Abstract
Background
It is well known that adjuvant tamoxifen treatment for breast cancer in postmenopausal women decreased bone loss. However, the effects of adjuvant tamoxifen therapy on bone mineral density (BMD) in premenopausal patients with breast cancer remains uncertain. Tamoxifen would have a potential impact of premenopausal BMD on health. The aim of this meta-analysis was to assess this in premenopausal women with primary breast cancer.
Methods
Through April 2020, studies reporting BMD changes of lumbar spine or hip in premenopausal women with primary breast cancer treated with adjuvant tamoxifen and tamoxifen plus chemotherapy or ovarian function suppression (OFS) were collected from EMBASE and PubMed. The meta-analysis was performed using random effects model of the standardized mean difference (SMD) of BMD in patients.
Results
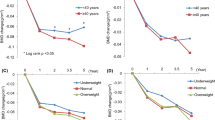
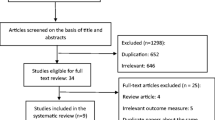
A total of 1432 premenopausal patients were enrolled in eight studies, involving 198 patients treated with tamoxifen alone in three studies. After a 3-year median follow-up, adjuvant tamoxifen decreased the lumbar spinal and hip BMD by as much as an SMD of −1.17 [95% confidence interval (CI); −1.58 to −0.76)] and −0.66 (95% CI, −1.55 to 0.23), respectively. In subgroup analysis in patients treated adjuvant tamoxifen and tamoxifen plus chemotherapy or OFS according to follow-up duration, the bone change of < 3 years follow-up group was −0.03 SMD (95% CI, −0.47 to 0.41) and that of ≥ 3 years follow-up group was −1.06 SMD (95% CI, −1.48 to −0.64). Compared with patients who received tamoxifen alone, patients who received combination therapy with chemotherapy or OFS showed lesser bone loss at the lumbar spine.
Conclusions
Our meta-analysis demonstrated that adjuvant tamoxifen therapy in premenopausal patients caused bone loss after 3 years of follow-up, especially at the lumbar spines. For a definite evaluation of the adverse effects of tamoxifen on bone, it is necessary to accumulate more relevant studies.




Similar content being viewed by others
Availability of data and materials
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Abbreviations
- BMD:
-
Bone mineral density
- CI:
-
Confidence interval
- OFS:
-
Ovarian function suppression
- SMD:
-
Standardized mean difference
References
Eifel P, et al. National Institutes of Health Consensus Development Conference Statement: adjuvant therapy for breast cancer, November 1–3, 2000. J Natl Cancer Inst. 2001;93:979–89. https://doi.org/10.1093/jnci/93.13.979.
Maximov PY, Lee TM, Jordan VC. The discovery and development of selective estrogen receptor modulators (SERMs) for clinical practice. Curr Clin Pharmacol. 2013;8:135–55. https://doi.org/10.2174/1574884711308020006.
Early Breast Cancer Trialists Collaborative Group. Tamoxifen for early breast cancer: an overview of the randomised trials. Lancet. 1998;351:1451–67.
Davies C, et al. Relevance of breast cancer hormone receptors and other factors to the efficacy of adjuvant tamoxifen: patient-level meta-analysis of randomised trials. Lancet. 2011;378:771–84. https://doi.org/10.1016/s0140-6736(11)60993-8.
Gray R, et al. aTTom: long-term effects of continuing adjuvant tamoxifen to 10 years versus stopping at 5 years in 6953 women with early breast cancer. J Clin Oncol. 2013. https://doi.org/10.1200/jco.2013.31.18_suppl.5.
Davies C, et al. Long-term effects of continuing adjuvant tamoxifen to 10 years versus stopping at 5 years after diagnosis of oestrogen receptor-positive breast cancer: ATLAS, a randomised trial. Lancet. 2013;381:805–16. https://doi.org/10.1016/s0140-6736(12)61963-1.
Ali S, et al. Molecular mechanisms and mode of tamoxifen resistance in breast cancer. Bioinformation. 2016;12:135–9. https://doi.org/10.6026/97320630012135.
