Abstract
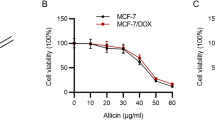
Polyphenols are known to induce apoptosis in many cancer cells and are proposed to be promising modulators of drug resistance. In the present study, we report that 3-geranyl-phloroacetophenone (3-GAP), a synthetic polyphenol, induces apoptosis and modulates drug resistance. In adriamycin-resistant MCF-7 human breast cancer (MCF-7/ADR) cells, which express a mutant form of p53, 3-GAP induced significant apoptosis, which was accompanied by no change in p53 transcriptional activity, but an increase in Bax expression, cyt c release, and activation of caspase-9, 7, and 3. In addition, 3-GAP significantly decreased the activity and expression level of glutathione S-transferase pi (GSTπ), a factor that induces drug resistance. Along with GSTπ inhibition, 3-GAP also induced a marked depletion of GSH, an endogenous antioxidant. The GST-inhibitory activity of 3-GAP correlated with the sensitization of MCF-7/ADR cells to doxorubicin. Under serum withdrawal conditions, the JNK inhibitor SP600125 significantly decreased the viability of the parent MCF-7 cells but not of MCF-7/ADR cells. In addition, the viability of 3-GAP-treated MCF-7/ADR cells was similar to those of MCF-7 cells treated with SP600125 alone or MCF-7/ADR cells co-treated with SP600125 and 3-GAP. These results indicate that JNK activity in MCF-7/ADR cells is halted by high levels of GSTπ, and that 3-GAP releases JNK from GSTπ’s inhibition. In conclusion, 3-GAP induces apoptosis in and sensitizes drug-resistant MCF-7/ADR cells. These effects are mediated through p53-independent caspase-3 activation and reduction of the capacity for cellular antioxidants, such as GSTπ and GSH.
Similar content being viewed by others
References
Adams, J. M. and Cory, S., The Bcl-2 protein family: arbiters of cell survival. Science, 281, 1322–1326 (1998).
Adler, V., Yin, Z., Fuchs, S. Y., Benezra, M., Rosario, L., Tew, K. D., Pincus, M. R., Sardana, M., Henderson, C. J., Wolf, C. R., Davis, R. J., and Ronai, Z., Regulation of JNK signaling by GSTp. EMBO J., 18, 1321–1334 (1999).
Aggarwal, B. B. and Shishodia, S., Molecular targets of dietary agents for prevention and therapy of cancer. Biochem. Pharmacol., 71, 1397–1421 (2006).
Cho, M.-Y., Park, S.-Y., Park, S., Lee, Y. R., Han, G. D., and Kim, J. A., Geranyl derivative of phloroacetophenone in duces cancer cell-specific apoptosis through Bax-mediated mitochondrial pathway in MCF-7 human breast cancer cells. Biol. Pharm. Bull., 35, 98–104 (2012).
Fairchild, C. R., Moscow, J. A., O’Brien, E. E., and Cowan, K. H., Multidrug resistance in cells transfected with human genes encoding a variant P-glycoprotein and glutathione S-transferase-pi. Mol. Pharmacol., 37, 801–809 (1990).
Ford, J. M., Hait, W. N., Matlin, S. A., and Benz, C. C., Modulation of resistance to alkylating agents in cancer cell by gossypol enantiomers. Cancer Lett., 56, 85–94 (1991).
Gehrmann, M. L., Fenselau, C., and Hathout, Y., Highly altered protein expression profile in the adriamycin resistant MCF-7 cell line. J. Proteome Res., 3, 403–409 (2004).
Germann, U. A., P-glycoprotein—a mediator of multidrug resistance in tumor cells. Eur. J. Cancer, 32A, 927–944 (1996).
Gottesman, M. M. and Ling, V., The molecular basis of multidrug resistance in cancer: the early years of Pglycoprotein research. FEBS Lett., 580, 998–1009 (2006).
