Abstract
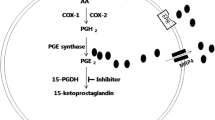
A novel series of benzylidenethiazolidine-2,4-dione derivatives was synthesized and investigated for 15-hydroxyprostaglandin dehydrogenase (15-PGDH)-scavenging activity, PGE2 release, and wound-healing activity. Among the tested derivatives, seven compounds (3, 9, 11, 12, 13, 14, and 25) resulted in a 50% inhibition of 15-PGDH at concentrations between 0.07 and 0.2 µM and increased PGE2 levels from 300 to over 600% in A549 cells treated with 5.0 and 10.0 µM of the compounds for 12 h. A scratch wound-healing assay using HaCaT cell line was conducted to verify the effects of 10 µM of these compounds on cell regeneration. The closure rate of the scratch wound healing showed that all compounds (3, 9, 11, 12, 13, 14, and 25) had greater wound regeneration effects than the cell growth factor, TGF-β1, which was used as the positive control. In particular, (Z)-N-benzyl-4-((2,4-dioxothiazolidin-5-ylidene)methyl)benzamide (compound 14) showed that the highest wound closure rate, which was 360%; this is about 3.6-fold higher than that of TGF-β1. Overall, these results show that compound 14 may be considered a promising candidate for the development of novel wound-healing agents.
Similar content being viewed by others
References
Sanon, S., D. A. Hart, and E. E. Tredget (2016) Molecular and cellular biology of wound healing and skin regeneration, Medicine, New York, Elsevier Inc., pp. 162–184.
Jeong, B. S. (2006) Structure-activity relationship study of asiatic acid derivatives for new wound healing agent. Arch. Pharma. Res. 29: 556–562.
Halehatty, P., N. Prakash, S. Halehatty, N. Bhojya, R. Thangali, N. Ravikumar, N. Raja, K. Gouthamchandra, R. Mahmood, and B. M. Khadeer Ahame (2009) Synthesis of novel benzo[h]quinolines. European J. Med. Chem. 44: 981–989.
Patrulea, V., N. Hirt-Bur, A. Jeannerat, L. A. Applegate, V. Ostafe, and O. G Jordan (2016) Borchard, Peptide-decorated chitosan derivatives enhance fibroblast adhesion and proliferation in wound healing. Carbohydrate Poly. 142: 114–123.
Patrulea, V., V. Ostafe, G. Borchard, and O. Jordan (2015) Chitosan as a starting material for wound healing applications. European J. Pharma. Biopharma. 97: 417–426.
Davidson, J. M., L. B. Nanney, K. N. Broadley, J. S. Whitsett, A. M. Aquino, M. Beccaro, and A. Rastrelli (1991) Hyaluronate derivatives and their application to wound healing. Clinical Mater. 8: 171–177.
Valacchi, G., G Grisci, C. Sticozzi, Y. S. Lim, M. Paolino, G. Giuliani, R. Mendichi, G Belmonte, R. Artusi, A. Zanardi, P. Garofalo, G. Giorgi, A. Cappelli, and L. Rovati (2015) Wound healing properties of hyaluronan derivatives bearing ferulate residues. J. Mater. Chem. 35: 7037–7045.
Lee, J. S., K. R. Lee, S. R. Lee, H.J. Lee, H. S. Yang, J. H. Yeo, J. M. Park, B. H. Choi and E. K. Hong (2017) Polysaccharides isolated from liquid culture broth of Inonotus obliquus inhibit the invasion of human non-small cell Lung carcinoma cells. Biotechnol. Bioprocess Eng. 22: 45–51.
Oh, M. S., Y. J. Kim, Y. J. Son, H. S. Yoo, and J. H Park (2017) Promotive effects of human induced pluripotent stem cell- conditioned medium on the proliferation and migration of dermal fibroblasts. Biotechnol. Bioprocess Eng. 22: 561–568.