Osborne CK. Tamoxifen in the treatment of breast cancer. N Engl J Med. 1998;339:1609–18. https://doi.org/10.1056/nejm199811263392207.
O’Regan RM, Jordan VC. The evolution of tamoxifen therapy in breast cancer: selective oestrogen-receptor modulators and downregulators. Lancet Oncol. 2002;3:207–14. https://doi.org/10.1016/s1470-2045(02)00711-8.
Powles TJ, et al. Oral clodronate and reduction in loss of bone mineral density in women with operable primary breast cancer. J Natl Cancer Inst. 1998;90:704–8. https://doi.org/10.1093/jnci/90.9.704.
Powles TJ, Hickish T, Kanis JA, Tidy A, Ashley S. Effect of tamoxifen on bone mineral density measured by dual-energy X-ray absorptiometry in healthy premenopausal and postmenopausal women. J Clin Oncol. 1996;14:78–84. https://doi.org/10.1200/jco.1996.14.1.78.
van Weelden WJ, Massuger L, Pijnenborg JMA, Romano A. Anti-estrogen treatment in endometrial cancer: a systematic review. Front Oncol. 2019;9:359. https://doi.org/10.3389/fonc.2019.00359.
Kristensen B, et al. Tamoxifen and bone metabolism in postmenopausal low-risk breast cancer patients: a randomized study. J Clin Oncol. 1994;12:992–7. https://doi.org/10.1200/jco.1994.12.5.992.
Saarto T, et al. Clodronate improves bone mineral density in post-menopausal breast cancer patients treated with adjuvant antioestrogens. Br J Cancer. 1997;75:602–5. https://doi.org/10.1038/bjc.1997.105.
Marttunen MB, Hietanen P, Tiitinen A, Ylikorkala O. Comparison of effects of tamoxifen and toremifene on bone biochemistry and bone mineral density in postmenopausal breast cancer patients. J Clin Endocrinol Metab. 1998;83:1158–62. https://doi.org/10.1210/jcem.83.4.4688.
Ward RL, Morgan G, Dalley D, Kelly PJ. Tamoxifen reduces bone turnover and prevents lumbar spine and proximal femoral bone loss in early postmenopausal women. Bone Miner. 1993;22:87–94. https://doi.org/10.1016/s0169-6009(08)80220-6.
Sverrisdóttir A, Fornander T, Jacobsson H, von Schoultz E, Rutqvist LE. Bone mineral density among premenopausal women with early breast cancer in a randomized trial of adjuvant endocrine therapy. J Clin Oncol. 2004;22:3694–9. https://doi.org/10.1200/jco.2004.08.148.
Kim M, et al. Changes in bone mineral density during 5 years of adjuvant treatment in premenopausal breast cancer patients. Breast Cancer Res Treat. 2020;180:657–63. https://doi.org/10.1007/s10549-020-05566-w.
Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. 2009;339: b2535. https://doi.org/10.1136/bmj.b2535.
Balduzzi S, Rücker G, Schwarzer G. How to perform a meta-analysis with R: a practical tutorial. Evid Based Ment Health. 2019;22:153–60. https://doi.org/10.1136/ebmental-2019-300117.
Viechtbauer W. Conducting meta-analyses in R with the metafor package. J Stat Softw. 2010;36:1–48.
Love RR, et al. Bone mineral density following surgical oophorectomy and tamoxifen adjuvant therapy for breast cancer. Cancer. 2013;119:3746–52. https://doi.org/10.1002/cncr.28302.
Hojan K, Milecki P, Molińska-Glura M, Roszak A, Leszczyński P. Effect of physical activity on bone strength and body composition in breast cancer premenopausal women during endocrine therapy. Eur J Phys Rehabil Med. 2013;49:331–9.
Nuzzo F, et al. Bone effect of adjuvant tamoxifen, letrozole or letrozole plus zoledronic acid in early-stage breast cancer: the randomized phase 3 HOBOE study. Ann Oncol. 2012;23:2027–33. https://doi.org/10.1093/annonc/mdr600.