Greenblatt, M. S., Bennett, W. P., Hollstein, M., and Harris, C. C., Mutations in the p53 tumor suppressor gene: clues to cancer etiology and molecular pathogenesis. Cancer Res., 54, 4855–4878 (1994).
Griffith, O. W., Determination of glutathione and glutathione disulfide using glutathione reductase and 2-vinylpyridine. Anal. Biochem., 106, 207–212 (1980).
Haldar, S., Negrini, M., Monne, M., Sabbioni, S., and Croce, C. M., Down-regulation of bcl-2 by p53 in breast cancer cells. Cancer Res., 54, 2095–2097 (1994).
Hochedlinger, K., Wagner, E. F., and Sabapathy, K., Differential effects of JNK1 and JNK2 on signal specific induction of apoptosis. Oncogene, 21, 2441–2445 (2002).
Kerbel, R. S., Kobayashi, H., and Graham, C. H., Intrinsic or acquired drug resistance and metastasis: are they linked phenotypes? J. Cell. Biochem., 56, 37–47 (1994).
Krajewski, S., Krajewska, M., Shabaik, A., Miyashita, T., Wang, H. G., and Reed, J. C., Immunohistochemical determination of in vivo distribution of Bax, a dominant inhibitor of Bcl-2. Am. J. Pathol., 145, 1323–1336 (1994).
Krajewski, S., Thor, A. D., Edgerton, S. M., Moore, D. H., 2nd, Krajewska, M., and Reed, J. C., Analysis of Bax and Bcl-2 expression in p53-immunopositive breast cancers. Clin. Cancer Res., 3, 199–208 (1997).
Krishna, R. and Mayer, L. D., Multidrug resistance (MDR) in cancer. Mechanisms, reversal using modulators of MDR and the role of MDR modulators in influencing the pharmacokinetics of anticancer drugs. Eur. J. Pharm. Sci., 11, 265–283 (2000).
Kroemer, G. and Reed, J. C., Mitochondrial control of cell death. Nat. Med., 6, 513–519 (2000).
Luqmani, Y. A., Mechanisms of drug resistance in cancer chemotherapy. Med. Princ. Pract., 14Suppl 1, 35–48 (2005).
Mei, Y., Wei, D., and Liu, J., Reversal of multidrug resistance in KB cells with tea polyphenol antioxidant capacity. Cancer Biol. Ther., 4, 468–473 (2005).
Mingo-Sion, A. M., Marietta, P. M., Koller, E., Wolf, D. M., and Van Den Berg, C. L., Inhibition of JNK reduces G2/M transit independent of p53, leading to endoreduplication, decreased proliferation, and apoptosis in breast cancer cells. Oncogene, 23, 596–604 (2004).
Miyashita, T., Krajewski, S., Krajewska, M., Wang, H. G., Lin, H. K., Liebermann, D. A., Hoffman, B., and Reed, J. C., Tumor suppressor p53 is a regulator of bcl-2 and bax gene expression in vitro and in vivo. Oncogene, 9, 1799–1805 (1994).
Moffat, G. J., McLaren, A. W., and Wolf, C. R., Involvement of Jun and Fos proteins in regulating transcriptional activation of the human pi class glutathione S-transferase gene in multidrug-resistant MCF7 breast cancer cells. J. Biol. Chem., 269, 16397–16402 (1994).
Ogretmen, B. and Safa, A. R., Down-regulation of apoptosis-related bcl-2 but not bcl-xL or bax proteins in multidrug-resistant MCF-7/Adr human breast cancer cells. Int. J. Cancer, 67, 608–614 (1996).
Ogretmen, B. and Safa, A. R., Expression of the mutated p53 tumor suppressor protein and its molecular and biochemical characterization in multidrug resistant MCF-7/Adr human breast cancer cells. Oncogene, 14, 499–506 (1997).
Oren, M., Decision making by p53: life, death and cancer. Cell Death Differ., 10, 431–442 (2003).
