Abstract
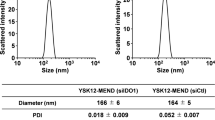
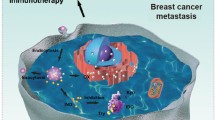
Researches on indoleamine-2,3-dioxygenase-1 (IDO1), a neoplastic pathogenesis-related protein, have provided a new angle of view to regulate malignancy-related immunosuppression. However, the therapeutic efficacy of IDO1 inhibitors is subject to key limitations as both cancer and dendritic cells tend to be trapped in the IDO1-mediated immune dysfunction, which poses challenges to the inhibitory potency of drug regimens in multiple targets. Here, we report on the fabrication technique of a biomimetic nanocarrier that is endowed with the whole array of cancer cell membrane proteins for encapsulating the most used IDO1 probe indoximod (IND). By fully utilizing the homologous adhesion proteins and antigenic motifs on cytomembrane, these nanoparticulate particles are capable of infiltrating tumors and actively accumulating in cancer and dendritic cells, as well as hitching a ride on dendritic cells to tumor-draining lymph nodes. Ultimately, by increasing the distribution of drugs in both tumor cells and dendritic cells in tumor-draining lymph nodes, these formulations greatly enhance the efficacy of IND without the aid of chemotherapeutic drugs, achieving substantial control of tumor growth. Overall, this leverage of bionanotechnology maximizes the therapeutic potential of IND and can provide a theoretical reference for the clinical application of IDO1 inhibitors.

Similar content being viewed by others
References
Fife, B. T.; Bluestone, J. A. Control of peripheral T-cell tolerance and autoimmunity via the CTLA-4 and PD-1 pathways. Immunol. Rev. 2008, 224, 166–182.
Waldman, A. D.; Fritz, J. M.; Lenardo, M. J. A guide to cancer immunotherapy: From T cell basic science to clinical practice. Nat. Rev. Immunol. 2020, 20, 651–668.
Xu, X. L.; Zhou, Z. C.; Li, H.; Fan, Y. Towards customized cancer vaccines: A promising filed in personalized cancer medicine. Expert Rev. Vaccines 2021, 20, 545–557.
Haslam, A.; Prasad, V. Estimation of the percentage of US patients with cancer who are eligible for and respond to checkpoint inhibitor immunotherapy drugs. JAMA Netw. Open 2019, 2, e192535.
Michot, J. M.; Bigenwald, C.; Champiat, S.; Collins, M.; Carbonnel, F.; Postel-Vinay, S.; Berdelou, A.; Varga, A.; Bahleda, R.; Hollebecque, A. et al. Immune-related adverse events with immune checkpoint blockade: A comprehensive review. Eur. J. Cancer 2016, 54, 139–148.
Das, S.; Johnson, D. B. Immune-related adverse events and antitumor efficacy of immune checkpoint inhibitors. J. Immunother. Cancer 2019, 7, 306.
Topalian, S. L.; Hodi, F. S.; Brahmer, J. R.; Gettinger, S. N.; Smith, D. C.; McDermott, D. F.; Powderly, J. D.; Carvajal, R. D.; Sosman, J. A.; Atkins, M. B. et al. Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N. Engl. J. Med. 2012, 366, 2443–2454.
Holmgaard, R. B.; Zamarin, D.; Li, Y. Y.; Gasmi, B.; Munn, D. H.; Allison, J. P.; Merghoub, T.; Wolchok, J. D. Tumor-expressed IDO recruits and activates MDSCs in a treg-dependent manner. Cell Rep. 2015, 13, 412–424.
Opitz, C. A.; Litzenburger, U. M.; Sahm, F.; Ott, M.; Tritschler, I.; Trump, S.; Schumacher, T.; Jestaedt, L.; Schrenk, D.; Weller, M. et al. An endogenous tumour-promoting ligand of the human aryl hydrocarbon receptor. Nature 2011, 478, 197–203.
Prendergast, G. C.; Mondal, A.; Dey, S.; Laury-Kleintop, L. D.; Muller, A. J. Inflammatory reprogramming with IDO1 inhibitors: Turning immunologically unresponsive “cold” tumors “hot”. Trends Cancer 2018, 4, 38–58.
Brochez, L.; Chevolet, I.; Kruse, V. The rationale of indoleamine 2, 3-dioxygenase inhibition for cancer therapy. Eur. J. Cancer 2017, 76, 167–182.
Platten, M.; Nollen, E. A. A.; Röhrig, U. F.; Fallarino, F.; Opitz, C. A. Tryptophan metabolism as a common therapeutic target in cancer, neurodegeneration and beyond. Nat. Rev. Drug Discov. 2019, 18, 379–401.
Munn, D. H.; Mellor, A. L. IDO in the tumor microenvironment: Inflammation, counter-regulation, and tolerance. Trends Immunol. 2016, 37, 193–207.
