Abstract
Introduction
Breast tumors often display an astonishing degree of spatial and temporal heterogeneity, which are associated with cancer progression, drug resistance, and relapse. Triple-negative breast cancer (TNBC) is a particularly aggressive and heterogeneous subtype for which targeted therapies are scarce. Consequently, patients with TNBC have a poorer overall prognosis compared to other breast cancer patients. Within heterogeneous tumors, individual clonal subpopulations may exhibit differences in their rates of growth and degrees of invasiveness. We hypothesized that such phenotypic heterogeneity at the single-cell level may accelerate tumor progression by enhancing the overall growth and invasion of the entire tumor.
Methods
To test this hypothesis, we isolated and characterized clonal subpopulations with distinct morphologies and biomarker expression from the inherently heterogeneous 4T1 mouse mammary carcinoma cell line. We then leveraged a 3D microfluidic tumor model to reverse-engineer intratumoral heterogeneity and thus investigate how interactions between phenotypically distinct subpopulations affect tumor growth and invasion.
Results
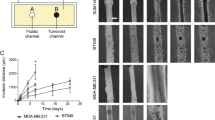
We found that the growth and invasion of multiclonal tumors were largely dictated by the presence of cells with epithelial and mesenchymal traits, respectively. The latter accelerated overall tumor invasion, even when these cells comprised less than 1% of the initial population. Consistently, tumor progression was delayed by selectively targeting the mesenchymal subpopulation.
Discussion
This work reveals that highly invasive cells can dominate tumor phenotype and that specifically targeting these cells can slow the progression of heterogeneous tumors, which may help inform therapeutic approaches.








Similar content being viewed by others
Data availability
RNA-seq data have been deposited at GEO under accession number GSE252177 and are available as of the date of publication.
Abbreviations
- CAM:
-
Chorioallantoic membrane
- EMT:
-
Epithelial–mesenchymal transition
- TNBC:
-
Triple-negative breast cancer
References
Kim, C., R. Gao, E. Sei, R. Brandt, J. Hartman, T. Hatschek, et al. Chemoresistance evolution in triple-negative breast cancer delineated by single-cell sequencing. Cell. 173(4):879–893, 2018. https://doi.org/10.1016/j.cell.2018.03.041.
Torab, P., Y. Yan, M. Ahmed, H. Yamashita, J. I. Warrick, J. D. Raman, et al. Intratumoral heterogeneity promotes collective cancer invasion through NOTCH1 variation. Cells. 2021. https://doi.org/10.3390/cells10113084.
Marusyk, A., and K. Polyak. Tumor heterogeneity: causes and consequences. Biochim. Biophys. Acta. 1805(1):105–117, 2010. https://doi.org/10.1016/j.bbcan.2009.11.002.
Inda, M. M., R. Bonavia, A. Mukasa, Y. Narita, D. Sah, S. Vandenberg, et al. Tumor heterogeneity is an active process maintained by a mutant EGFR-induced cytokine circuit in glioblastoma. Genes Dev. 2010. https://doi.org/10.1101/gad.1890510.
Summerbell, E. R., J. K. Mouw, J. S. K. Bell, C. M. Knippler, B. Pedro, J. L. Arnst, et al. Epigenetically heterogeneous tumor cells direct collective invasion through filopodia-driven fibronectin micropatterning. Sci. Adv. 2020. https://doi.org/10.1126/sciadv.aaz6197.
Wu, P. H., D. M. Gilkes, J. M. Phillip, A. Narkar, T. W. Cheng, J. Marchand, et al. Single-cell morphology encodes metastatic potential. Sci. Adv. 2020. https://doi.org/10.1126/sciadv.aaw6938.
Fedele, C., R. W. Tothill, and G. A. McArthur. Navigating the challenge of tumor heterogeneity in cancer therapy. Cancer Discov. 4(2):146–148, 2014. https://doi.org/10.1158/2159-8290.CD-13-1042.