Anderson RA, Cameron DA. Pretreatment serum anti-müllerian hormone predicts long-term ovarian function and bone mass after chemotherapy for early breast cancer. J Clin Endocrinol Metab. 2011;96:1336–43. https://doi.org/10.1210/jc.2010-2582.
Henry NL, et al. Lack of association between oestrogen receptor polymorphisms and change in bone mineral density with tamoxifen therapy. Br J Cancer. 2010;102:294–300. https://doi.org/10.1038/sj.bjc.6605460.
Gnant MF, et al. Zoledronic acid prevents cancer treatment-induced bone loss in premenopausal women receiving adjuvant endocrine therapy for hormone-responsive breast cancer: a report from the Austrian Breast and Colorectal Cancer Study Group. J Clin Oncol. 2007;25:820–8. https://doi.org/10.1200/jco.2005.02.7102.
Lee J, et al. Effect of tamoxifen on the risk of osteoporosis and osteoporotic fracture in younger breast cancer survivors: a nationwide study. Front Oncol. 2020;10:366. https://doi.org/10.3389/fonc.2020.00366.
Stumpf U, Kostev K, Kyvernitakis J, Böcker W, Hadji P. Incidence of fractures in young women with breast cancer—a retrospective cohort study. J Bone Oncol. 2019;18:100254. https://doi.org/10.1016/j.jbo.2019.100254.
Bradley R. Aromatase inhibitors versus tamoxifen in premenopausal women with oestrogen receptor-positive early-stage breast cancer treated with ovarian suppression: a patient-level meta-analysis of 7030 women from four randomised trials. Lancet Oncol. 2022;23:382–92. https://doi.org/10.1016/s1470-2045(21)00758-0.
Love RR, et al. Effects of tamoxifen on bone mineral density in postmenopausal women with breast cancer. N Engl J Med. 1992;326:852–6. https://doi.org/10.1056/nejm199203263261302.
Grey AB, et al. The effect of the antiestrogen tamoxifen on bone mineral density in normal late postmenopausal women. Am J Med. 1995;99:636–41. https://doi.org/10.1016/s0002-9343(99)80251-4.
Parfitt A. Morphologic basis of bone mineral measurements: transient and steady state effect of treatment in osteoporosis. Miner Electorlyte Metab. 1980;4:273–87.
Sibonga JD, Evans GL, Hauck ER, Bell NH, Turner RT. Ovarian status influences the skeletal effects of tamoxifen in adult rats. Breast Cancer Res Treat. 1996;41:71–9. https://doi.org/10.1007/bf01807038.
Ramchand SK, Cheung YM, Yeo B, Grossmann M. The effects of adjuvant endocrine therapy on bone health in women with breast cancer. J Endocrinol. 2019;241:R111-r124. https://doi.org/10.1530/joe-19-0077.
Watts NB. Clinical utility of biochemical markers of bone remodeling. Clin Chem. 1999;45:1359–68.
Barrett-Connor E, et al. Osteoporosis and fracture risk in women of different ethnic groups. J Bone Miner Res. 2005;20:185–94. https://doi.org/10.1359/jbmr.041007.
Zengin A, Prentice A, Ward KA. Ethnic differences in bone health. Front Endocrinol (Lausanne). 2015;6:24. https://doi.org/10.3389/fendo.2015.00024.
Acknowledgements
We would like to thank Editage (www.editage.co.kr) for English language editing.
Funding
There is no funding source.
Author information
Authors and Affiliations
Contributions
Study concept and design: CDC, SJL, MSC. Data acquisition: CDC, YYC. Quality control of data: CDC, SJL. Data analysis and interpretation: CDC, SJL, HH. Drafting of the manuscript: CDC, SJL, YYC, HH. Revision of the manuscript for important intellectual content: All authors. Manuscript review: MSC. Study supervision: MSC.
Corresponding author
Ethics declarations
Conflict of interest
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Lee, S.J., Cha, C.D., Hong, H. et al. Adverse effects of tamoxifen treatment on bone mineral density in premenopausal patients with breast cancer: a systematic review and meta-analysis. Breast Cancer (2024). https://doi.org/10.1007/s12282-024-01586-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12282-024-01586-2