Abstract
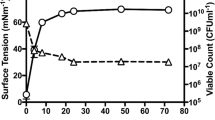
Pseudomonas protegens is a gram-negative bacterium with an excellent biological control effect. Compared to standard cells growing in NaCl-free media, the ability of the hyperosmotic cells (450 mM NaCl) to resist high temperatures and freezing was significantly improved. It is of great significance to apply the P. protegens to elaborate on the hyperosmotic adaptation mechanism. RNA-seq was used to sequence P. protegens cultured with 0 mM and 450 mM NaCl. Comparative transcriptomic analyses of the different treatments were performed using gene ontology and the Kyoto encyclopedia of genes and genome. The results revealed that hyperosmotic stress had prominent impacts on the genes involving in multiple cellular functions. The hyperosmotic environment significantly affected carbohydrate, energy, and amino acid metabolism, as well as membrane system and cell motility. Our findings indicated that P. protegens adopted a series of approaches, including the high cytoplasmic concentrations of potassium ions and the uptake or synthesis of osmoprotectants, for surviving hyperosmotic stress. Among these, trehalose and proline synthesis appeared to be an important method to withstand prolonged hyperosmotic stress in P. protegens. These data provide crucial resource that may determine specific responses to the hyperosmotic environment in P. protegens.
Similar content being viewed by others
Reference
Bonaterra, A., L. Ruz, E. Badosa, J. Pinochet, and E. Montesinos (2003) Growth promotion of Prunus rootstocks by root treatment with specific bacterial strains. Plant Soil. 255: 555–569.
Krimm, U., D. Abanda-Nkpwatt, W. Schwab, and L. Schreiber (2005) Epiphytic microorganisms on strawberry plants (Fragaria ananassa cv. Elsanta): identification of bacterial isolates and analysis of their interaction with leaf surfaces. FEMS Microbiol Ecol. 53: 483–492.
Walsh, U. F., J. P. Morrissey, and F. O’Gara (2001) Pseudomonas for biocontrol of phytopathogens: from functional genomics to commercial exploitation. Curr. Opin. Biotechnol. 12: 289–295.
Rajkumar, M., L. B. Bruno, and J. R. Banu (2017) Alleviation of environmental stress in plants: The role of beneficial Pseudomonas spp. Crit. Rev. Environ. Sci. Technol. 47: 372–407.
Ramette, A., M. Frapolli, M. Fischer-Le Saux, C. Gruffaz, J. M. Meyer, G. Defago, L. Sutra, and Y. Moenne-Loccoz (2011) Pseudomonas protegens sp. nov., widespread plant-protecting bacteria producing the biocontrol compounds 2,4-diacetylphloroglucinol and pyoluteorin. Syst Appl Microbiol. 34: 180–188.
Haas, D. and G. Defago (2005) Biological control of soil-borne pathogens by fluorescent pseudomonads. Nat. Rev. Microbiol. 3: 307–319.
Lou, H., X. Wang, J. Chen, B. Wang, and W. Wang (2018) Transcriptomic response of Ralstonia solanacearum to antimicrobial Pseudomonas fluorescens SN15-2 metabolites. Can. J. Microbiol. 64: 816–825.
den Besten, H. M. W., E. J. van der Mark, L. Hensen, T. Abee, and M. H. Zwietering (2010) Quantification of the effect of culturing temperature on salt-induced heat resistance of Bacillus species. Appl Environ Microbiol. 76: 4286–4292.
Amiet-Charpentier, C., P. Gadille, and J. P. Benoit (1999) Rhizobacteria microencapsulation: properties of microparticles obtained by spray-drying. J. Microencapsul. 16: 215–229.
Caspeta, L., Y. Chen, P. Ghiaci, A. Feizi, S. Buskov, B. M. Hallstrom, D. Petranovic, and J. Nielsen (2014) Altered sterol composition renders yeast thermotolerant. Science. 346: 75–78.
Cabrefiga, J., J. Frances, E. Montesinos, and A. Bonaterra (2011) Improvement of fitness and efficacy of a fire blight biocontrol agent via nutritional enhancement combined with osmoadaptation. Appl Environ Microbiol. 77: 3174–3181.
Bonaterra, A., J. Camps, and E. Montesinos (2005) Osmotically induced trehalose and glycine betaine accumulation improves tolerance to desiccation, survival and efficacy of the postharvest biocontrol agent Pantoea agglomerans EPS125. FEMS Microbiol. Lett. 250: 1–8.
Cabrefiga, J., J. Frances, E. Montesinos, and A. Bonaterra (2014) Improvement of a dry formulation of Pseudomonas fluorescens EPS62e for fire blight disease biocontrol by combination of culture osmoadaptation with a freeze-drying lyoprotectant. J. Appl. Microbiol. 117: 1122–1131.