Potapova, O., Gorospe, M., Dougherty, R. H., Dean, N. M., Gaarde, W. A., and Holbrook, N. J., Inhibition of c-Jun N-terminal kinase 2 expression suppresses growth and induces apoptosis of human tumor cells in a p53-dependent manner. Mol. Cell. Biol., 20, 1713–1722 (2000).
Sadzuka, Y., Sugiyama, T., and Sonobe, T., Efficacies of tea components on doxorubicin induced antitumor activity and reversal of multidrug resistance. Toxicol. Lett., 114, 155–162 (2000).
Sartippour, M. R., Pietras, R., Marquez-Garban, D. C., Chen, H. W., Heber, D., Henning, S. M., Sartippour, G., Zhang, L., Lu, M., Weinberg, O., Rao, J. Y., and Brooks, M. N., The combination of green tea and tamoxifen is effective against breast cancer. Carcinogenesis, 27, 2424–2433 (2006).
Silvestrini, R., Benini, E., Veneroni, S., Daidone, M. G., Tomasic, G., Squicciarini, P., and Salvadori, B., p53 and bcl-2 expression correlates with clinical outcome in a series of node-positive breast cancer patients. J. Clin. Oncol., 14, 1604–1610 (1996).
Silvestrini, R., Veneroni, S., Benini, E., Daidone, M. G., Luisi, A., Leutner, M., Maucione, A., Kenda, R., Zucali, R., and Veronesi, U., Expression of p53, glutathione S-transferase-pi, and Bcl-2 proteins and benefit from adjuvant radiotherapy in breast cancer. J. Natl. Cancer Inst., 89, 639–645 (1997).
Susan, B., Moyers, S. B., Nagi, B., and Kumar, N. B., Green tea polyphenols and cancer chemoprevention: multiple mechanisms and endpoints for phase II trials. Nutr. Rev., 62, 204–211 (2004).
Tew, K. D., Bomber, A. M., and Hoffman, S. J., Ethacrynic acid and piriprost as enhancers of cytotoxicity in drug resistant and sensitive cell lines. Cancer Res., 48, 3622–3625 (1988).
Tournier, C., Hess, P., Yang, D. D., Xu, J., Turner, T. K., Nimnual, A., Bar-Sagi, D., Jones, S. N., Flavell, R. A., and Davis, R. J., Requirement of JNK for stress-induced activation of the cytochrome c-mediated death pathway. Science, 288, 870–874 (2000).
Townsend, D. M. and Tew, K. D., The role of glutathione-Stransferase in anti-cancer drug resistance. Oncogene, 22, 7369–7375 (2003).
van Zanden, J. J., Wortelboer, H. M., Bijlsma, S., Punt, A., Usta, M., Bladeren, P. J., Rietjens, I. M., and Cnubben, N. H., Quantitative structure activity relationship studies on the flavonoid mediated inhibition of multidrug resistance proteins 1 and 2. Biochem. Pharmacol., 69, 699–708 (2005).
Yu, S. T., Chen, T. M., Chern, J. W., Tseng, S. Y., and Chen, Y. H., Downregulation of GSTpi expression by tryptanthrin contributing to sensitization of doxorubicin-resistant MCF-7 cells through c-jun NH2-terminal kinase-mediated apoptosis. Anticancer Drugs, 20, 382–388 (2009).
Zhang, S., Yang, X., and Morris, M. E., Flavonoids are inhibitors of breast cancer resistance protein (ABCG2)-mediated transport. Mol. Pharmacol., 65, 1208–1216 (2004).
Zhao, G. and Wang, X., Advance in antitumor agents targeting glutathione-S-transferase. Curr. Med. Chem., 13, 1461–1471 (2006).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cho, MY., Park, SY., Park, S. et al. Effects of geranyl-phloroacetophenone on the induction of apoptosis and chemosensitization of adriamycin-resistant MCF-7 human breast cancer cells. Arch. Pharm. Res. 35, 911–919 (2012). https://doi.org/10.1007/s12272-012-0517-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12272-012-0517-9