Kalluri, R., and R. Weinberg. The basics of epithelial–mesenchymal transition. J. Clin. Investig. 119(6):1420–1428, 2009. https://doi.org/10.1172/JCI39104.
Yang, J., P. Antin, G. Berx, C. Blanpain, T. Brabletz, M. Bronner, et al. Guidelines and definitions for research on epithelial–mesenchymal transition. Nat. Rev. Mol. Cell Biol. 21(6):341–352, 2020. https://doi.org/10.1038/s41580-020-0237-9.
Liu, X., J. Li, B. L. Cadilha, A. Markota, C. Voigt, Z. Huang, et al. Epithelial-type systemic breast carcinoma cells with a restricted mesenchymal transition are a major source of metastasis. Sci. Adv. 2019. https://doi.org/10.1126/sciadv.aav4275.
Carey, S. P., A. Starchenko, A. L. McGregor, and C. A. Reinhart-King. Leading malignant cells initiate collective epithelial cell invasion in a three-dimensional heterotypic tumor spheroid model. Clin. Exp. Metastasis. 2013. https://doi.org/10.1007/s10585-013-9565-x.
Hapach, L. A., S. P. Carey, S. C. Schwager, P. V. Taufalele, W. W. Wang, J. A. Mosier, et al. Phenotypic heterogeneity and metastasis of breast cancer cells. Cancer Res. 2021. https://doi.org/10.1158/0008-5472.CAN-20-1799.
Young, K. M., and C. A. Reinhart-King. Phenotypic heterogeneity and cooperation in the metastatic cascade. Oncoscience. 9:55–56, 2022. https://doi.org/10.18632/oncoscience.565.
Hapach, L. A., W. Wang, S. C. Schwager, D. Pokhriyal, E. D. Fabiano, and C. A. Reinhart-King. Phenotypically sorted highly and weakly migratory triple negative breast cancer cells exhibit migratory and metastatic commensalism. Breast Cancer Res. 25(1):102, 2023. https://doi.org/10.1186/s13058-023-01696-3.
Martín-Pardillos, A., Á. Valls Chiva, G. Bande Vargas, P. Hurtado Blanco, R. Piñeiro Cid, P. J. Guijarro, et al. The role of clonal communication and heterogeneity in breast cancer. BMC Cancer 19(1):1–26, 2019. https://doi.org/10.1186/s12885-019-5883-y.
Tao, K., M. Fang, J. Alroy, and G. G. Sahagian. Imagable 4T1 model for the study of late stage breast cancer. BMC Cancer. 8:228, 2008. https://doi.org/10.1186/1471-2407-8-228.
Heppner, G. H., F. R. Miller, and P. M. Shekhar. Nontransgenic models of breast cancer. Breast Cancer Res. 2(5):331–334, 2000. https://doi.org/10.1186/bcr77.
Ilina, O., L. Campanello, P. G. Gritsenko, M. Vullings, C. Wang, P. Bult, et al. Intravital microscopy of collective invasion plasticity in breast cancer. Dis. Model Mech. 2018. https://doi.org/10.1242/dmm.034330.
Wagenblast, E., M. Soto, S. Gutiérrez-Ángel, C. A. Hartl, A. L. Gable, A. R. Maceli, et al. A model of breast cancer heterogeneity reveals vascular mimicry as a driver of metastasis. Nature. 520(7547):358–362, 2015. https://doi.org/10.1038/nature14403.
Wang, R., C. Jin, and X. Hu. Evidence of drug-response heterogeneity rapidly generated from a single cancer cell. Oncotarget. 8(25):41113–41124, 2017. https://doi.org/10.18632/oncotarget.17064.
Yeo, S. K., X. Zhu, T. Okamoto, M. Hao, C. Wang, P. Lu, et al. Single-cell RNA-sequencing reveals distinct patterns of cell state heterogeneity in mouse models of breast cancer. Elife. 2020. https://doi.org/10.7554/eLife.58810.