Liu, L., L. Si, X. Meng, and L. Luo (2015) Comparative transcriptomic analysis reveals novel genes and regulatory mechanisms of Tetragenococcus halophilus in response to salt stress. J. Ind. Microbiol. Biotechnol. 42: 601–616.
Obruca, S., P. Sedlacek, V. Krzyzanek, F. Mravec, K. Hrubanova, O. Samek, D. Kucera, P. Benesova, and I. Marova (2016) Accumulation of poly(3-hydroxybutyrate) helps bacterial cells to survive freezing. PLoS One. 11: e0157778.
Li, B. and C. N. Dewey (2011) RSEM: accurate transcript quantification from RNA-Seq data with or without a reference genome. BMC Bioinformatics. 12: 323.
Robinson, M. D., D. J. McCarthy, and G. K. Smyth (2010) edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics. 26: 139–140.
Xie, C., X. Mao, J. Huang, Y. Ding, J. Wu, S. Dong, L. Kong, G. Gao, C. Y. Li, and L. Wei (2011) KOBAS 2.0: a web server for annotation and identification of enriched pathways and diseases. Nucleic Acids Res. 39: W316–W322.
Zhang, L., J. Xu, J. Xu, H. Zhang, L. He, and J. Feng (2014) TssB is essential for virulence and required for Type VI secretion system in Ralstonia solanacearum. Microb. Pathog. 74: 1–7.
Corbella, M. E. and A. Puyet (2003) Real-time reverse transcription-PCR analysis of expression of halobenzoate and salicylate catabolism-associated operons in two strains of Pseudomonas aeruginosa. Appl. Environ. Microbiol. 69: 2269–2275.
Beales, N. (2004) Adaptation of microorganisms to cold temperatures, weak acid preservatives, low pH, and osmotic stress: A review. Compr. Rev. Food Sci. Food Saf. 3: 1–20.
Marles-Wright, J. and R. J. Lewis (2007) Stress responses of bacteria. Curr. Opin. Struct. Biol. 17: 755–760.
Koga, T., T. Katagiri, H. Hori, and K. Takumi (2002) Alkaline adaptation induces cross-protection against some environmental stresses and morphological change in Vibrio parahaemolyticus. Microbiol. Res. 157: 249–255.
Ayub, N. D., M. J. Pettinari, B. S. Mendez, and N. I. Lopez (2007) The polyhydroxyalkanoate genes of a stress resistant Antarctic Pseudomonas are situated within a genomic island. Plasmid. 58: 240–248.
Wu, D., J. He, Y. Gong, D. Chen, X. Zhu, N. Qiu, M. Sun, M. Li, and Z. Yu (2011) Proteomic analysis reveals the strategies of Bacillus thuringiensis YBT-1520 for survival under long-term heat stress. Proteomics. 11: 2580–2591.
Raiger Iustman, L. J., P. M. Tribelli, J. G. Ibarra, M. V. Catone, E. C. Solar Venero, and N. I. López (2015) Genome sequence analysis of Pseudomonas extremaustralis provides new insights into environmental adaptability and extreme conditions resistance. Extremophiles. 19: 207–220.
Pavez, P., J. L. Castillo, C. González, and M. Martínez (2009) Poly-β-hydroxyalkanoate exert a protective effect against carbon starvation and frozen conditions in Sphingopyxis chilensis. Curr. Microbiol. 59: 636–640.
Obruca, S., P. Sedlacek, F. Mravec, V. Krzyzanek, J. Nebesarova, O. Samek, D. Kucera, P. Benesova, K. Hrubanova, M. Milerova, and I. Marova (2017) The presence of PHB granules in cytoplasm protects non-halophilic bacterial cells against the harmful impact of hypertonic environments. N Biotechnol. 39: 68–80.
Yaakop, A. S., K. G. Chan, R. Ee, Y. L. Lim, S. K. Lee, F. Abd Manan, and K. M. Goh (2016) Characterization of the mechanism of prolonged adaptation to osmotic stress of Jeotgalibacillus malaysiensis via genome and transcriptome sequencing analyses. Sci. Rep. 6: 33660.
Smulek, W., Z. Cybulski, U. Guzik, T. Jesionowski, and E. Kaczorek (2019) Three chlorotoluene-degrading bacterial strains: Differences in biodegradation potential and cell surface properties. Chemosphere. 237: 124452.
Munns, R. (2002) Comparative physiology of salt and water stress. Plant Cell Environ. 25: 239–250.
Fujita, Y., H. Matsuoka, and K. Hirooka (2007) Regulation of fatty acid metabolism in bacteria. Mol. Microbiol. 66: 829–839.
Leesong, M., B. S. Henderson, J. R. Gillig, J. M. Schwab, and J. L. Smith (1996) Structure of a dehydratase-isomerase from the bacterial pathway for biosynthesis of unsaturated fatty acids: two catalytic activities in one active site. Structure. 4: 253–264.
