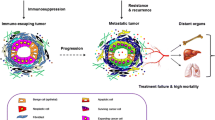
Abstract
The tumorigenesis and cancer cell progression are complicated processes starting from primary tumor formation by dysregulated vascularization to systemic metastasis. The tumor tends to thrive in a microenvironment that anchors its growth, progression, activation, and malignancies in the nearby tissues. The tumor microenvironment (TME) comprises different types of immune cells such as neutrophils, dendritic cells, macrophages, and fibroblasts, which have important functions in the activation of tumor suppression, and an extracellular matrix with cytokines, chemokines, and interleukins for cell signaling. This microenvironment niche has emerged as a significant way to target tumor cells and is implied in cancer immunotherapy techniques. The deeper insights into diverse cell types of cancer, carcinoma ligands, spatial arrangements, and leaky blood vessels have provided favorable circumstances for modulating therapeutic conciliation. This mini-review discusses the nature of the TME, its varied constituents, and its role in cancer therapeutics to target tumors. Overall, this review provides a brief account of the tumor microenvironment and its promising role in cancer treatment.



Similar content being viewed by others
Data Availability
Not applicable.
References
Sung H, Ferlay J, Siegel RL et al (2021) Global cancer statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin 71:209–249. https://doi.org/10.3322/CAAC.21660
Shelton SE, Nguyen HT, Barbie DA, Kamm RD (2021) Engineering approaches for studying immune-tumor cell interactions and immunotherapy. iScience 24:101985. https://doi.org/10.1016/J.ISCI.2020.101985
Abou Khouzam R, Goutham HV, Zaarour RF et al (2020) Integrating tumor hypoxic stress in novel and more adaptable strategies for cancer immunotherapy. Semin Cancer Biol 65:140–154. https://doi.org/10.1016/J.SEMCANCER.2020.01.003
Wang JJ, Lei KF, Han F (2018) Tumor microenvironment: recent advances in various cancer treatments. Eur Rev Med Pharmacol Sci 22(12):3855–3864. http://www.europeanreview.org/wp/wp-content/uploads/3855-3864.pdf
Binnewies M, Roberts EW, Kersten K et al (2018) Understanding the tumor immune microenvironment (TIME) for effective therapy. Nat Med 24(5):541–550. https://doi.org/10.1038/s41591-018-0014-x
Bergers G (2021) Fendt SM (2021) The metabolism of cancer cells during metastasis. Nat Rev Cancer 21(3):162–180. https://doi.org/10.1038/s41568-020-00320-2
Freeman JW (2021) Structural biology of the tumor microenvironment. Adv Exp Med Biol 1350:91–100. https://doi.org/10.1007/978-3-030-83282-7_4/COVER
Greten FR, Grivennikov SI (2019) Inflammation and cancer: triggers, mechanisms, and consequences. Immunity 51:27–41. https://doi.org/10.1016/J.IMMUNI.2019.06.025
Zhao H, Wu L, Yan G et al (2021) Inflammation and tumor progression: signaling pathways and targeted intervention. Signal Trans Target Ther 6:1 6:1–46. https://doi.org/10.1038/s41392-021-00658-5
Korneev KV, Atretkhany KSN, Drutskaya MS et al (2017) TLR-signaling and proinflammatory cytokines as drivers of tumorigenesis. Cytokine 89:127–135. https://doi.org/10.1016/J.CYTO.2016.01.021
Poeta VM, Massara M, Capucetti A, Bonecchi R (2019) Chemokines and chemokine receptors: new targets for cancer immunotherapy. Front Immunol 10:379. https://doi.org/10.3389/FIMMU.2019.00379/BIBTEX
Walker C, Mojares E, Del RíoHernández A (2018) Role of extracellular matrix in development and cancer progression. Int J Mol Sci 19:3028. https://doi.org/10.3390/IJMS19103028
Hill BS, Sarnella A, D’Avino G, Zannetti A (2020) Recruitment of stromal cells into tumour microenvironment promote the metastatic spread of breast cancer. Semin Cancer Biol 60:202–213. https://doi.org/10.1016/J.SEMCANCER.2019.07.028
Baghban R, Roshangar L, Jahanban-Esfahlan R et al (2020) Tumor microenvironment complexity and therapeutic implications at a glance. Cell Commun Signal 18:1–19. https://doi.org/10.1186/S12964-020-0530-4
Jin MZ, Jin WL (2020) The updated landscape of tumor microenvironment and drug repurposing. Signal Transduct Target Ther 5(1):16. https://doi.org/10.1038/s41392-020-00280-x
Jing X, Yang F, Shao C et al (2019) Role of hypoxia in cancer therapy by regulating the tumor microenvironment. Mol Cancer 18:1–15. https://doi.org/10.1186/S12943-019-1089-9