Schrors, B., S. Boegel, C. Albrecht, T. Bukur, V. Bukur, C. Holtstrater, et al. Multi-omics characterization of the 4T1 murine mammary gland tumor model. Front. Oncol. 10:1195, 2020. https://doi.org/10.3389/fonc.2020.01195.
Heppner, G. H., D. L. Dexter, T. DeNucci, F. R. Miller, and P. Calabresi. Heterogeneity in drug sensitivity among tumor cell subpopulations of a single mammary tumor. Cancer Res. 38(11 Pt 1):3758–3763, 1978.
Aslakson, C. J., and F. R. Miller. Selective events in the metastatic process defined by analysis of the sequential dissemination of subpopulations of a mouse mammary tumor. Cancer Res. 52(6):1399–1405, 1992.
Bhatia, S., J. Monkman, T. Blick, P. H. Duijf, S. H. Nagaraj, and E. W. Thompson. Multi-omics characterization of the spontaneous mesenchymal–epithelial transition in the PMC42 breast cancer cell lines. J. Clin. Med. 8(8):1253, 2019. https://doi.org/10.3390/jcm8081253.
Shcherbakova, D. M., and V. V. Verkhusha. Near-infrared fluorescent proteins for multicolor in vivo imaging. Nat. Methods. 10(8):751–754, 2013. https://doi.org/10.1038/nmeth.2521.
Oliva-Trastoy, M., M. Defais, and F. Larminat. Resistance to the antibiotic Zeocin by stable expression of the Sh ble gene does not fully suppress Zeocin-induced DNA cleavage in human cells. Mutagen. 20(2):111–114, 2005. https://doi.org/10.1093/mutage/gei016.
Piotrowski-Daspit, A. S., and C. M. Nelson. Engineering three-dimensional epithelial tissues embedded within extracellular matrix. J. Vis. Exp. 2016. https://doi.org/10.3791/54283.
Piotrowski-Daspit, A. S., J. Tien, and C. M. Nelson. Interstitial fluid pressure regulates collective invasion in engineered human breast tumors via Snail, vimentin, and E-cadherin. Integr. Biol. 8(3):319–331, 2016. https://doi.org/10.1039/c5ib00282f.
Piotrowski-Daspit, A. S., A. K. Simi, M. Pang, J. Tien, and C. M. Nelson. A 3D culture model to study how fluid pressure and flow affect the behavior of aggregates of epithelial cells. In: Mammary Gland Development: Methods and Protocols (Methods in Molecular Biology Series), edited by F. Martin, T. Stein, and J. Howlin. New York: Springer, 2017, pp. 245–257.
Teng, Y. Hanging drop aggregation assay of breast cancer cells. Bio-Protocols. 2015. https://doi.org/10.21769/BioProtoc.1393.
Foty, R. A simple hanging drop cell culture protocol for generation of 3D spheroids. J. Vis. Exp. 2011. https://doi.org/10.3791/2720.
Spurlin III, J., and P. Lwigale. A technique to increase accessibility to late-stage chick embryos for in ovo manipulations. Dev. Dyn. 2013. https://doi.org/10.1002/dvdy.23907.
Aleskandarany, M. A., M. E. Vandenberghe, C. Marchiò, I. O. Ellis, A. Sapino, and E. A. Rakha. Tumour heterogeneity of breast cancer: from morphology to personalised medicine. Pathobiology. 85(1–2):23–34, 2018. https://doi.org/10.1159/000477851.
Aiello, N. M., R. Maddipati, R. J. Norgard, D. Balli, J. Li, S. Yuan, et al. EMT subtype influences epithelial plasticity and mode of cell migration. Dev. Cell. 2018. https://doi.org/10.1016/j.devcel.2018.05.027.
Mendez, M. G., S. Kojima, and R. D. Goldman. Vimentin induces changes in cell shape, motility, and adhesion during the epithelial to mesenchymal transition. FASEB J. 2010. https://doi.org/10.1096/fj.09-151639.