Reina-Bueno, M., M. Argandoña, J. J. Nieto, A. Hidalgo-García, F. Iglesias-Guerra, M. J. Delgado, and C. Vargas (2012) Role of trehalose in heat and desiccation tolerance in the soil bacterium Rhizobium etli. BMC Microbiol. 12: 207.
Yanhe, M., E. A. Galinski, W. D. Grant, A. Oren, and V. Antonio (2010) Halophiles 2010: life in saline environments. Appl. Environ. Microbiol. 76: 6971–6981.
Holtmann, G., E. P. Bakker, N. Uozumi, and E. Bremer (2003) KtrAB and KtrCD: Two K+ uptake systems in Bacillus subtilis and their role in adaptation to hypertonicity. J. Bacteriol. 185: 1289–1298.
Aspedon, A., K. Palmer, and M. Whiteley (2006) Microarray analysis of the osmotic stress response in Pseudomonas aeruginosa. J. Bacteriol. 188: 2721–2725.
Seo, S. W., Y. Gao, D. Kim, R. Szubin, J. Yang, B. K. Cho, and B. O. Palsson (2017) Revealing genome-scale transcriptional regulatory landscape of OmpR highlights its expanded regulatory roles under osmotic stress in Escherichia coli K-12 MG1655. Sci. Rep. 7: 2181.
Pastor, J. M., V. Bernal, M. Salvador, M. Argandona, C. Vargas, L. Csonka, A. Sevilla, J. L. Iborra, J. J. Nieto, and M. Canovas (2013) Role of central metabolism in the osmoadaptation of the halophilic bacterium Chromohalobacter salexigens. J. Biol. Chem. 288: 17769–17781.
Sleator, R. D. and C. Hill (2002) Bacterial osmoadaptation: the role of osmolytes in bacterial stress and virulence. FEMS Microbiol. Rev. 26: 49–71.
Roberts, M. F. (2005) Organic compatible solutes of halotolerant and halophilic microorganisms. Saline Systems. 1: 5.
Tonon, T. and A. Lonvaud-Funel (2000) Metabolism of arginine and its positive effect on growth and revival of Oenococcus oeni. J. Appl. Microbiol. 89: 526–531.
Mira de Orduna, R., M. L. Patchett, S. Q. Liu, and G. J. Pilone (2001) Growth and arginine metabolism of the wine lactic acid bacteria Lactobacillus buchneri and Oenococcus oeni at different pH values and arginine concentrations. Appl. Environ. Microbiol. 67: 1657–1662.
Barron, A., G. May, E. Bremer, and M. Villarejo (1986) Regulation of envelope protein composition during adaptation to osmotic stress in Escherichia coli. J. Bacteriol. 167: 433–438.
Bremer, E. (2000) Coping with osmotic challenges: osmoregulation through accumulation and release of compatible solutes in B. subtilis. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 126: S17.
Marion, R. M., A. Regev, E. Segal, Y. Barash, D. Koller, N. Friedman, and E. K. O’Shea (2004) Sfp1 is a stress- and nutrient-sensitive regulator of ribosomal protein gene expression. Proc. Natl. Acad. Sci. USA. 101: 14315–14322.
Maier, T., A. Schmidt, M. Guell, S. Kuhner, A. C. Gavin, R. Aebersold, and L. Serrano (2011) Quantification of mRNA and protein and integration with protein turnover in a bacterium. Mol. Syst. Biol. 7: 511.
Zgur-Bertok, D. (2013) DNA Damage repair and bacterial pathogens. PLoS Pathog. 9: e1003711.
Liu, Y., W. Gao, Y. Wang, L. Wu, X. Liu, T. Yan, E. Alm, A. Arkin, D. K. Thompson, M. W. Fields, and J. Zhou (2005) Transcriptome analysis of Shewanella oneidensis MR-1 in response to elevated salt conditions. J. Bacteriol. 187: 2501–2507.
Steil, L., T. Hoffmann, I. Budde, U. Völker, and E. Bremer (2003) Genome-wide transcriptional profiling analysis of adaptation of Bacillus subtilis to high salinity. J Bacteriol. 185: 6358–6370.
Acknowledgements
Funding: This work was supported by the National Key Research and Development Program of China under Grant number 2017YFD020040; the Project of Prospering Agriculture through Science and Technology of Shanghai, China under Grant number Hu NongKeChuangZi (2018) No. 2–5.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
No potential conflict of interest was reported by the authors. Neither ethical approval nor informed consent was required for this study.
Additional information
Publisher’s Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Wang, X., Tang, D. & Wang, W. Hyperosmotic Adaptation of Pseudomonas protegens SN15-2 Helps Cells to Survive at Lethal Temperatures. Biotechnol Bioproc E 25, 403–413 (2020). https://doi.org/10.1007/s12257-019-0430-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-019-0430-x