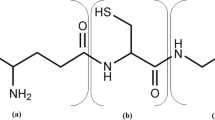
Abstract
The effect of different sugars (glucose, glycerol, maltose, galactose and lactose) on cell-membrane-associated proteinase production by Lactobacillus delbrueckii subsp. lactis 313 (LDL 313) was investigated. The experimental results showed that aside glycerol and galactose, all the other sugars supported high growth levels of LDL 313, with glucose displaying the maximum biomass concentration of 0.85 mg/mL dry cell weight for cells harvested at the mid-exponential phase of ∼12 h after inoculation. The specific proteinase yield, a measure of the rate of proteinase production relative to cell wall biosynthesis, was used to evaluate the preferential degree of proteinase metabolism as induced by the consumption of different sugar substrates by LDL 313. It was found that maltose displayed the highest specific proteinase yield of 12.59 U/mg sugar consumed. Further, molecular differences were observed in the SDS electrophoretic profile of cell surface proteins generated for the different carbon substrates. This is a preliminary study which supports the inference that different sugars stimulate the production of different cell-surface proteins with a significant effect on cell proteinase activity.
Similar content being viewed by others
References
Tsakalidou, E., R. Anastasiou, I. Vandenberghe, J. van Beeumen, and G. Kalantzopoulos (1999) Cell-Wall-Bound Proteinase of Lactobacillus delbrueckii subsp. lactis ACA-DC 178: Characterization and Specicity for β-Casein. Appl. Environ. Microbiol. 65: 2035–2040.
Espeche Turbay, M. A. B., G. Savoy de Giori, and E. M. Hebert (2009) Release of the Cell-envelope-associated proteinase of Lactobacillus delbrueckii subspecies lactis CRL 581 is dependent upon pH and temperature. J. Agric. Food Chem. 57: 8607–8611.
Axelsson, L. (2004) Lactic acid bacteria: Classification and physiology, in Lactic Acid Bacteria. pp. 1–66. In: S. Salminen, A. von Wright, and A. Ouwehand (ed.). Microbiological and Functional Aspects. Marcel Dekker, NY.
Kunji, E. R. S., I. Mierau, A. Hagting, B. Poolman, and W. N. Konings (1996) The proteotytic systems of lactic acid bacteria. Anton. Leeuw. 70: 187–221.
Khalid, N. M. and E. H. Marth (1990) Lactobacilli — Their enzymes and role in ripening and spoilage of cheese: A Review. J. Dairy Sci. 73: 2669–2684.
Gupta, R., Q. K. Beg, S. Khan, and B. Chauhan (2002) An overview on fermentation, downstream processing and properties of microbial alkaline proteases. Appl. Microbiol. Biotechnol. 60: 381–395.
Potumarthi, R., S. Ch, and A. Jetty (2007) Alkaline protease production by submerged fermentation in stirred tank reactor using Bacillus licheniformis NCIM-2042: Effect of aeration and agitation regimes. Biochem. Eng. J. 34: 185–192.
Titgemeyer, F. and W. Hillen (2002) Global control of sugar metabolism: A Gram-positive solution. Anton. Leeuw. 82: 59–71.
Schick, J., B. Weber, J. R. Klein, and B. Henrich (1999) PepR1, a CcpA-like transcription regulator of Lactobacillus delbrueckii subsp. lactis. Microb. 145: 3147–3154.
Kim, J. -H., S. P. Shoemaker, and D. A. Mills (2009) Relaxed control of sugar utilization in Lactobacillus brevis. Microb. 155: 1351–1359.
Morel, F., J. Frot-Coutaz, D. Aubel, R. Portalier, and D. Atlan (1999) Characterization of a prolidase from Lactobacillus delbrueckii subsp. bulgaricus CNRZ 397 with an unusual regulation of biosynthesis. Microb. 145: 437–446.
Mahr, K., W. Hillen, and F. Titgemeyer (2000) Carbon catabolite repression in Lactobacillus pentosus: Analysis of the ccpA region. Appl. Environ. Microbiol. 66: 277–283.
van den Bogaard, P. T. C. (2002) Catabolite Control of Sugar Metabolism in Streptococcus thermophilus, in Laboratory of Microbiology. p. 158. Wageningen University: Wageningen.
Chervaux, C., S. D. Ehrlich, and E. Maguin (2000) Physiological study of Lactobacillus delbrueckii subsp. bulgaricus strains in a novel chemically defined medium. Appl. Environ. Microbiol. 66: 5306–5311.
Schiraldi, C., V. Adduci, V. Valli, C. Maresca, M. Giuliano, M. Lamberti, M. Cartenì, and M. De Rosa (2003) High cell density cultivation of probiotics and lactic acid production. Biotechnol. Bioeng. 82: 213–222.
Kim, J. -H., D. E. Block, S. P. Shoemaker, and D. A. Mills (2010) Conversion of rice straw to bio-based chemicals: An integrated process using Lactobacillus brevis. Appl. Microbiol. Biotechnol. 86: 1375–1385.
Fournier, E. (2001) Colorimetric Quantification of Carbohydrates. Current Protocols in Food Analytical Chemistry. pp. E1.1.1–E1.1.8. John Wiley & Sons, Inc.
Exterkate, F. A. (1990) Differences in short peptide-substrate cleavage by two cell-envelope-located serine proteinases of Lactococcus lactis subsp. cremoris are related to secondary binding specificity. Appl. Microbiol. Biotechnol. 33: 401–406.
