.
Palay SL, Chan-Palay V. Cerebellar cortex: cytology and organization. New York: Springer; 1974.
Albrecht MA et al. Multiple modes of calcium-induced calcium release in sympathetic neurons I: attenuation of endoplasmic reticulum Ca2+ accumulation at low [Ca2+](i) during weak depolarization. J Gen Physiol. 2001;118(1):83–100.
Zhang CL, Messing A, Chiu SY. Specific alteration of spontaneous GABAergic inhibition in cerebellar purkinje cells in mice lacking the potassium channel Kv1.1. J Neurosci. 1999;19(8):2852–64.
Fortier PA, Smith AM, Kalaska JF. Comparison of cerebellar and motor cortex activity during reaching: directional tuning and response variability. J Neurophysiol. 1993;69(4):1136–49.
Krauzlis RJ, Lisberger SG. Directional organization of eye movement and visual signals in the floccular lobe of the monkey cerebellum. Exp Brain Res. 1996;109(2):289–302.
Mano N, Ito Y, Shibutani H. Saccade-related Purkinje cells in the cerebellar hemispheres of the monkey. Exp Brain Res. 1991;84(3):465–70.
de Solages C et al. High-frequency organization and synchrony of activity in the purkinje cell layer of the cerebellum. Neuron. 2008;58(5):775–88.
Vincent P, Marty A. Neighboring cerebellar Purkinje cells communicate via retrograde inhibition of common presynaptic interneurons. Neuron. 1993;11(5):885–93.
Chacron MJ, Pakdaman K, Longtin A. Interspike interval correlations, memory, adaptation, and refractoriness in a leaky integrate-and-fire model with threshold fatigue. Neural Comput. 2003;15(2):253–78.
Callaway JC, Lasser-Ross N, Ross WN. IPSPs strongly inhibit climbing fiber-activated [Ca2+]i increases in the dendrites of cerebellar Purkinje neurons. J Neurosci. 1995;15(4):2777–87.
Acknowledgments
David Friel would like to thank Dr. Roberto F. Galán for discussions regarding sIPSC burst statistics. This work was supported by a grant from NIH/NINDS (NS 33514) to DDF.
Conflict of interest
The authors report no conflicts of interest regarding the contents of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Supplementary Fig. 1
Comparison between tonic firing in PCs under whole cell and cell-attached recording conditions. a, b Representative examples of spontaneous tonic firing activity recorded in wild-type (a) and leaner (b) PCs under cell-attached (top) and whole cell (bottom) recording conditions. Note the pronounced irregular spiking of leaner PCs under both experimental conditions, in contrast to the more regular firing of WT PCs. c, d Comparison between measurements of firing rate and CV in WT and leaner PCs obtained under cell-attached (CA, WT: n = 24; leaner: n = 13) and whole cell (WC, wild type: n = 18; leaner: n = 15) recording conditions. The similar values for firing rate and CV obtained under cell-attached and whole cell conditions supports the use of both experimental approaches for evaluating PC spike activity in slices. e, f A typical cell-attached recording of spontaneous spiking from WT PC and after the transition to whole cell recording mode (black arrow), during which firing rate and CV remained approximately constant. f Collected results from three WT PCs in which recordings we made as in e (JPEG 623 kb)
Supplementary Fig. 2
Detection and analysis of sIPSCs. a Representative current recording from a leaner PC under voltage clamp at −75 mV in the presence of kynurenic acid (5 mM) illustrating spontaneous IPSCs. Dashed line indicates baseline holding current. b Negative derivative of the current trace in a. Dashed line indicates zero and dotted line shows the threshold used for identifying synaptic currents. The threshold was defined as 2.5 times the standard deviation of −dI/dt during a manually defined time interval that lacked obvious synaptic currents (see horizontal bar above trace in a and corresponding values of −dI/dt in b). c Normalized distribution of −dI/dt during the event-free period illustrated in a. Mean value differs from zero due to the small non-zero current slope during the event-free time interval in a. d Normalized distribution of inter-event intervals from the same cell. This distribution could be approximated by a single exponential function, consistent with a homogeneous Poisson process. Mean sIPSC frequency in this cell was 50.2 Hz. Sample interval, 50 μs (JPEG 278 kb)
Supplementary Fig. 3
Variability of firing rate and CV measurements in PCs and molecular layer interneurons. a Distribution of firing rate (top) and interspike interval CV (bottom) under whole cell current-clamp conditions in WT and leaner Purkinje neurons before (left) and after (right) exposure to kynurenic acid (5 mM) and picrotoxin (200 μM) to block spontaneous synaptic currents. Measurements were from 19 WT and 16 leaner PCs (pre-block) and 18 WT and 12 leaner cells (post-block). b Distribution of firing rate (top) and CV (bottom) in molecular layer interneurons under cell-attached recording conditions. Measurements were from 13 WT to 7 leaner basket neurons. In each panel, horizontal bars indicate mean values (JPEG 381 kb)
Rights and permissions
About this article
Cite this article
Ovsepian, S.V., Friel, D.D. Enhanced Synaptic Inhibition Disrupts the Efferent Code of Cerebellar Purkinje Neurons in Leaner Cav2.1 Ca2+ Channel Mutant Mice. Cerebellum 11, 666–680 (2012). https://doi.org/10.1007/s12311-010-0210-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12311-010-0210-9