Xiao Y, Yu D (2021) Tumor microenvironment as a therapeutic target in cancer. Pharmacol Ther 221:107753. https://doi.org/10.1016/J.PHARMTHERA.2020.107753
Granot Z (2019) Neutrophils as a therapeutic target in cancer. Front Immunol 10:1710. https://doi.org/10.3389/FIMMU.2019.01710/BIBTEX
Blando J, Sharma A, Higa MG et al (2019) Comparison of immune infiltrates in melanoma and pancreatic cancer highlights VISTA as a potential target in pancreatic cancer. Proc Natl Acad Sci U S A 116:1692–1697. https://doi.org/10.1073/PNAS.1811067116/SUPPL_FILE/PNAS.1811067116.SAPP.PDF
Su S, Chen J, Yao H et al (2018) CD10+GPR77+ Cancer-associated fibroblasts promote cancer formation and chemoresistance by sustaining cancer stemness. Cell 172:841-856.e16. https://doi.org/10.1016/J.CELL.2018.01.009
Schito L, Semenza GL (2016) Hypoxia-inducible factors: master regulators of cancer progression. Trends Cancer 2:758–770. https://doi.org/10.1016/J.TRECAN.2016.10.016
Jiang X, Wang J, Deng X et al (2020) (2020) The role of microenvironment in tumor angiogenesis. J Exp Clin Cancer Res 39(1):1–19. https://doi.org/10.1186/S13046-020-01709-5
Zhang Q, Han Z, Zhu Y et al (2021) Role of hypoxia inducible factor-1 in cancer stem cells (Review). Mol Med Rep 23:1–1. https://doi.org/10.3892/MMR.2020.11655/HTML
Hodroj MH, Jardaly A, Raad SA et al (2018) Andrographolide potentiates the antitumor effect of topotecan in acute myeloid leukemia cells through an intrinsic apoptotic pathway. Cancer Manag Res 10:1079. https://doi.org/10.2147/CMAR.S160924
Jiang B (2017) Aerobic glycolysis and high level of lactate in cancer metabolism and microenvironment. Genes Dis 4:25–27. https://doi.org/10.1016/J.GENDIS.2017.02.003
Raza S, Rajak S, Tewari A et al (2022) Multifaceted role of chemokines in solid tumors: From biology to therapy. Semin Cancer Biol 86:1105–1121. https://doi.org/10.1016/J.SEMCANCER.2021.12.011
Zuazo-Gaztelu I, Casanovas O (2018) Unraveling the role of angiogenesis in cancer ecosystems. Front Oncol 8:248. https://doi.org/10.3389/FONC.2018.00248/BIBTEX
Zhang H, Shen YW, Zhang LJ et al (2021) Targeting endothelial cell-specific molecule 1 protein in cancer: a promising therapeutic approach. Front Oncol 11:2002. https://doi.org/10.3389/FONC.2021.687120/BIBTEX
Christofi T, Baritaki S, Falzone L et al (2019) Current perspectives in cancer immunotherapy. Cancers 11:1472. https://doi.org/10.3390/CANCERS11101472
Chanu TM, Kma L, Sharan RN (2023) Cancer biomarkers: status and its future direction. Indian J Surg 1–13. https://link.springer.com/article/10.1007/s12262-023-03723-1
Singh G, Yoshida EM, Rathi S et al (2020) Biomarkers for hepatocellular cancer. World J Hepatol 12:558. https://doi.org/10.4254/WJH.V12.I9.558
Wanyama FM, Blanchard V (2021) Glycomic-based biomarkers for ovarian cancer: advances and challenges. Diagnostics 11:643. https://doi.org/10.3390/DIAGNOSTICS11040643
Hristova VA, Chan DW (2019) Cancer biomarker discovery and translation: proteomics and beyond. Expert Rev Proteomics 16(2):93–103. https://doi.org/10.1080/14789450.2019.1559062
Asif S, Teply BA (2021) Biomarkers for treatment response in advanced prostate cancer. Cancers 13:5723. https://doi.org/10.3390/CANCERS13225723
Huang H, Yang Y, Zhu Y et al (2022) Blood protein biomarkers in lung cancer. Cancer Lett 551:215886. https://doi.org/10.1016/J.CANLET.2022.215886
Acknowledgements
ST and BS have collected the data and drafted the manuscript. SS prepared the figures. AP provided the final version of manuscript.
Funding
AP thanks Department of Biotechnology, Ministry of Science and Technology, Government of India, for MK Bhan Young researcher fellowship program (HRD-12/4/2020-AFS-DBT-Part (1) (13815).
Author information
Authors and Affiliations
Contributions
ST and BS have collected the data and drafted the manuscript. SS prepared the figures. AP provided the final version of manuscript.
Corresponding author
Ethics declarations
Ethics Approval and Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Conflict of Interest
There authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tiwari, S., Siddiqui, B., Singh, S. et al. Tumor Microenvironment: a Therapeutic Aid in Cancer. Indian J Surg 86, 57–63 (2024). https://doi.org/10.1007/s12262-023-03828-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12262-023-03828-7