Gaudana, S. B., A. S. Dhanani, and T. Bagchi (2010) Probiotic attributes of Lactobacillus strains isolated from food and of human origin. Br. J. Nutr. 103: 1620–1628.
Bevilacqua, A., M. Corbo, M. Mastromatteo, and M. Sinigaglia (2008) Combined effects of pH, yeast extract, carbohydrates and di-ammonium hydrogen citrate on the biomass production and acidifying ability of a probiotic Lactobacillus plantarum strain, isolated from table olives, in a batch system. World J. Microb. Biotechnol. 24: 1721–1729.
Veenhoff, L. M., E. H. M. L. Heuberger, and B. Poolman (2001) The lactose transport protein is a cooperative dimer with two sugar translocation pathways. EMBO J. 20: 3056–3062.
van den Bogaard, P. T. C., M. Kleerebezem, O. P. Kuipers, and W. M. de Vos (2000) Control of lactose transport, beta -galactosidase activity, and glycolysis by CcpA in Streptococcus thermophilus: Evidence for carbon catabolite repression by a Nonphosphoenolpyruvate-dependent phosphotransferase system sugar. J. Bacteriol. 182: 5982–5989.
Frey, P. (1996) The Leloir pathway: A mechanistic imperative for three enzymes to change the stereochemical configuration of a single carbon in galactose. The FASEB J. 10: 461–470.
Bettenbrock, K. and C. -A. Alpert (1998) The gal Genes for the Leloir Pathway of Lactobacillus casei 64H. Appl. Environ. Microbiol. 64: 2013–2019.
Hickey, M. W., A. J. Hillier, and G. R. Jago (1986) Transport and metabolism of lactose, glucose, and galactose in homofermentative lactobacilli. Appl. Environ. Microbiol. 51: 825–831.
Hebert, E., G. Mamone, G. Picariello, R. Raya, G. Savoy, P. Ferranti, and F. Addeo (2008) Characterization of the pattern of ás1-and & a-casein breakdown and release of a bioactive peptide by a cell envelope proteinase from Lactobacillus delbrueckii subsp. lactis CRL 581. Appl. Environ. Microbiol. 74: 3682–3689.
Neubauer, H., E. Glaasker, W. P. Hammes, B. Poolman, and W. N. Konings (1994) Mechanism of maltose uptake and glucose excretion in Lactobacillus sanfrancisco. J. Bacteriol. 176: 3007–3012.
Schär-Zammaretti, P. and J. Ubbink (2003) The cell wall of lactic acid bacteria: Surface constituents and macromolecular conformations. Biophys. J. 85: 4076–4092.
Otto, A., J. Bernhardt, H. Meyer, M. Schaffer, F. -A. Herbst, J. Siebourg, U. Mäder, M. Lalk, M. Hecker, and D. Becher (2010) Systems-wide temporal proteomic profiling in glucose-starved Bacillus subtilis. Nat. Commun. 1: 137.
Schar-Zammaretti, P., M. -L. Dillmann, N. D’Amico, M. Affolter, and J. Ubbink (2005) Influence of fermentation medium composition on physicochemical surface properties of Lactobacillus acidophilus. Appl. Environ. Microbiol. 71: 8165–8173.
Tymczyszyn, E. E., A. Gómez-Zavaglia, and E. A. Disalvo (2007) Effect of sugars and growth media on the dehydration of Lactobacillus delbrueckii ssp. bulgaricus. J. Appl. Microbiol. 102: 845–851.
Petranovic, D., E. Guedon, B. Sperandio, C. Delorme, D. Ehrlich, and P. Renault (2004) Intracellular effectors regulating the activity of the Lactococcus lactis CodY pleiotropic transcription regulator. Mol. Microbiol. 53: 613–621.
Dineen, S. S., S. M. McBride, and A. L. Sonenshein (2010) Integration of metabolism and virulence by Clostridium difficile CodY. J. Bacteriol. 192: 5350–5362.
Guedon, E., P. Renault, S. D. Ehrlich, and C. Delorme (2001) Transcriptional pattern of genes coding for the proteolytic system of Lactococcus lactis and evidence for coordinated regulation of key enzymes by peptide supply. J. Bacteriol. 183: 3614–3622.
Sánchez, B., P. Bressollier, S. Chaignepain, J. -M. Schmitter, and M. C. Urdaci (2009) Identification of surface-associated proteins in the probiotic bacterium Lactobacillus rhamnosus GG. Int. Dairy J. 19: 85–88.
Deepika, G., R. Green, R. Frazier, and D. Charalampopoulos (2009) Effect of growth time on the surface and adhesion properties of Lactobacillus rhamnosus GG. J. Appl. Microbiol. 107: 1230–1240.
Lortal, S., J. Van Heijenoort, K. Gruber, and U. B. Sleytr (1992) S-layer of Lactobacillus helveticus ATCC 12046: Isolation, chemical characterization and re-formation after extraction with lithium chloride. J. Gen. Microbiol. 138: 611–618.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Agyei, D., Danquah, M.K. Carbohydrate utilization affects Lactobacillus delbrueckii subsp. lactis 313 cell-enveloped-associated proteinase production. Biotechnol Bioproc E 17, 787–794 (2012). https://doi.org/10.1007/s12257-012-0106-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12257-012-0106-2