Prendergast, G. C.; Malachowski, W. P.; DuHadaway, J. B.; Muller, A. J. Discovery of IDO1 inhibitors: From bench to bedside. Cancer Res. 2017, 77, 6795–6811.
Opitz, C. A.; Patterson, L. F. S.; Mohapatra, S. R.; Dewi, D. L.; Sadik, A.; Platten, M.; Trump, S. The therapeutic potential of targeting tryptophan catabolism in cancer. Br. J. Cancer 2020, 122, 30–44.
Muller, A. J.; DuHadaway, J. B.; Donover, P. S.; Sutanto-Ward, E.; Prendergast, G. C. Inhibition of indoleamine 2, 3-dioxygenase, an immunoregulatory target of the cancer suppression gene Bin1, potentiates cancer chemotherapy. Nat. Med. 2005, 11, 312–319.
Hou, D. Y.; Muller, A. J.; Sharma, M. D.; DuHadaway, J.; Banerjee, T.; Johnson, M.; Mellor, A. L.; Prendergast, G. C.; Munn, D. H. Inhibition of indoleamine 2, 3-dioxygenase in dendritic cells by stereoisomers of 1-methyl-tryptophan correlates with antitumor responses. Cancer Res. 2007, 67, 792–801.
Holmgaard, R. B.; Zamarin, D.; Munn, D. H.; Wolchok, J. D.; Allison, J. P. Indoleamine 2, 3-dioxygenase is a critical resistance mechanism in antitumor T cell immunotherapy targeting CTLA-4. J. Exp. Med. 2013, 210, 1389–1402.
Jia, L. E; Schweikart, K.; Tomaszewski, J.; Page, J. G.; Noker, P. E.; Buhrow, S. A.; Reid, J. M.; Ames, M. M.; Munn, D. H. Toxicology and pharmacokinetics of 1-methyl-D-tryptophan: Absence of toxicity due to saturating absorption. Food Chem. Toxicol. 2008, 46, 203–211.
Zhang, X. D.; Wang, C.; Wang, J. Q.; Hu, Q. Y.; Langworthy, B.; Ye, Y. Q.; Sun, W. J.; Lin, J.; Wang, T. F.; Fine, J. et al. PD-1 blockade cellular vesicles for cancer immunotherapy. Adv. Mater. 2018, 30, 1707112.
Huang, T. L.; Li, S.; Fang, J. C.; Li, F. L.; Tu, S. P. Antibody-activated trans-endothelial delivery of mesoporous organosilica nanomedicine augments tumor extravasation and anti-cancer immunotherapy. Bioact. Mater. 2021, 6, 2158–2172.
Du, W.; Chen, C.; Sun, P.; Zhang, S. C.; Zhang, J.; Zhang, X. Y.; Liu, Y.; Zhang, R.; Yan, C. Z.; Fan, C. C. et al. Eliciting an immune hot tumor niche with biomimetic drug-based multi-functional nanohybrids augments immune checkpoint blockade-based breast cancer therapy. Nanoscale 2020, 12, 3317–3329.
Yang, W. J.; Zhang, F. W.; Deng, H. Z.; Lin, L. S.; Wang, S.; Kang, F.; Yu, G. C.; Lau, J.; Tian, R.; Zhang, M. R. et al. Smart nanovesicle-mediated immunogenic cell death through tumor microenvironment modulation for effective photodynamic immunotherapy. ACS Nano 2020, 14, 620–631.
Li, Q.; Zhang, D.; Zhang, J.; Jiang, Y.; Song, A. X.; Li, Z. H.; Luan, Y. X. A three-in-one immunotherapy nanoweapon via cascade-amplifying cancer-immunity cycle against tumor metastasis, relapse, and postsurgical regrowth. Nano Lett. 2019, 19, 6647–6657.
Kuang, J.; Song, W.; Yin, J.; Zeng, X.; Han, S.; Zhao, Y. P.; Tao, J.; Liu, C. J.; He, X. H.; Zhang, X. Z. iRGD modified chemoimmunotherapeutic nanoparticles for enhanced immunotherapy against glioblastoma. Adv. Funct. Mater. 2018, 28, 1800025.
Li, Q. L.; Liu, J.; Fan, H. L.; Shi, L.; Deng, Y.; Zhao, L.; Xiang, M. X.; Xu, Y. R.; Jiang, X. L.; Wang, G. B. et al. IDO-inhibitor potentiated immunogenic chemotherapy abolishes primary tumor growth and eradicates metastatic lesions by targeting distinct compartments within tumor microenvironment. Biomaterials 2021, 269, 120388.
Yang, Z.; Gao, D.; Guo, X. Q.; Jin, L.; Zheng, J. J.; Wang, Y.; Chen, S. J.; Zheng, X. W.; Zeng, L.; Guo, M. et al. Fighting immune cold and reprogramming immunosuppressive tumor microenvironment with red blood cell membrane-camouflaged nanobullets. ACS Nano 2020, 14, 17442–17457.