Manjal, S. K., R. Kaur, R. Bhatia, K. Kumar, V. Singh, R. Shankar, R. Kaur, and R. K. Rawal (2017) Synthetic and medicinal perspective of thiazolidinones. Bioorganic Chem. 75: 406–423.
Jo, D. R., Y. O. Kim, R. Kim, Y. C. Chang, D. B. Choi, and H. Cho (2017) Novel rhodanine derivatives are selective algicides against Microcystis aeruginosa. Biotechnol. Bioprocess Eng. 22: 748–757.
You, D. S., Y. W. Lee, D. B. Choi, Y. C. Chang, and H. Cho (2017) Algicidal effects of thiazolinedione derivatives against Microcystis aeruginosa, Korean J. Chem. Eng. 34: 139–149.
Choi, D. B., S. J. Yu, S. H. Baek, Y. H. Kang, Y. C. Chang, and H. Cho (2016) Synthesis and algicidal activity of new dichloro- benzylamine derivatives against harmful red tides. Biotechnol. Bioprocess Eng. 21: 463–476.
Cho, H. and H. H. Tai (2002) Thiazolidinediones as a novel class of NAD+-dependent 15-hydroxyprostaglandin dehydrogenase inhibitors. Arch. Biochem. Biophys. 405: 247–251.
Wu, Y., H. H. Tai, and H. Cho (2010) Synthesis and SAR of thiazolidinedione derivatives as 15-PGDH inhibitors. Bioorg. Medicin. Chem. 18: 1428–1433.
Niesen, F. H., L. Schultz, A. Jadhav, C. Bhatia 1, K. Guo, D. J. Maloney, E. S. Pilka, M. Wang, U. Oppermann, T. D. Heightman, and A. Simeonov (2010) High-affinity inhibitors of human NAD -dependent 15-hydroxyprostaglandin dehydrogenase: mechanisms of inhibition and structure activity relationships. PLoS One 5: 13719–13722.
Wu, Y., S. D. Karna, C. H. Choi, M. Tong, H. H. Tai, D. H. Na, C. H. Jang, and H. Cho (2011) Synthesis and biological evaluation of novel thiazolidinedione analogues as 15-Hydroxyprostaglandin dehydrogenase inhibitors. J. Med. Chem. 54: 5260–5264.
Choi, D. B., Y. L. Piao, Y. Wub, and H. Cho (2013) Control of the intracellular levels of prostaglandin E2 through inhibition of the 15-hydroxyprostaglandin dehydrogenase for wound healing. Bioorganic Med. Chem. 21: 4477–4484.
Piao, Y.L., S. A. Ram, and H. Cho (2015) Cell-based biological evaluations of 5-(3-bromo-4-phenethoxybenzylidene)thiazolidine- 2,4-dione as promising wound healing agent. Bioorganic Med. Chem. 23: 2098–103.
Clark, R. A (1996) The molecular and cellular biology of wound repair, 2nd ed.; New York, Plenum, pp. 188–206.
Martin, P. (1997) Wound healing—aiming for perfect skin regeneration. Science 276: 75–81.
Werner, S. and R. Grose (2003) Regulation of wound healing by growth factors and cytokines. Physiol. Rev. 83: 835–870.
Kolodsick, J. E., M. Peters-Golden, J. Larios, G. B. Toews, V. J. Thannickal, and B. B. Moore (2003) Prostaglandin E inhibits fibroblast to myofibroblast transition via E. prostanoid receptor 2 signaling and cyclic adenosine monophosphate elevation. American J. Resp. Cell Mole. Bio. 29: 537–544.
Acknowledgment
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2016R1D1A1B04930255).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Yu, I., Choi, D., Lee, HK. et al. Synthesis and Biological Evaluation of New Benzylidenethiazolidine-2,4-dione Derivatives as 15-hydroxyprostaglandin Dehydrogenase Inhibitors to Control the Intracellular Levels of Prostaglandin E2 for Wound Healing. Biotechnol Bioproc E 24, 464–475 (2019). https://doi.org/10.1007/s12257-019-0015-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-019-0015-8