Liu, R.; An, Y.; Jia, W. F.; Wang, Y. S.; Wu, Y.; Zhen, Y. H.; Cao, J.; Gao, H. L. Macrophage-mimic shape changeable nanomedicine retained in tumor for multimodal therapy of breast cancer. J. Controlled Release 2020, 321, 589–601.
Peer, D.; Karp, J. M.; Hong, S.; FaroKhzad, O. C.; Margalit, R.; Langer, R. Nanocarriers as an emerging platform for cancer therapy. Nat. Nanotechnol. 2007, 2, 751–760.
Munn, D. H.; Mellor, A. L. The tumor-draining lymph node as an immune-privileged site. Immunol. Rev. 2006, 213, 146–158.
Swartz, M. A.; Lund, A. W. Lymphatic and interstitial flow in the tumour microenvironment: Linking mechanobiology with immunity. Nat. Rev. Cancer 2012, 12, 210–219.
Chen, Z.; Zhao, P. F.; Luo, Z. Y.; Zheng, M. B.; Tian, H.; Gong, P.; Gao, G. H.; Pan, H.; Liu, L. L.; Ma, A. Q. et al. Cancer cell membrane-biomimetic nanoparticles for homologous-targeting dualmodal imaging and photothermal therapy. ACS Nano 2016, 10, 10049–10057.
Rao, L.; Bu, L. L.; Cai, B.; Xu, J. H.; Li, A.; Zhang, W. F.; Sun, Z. J.; Guo, S. S.; Liu, W.; Wang, T. H. et al. Cancer cell membrane-coated upconversion nanoprobes for highly specific tumor imaging. Adv. Mater. 2016, 28, 3460–3466.
Shao, D.; Li, M. Q.; Wang, Z.; Zheng, X.; Lao, Y. H.; Chang, Z. M.; Zhang, F.; Lu, M. M.; Yue, J.; Hu, H. Z. et al. Bioinspired diselenide-bridged mesoporous silica nanoparticles for dual-responsive protein delivery. Adv. Mater. 2018, 30, 1801198.
Kroll, A. V.; Fang, R. H.; Jiang, Y.; Zhou, J.; Wei, X.; Yu, C. L.; Gao, J.; Luk, B. T.; Dehaini, D.; Gao, W. et al. Nanoparticulate delivery of cancer cell membrane elicits multiantigenic antitumor immunity. Adv. Mater. 2017, 29, 1703969.
Liu, W. L.; Zou, M. Z.; Liu, T.; Zeng, J. Y.; Li, X.; Yu, W. Y.; Li, C. X.; Ye, J. J.; Song, W.; Feng, J. et al. Cytomembrane nanovaccines show therapeutic effects by mimicking tumor cells and antigen presenting cells. Nat. Commun. 2019, 10, 3199.
Yang, R.; Xu, J.; Xu, L. G.; Sun, X. Q.; Chen, Q.; Zhao, Y. H.; Peng, R.; Liu, Z. Cancer cell membrane-coated adjuvant nanoparticles with mannose modification for effective anticancer vaccination. ACS Nano 2018, 12, 5121–5129.
Worbs, T.; Hammerschmidt, S. I.; Förster, R. Dendritic cell migration in health and disease. Nat. Rev. Immunol. 2017, 17, 30–48.
Zhang, Q. Z.; Dehaini, D.; Zhang, Y.; Zhou, J. L.; Chen, X. Y.; Zhang, L. F.; Fang, R. H.; Gao, W. W.; Zhang, L. F. Neutrophil membrane-coated nanoparticles inhibit synovial inflammation and alleviate joint damage in inflammatory arthritis. Nat. Nanotechnol. 2018, 13, 1182–1190.
Wang, C.; Chen, S. Q.; Wang, Y. X.; Liu, X. R.; Hu, F. Q.; Sun, J. H.; Yuan, H. Lipase-triggered water-responsive “pandora’s box” for cancer therapy: Toward induced neighboring effect and enhanced drug penetration. Adv. Mater. 2018, 30, 1706407.
Ye, Y. Q.; Wang, J. Q.; Hu, Q. Y.; Hochu, G. M.; Xin, H. L.; Wang, C.; Gu, Z. Synergistic transcutaneous immunotherapy enhances antitumor immune responses through delivery of checkpoint inhibitors. ACS Nano 2016, 10, 8956–8963.
Acknowledgements
The authors acknowledge the support from the National Natural Science Foundation of China (Nos. 81773648 and 81973267) and the Zhejiang Provincial Natural Science Foundation of China (Nos. LD19H300001 and LQ20H300004).
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Yu, T., Jin, X., Yu, F. et al. Tumor and dendritic cell dual-targeting nanocarriers maximize the therapeutic potential of IDO1 inhibitor in vivo. Nano Res. 15, 9204–9214 (2022). https://doi.org/10.1007/s12274-022-4597-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12274-022-4597-7