Prasad, A., and E. Alizadeh. Cell form and function: interpreting and controlling the shape of adherent cells. Trends Biotechnol. 2019. https://doi.org/10.1016/j.tibtech.2018.09.007.
Mueller-Klieser, W. Multicellular spheroids. A review on cellular aggregates in cancer research. J. Cancer Res. Clin. Oncol. 1987. https://doi.org/10.1007/BF00391431.
Graziani, V., I. Rodriguez-Hernandez, O. Maiques, and V. Sanz-Moreno. The amoeboid state as part of the epithelial-to-mesenchymal transition programme. Trends Cell Biol. 32(3):228–242, 2022. https://doi.org/10.1016/j.tcb.2021.10.004.
Tien, J., J. G. Truslow, and C. M. Nelson. Modulation of invasive phenotype by interstitial pressure-driven convection in aggregates of human breast cancer cells. PLoS ONE. 2012. https://doi.org/10.1371/journal.pone.0045191.
Tien, J., U. Ghani, Y. W. Dance, A. J. Seibel, M. C. Karakan, K. L. Ekinci, et al. Matrix pore size governs escape of human breast cancer cells from a microtumor to an empty cavity. Iscience. 2020. https://doi.org/10.1016/j.isci.2020.101673.
Liu, Y., Y. Fang, L. Bao, F. Wu, S. Wang, and S. Hao. Intercellular communication reveals therapeutic potential of epithelial–mesenchymal transition in triple-negative breast cancer. Biomolecules. 2022. https://doi.org/10.3390/biom12101478.
Konen, J., E. Summerbell, B. Dwivedi, K. Galior, Y. Hou, L. Rusnak, et al. Image-guided genomics of phenotypically heterogeneous populations reveals vascular signalling during symbiotic collective cancer invasion. Nat. Commun. 8:15078, 2017. https://doi.org/10.1038/ncomms15078.
Saenz-de-Santa-Maria, I., L. Celada, and M. D. Chiara. The leader position of mesenchymal cells expressing N-cadherin in the collective migration of epithelial cancer. Cells. 2020. https://doi.org/10.3390/cells9030731.
Hallou, A., J. Jennings, and A. J. Kabla. Tumour heterogeneity promotes collective invasion and cancer metastatic dissemination. R. Soc. Open Sci.4(8):161007, 2017. https://doi.org/10.1098/rsos.161007.
Zhao, B., J. R. Pritchard, D. A. Lauffenburger, and M. T. Hemann. Addressing genetic tumor heterogeneity through computationally predictive combination therapy. Cancer Discov. 4(2):166–174, 2014. https://doi.org/10.1158/2159-8290.CD-13-0465.
Zhao, B., M. T. Hemann, and D. A. Lauffenburger. Intratumor heterogeneity alters most effective drugs in designed combinations. Proc. Natl Acad. Sci. USA. 111(29):10773–10778, 2014. https://doi.org/10.1073/pnas.1323934111.
McQuerry, J. A., J. T. Chang, D. D. L. Bowtell, A. Cohen, and A. H. Bild. Mechanisms and clinical implications of tumor heterogeneity and convergence on recurrent phenotypes. J. Mol. Med. 2017. https://doi.org/10.1007/s00109-017-1587-4.
Kleppe, M., and R. L. Levine. Tumor heterogeneity confounds and illuminates: assessing the implications. Nat. Med. 2014. https://doi.org/10.1038/nm.3522.
Tammela, T., and J. Sage. Investigating tumor heterogeneity in mouse models. Annu. Rev. Cancer Biol. 2020. https://doi.org/10.1146/annurev-cancerbio-030419-033413.
Denison, T. A., and Y. H. Bae. Tumor heterogeneity and its implication for drug delivery. J. Control Release. 164(2):187–191, 2012. https://doi.org/10.1016/j.jconrel.2012.04.014.
Marusyk, A., M. Janiszewska, and K. Polyak. Intratumor heterogeneity: The Rosetta Stone of therapy resistance. Cancer Cell. 37(4):471–484, 2020. https://doi.org/10.1016/j.ccell.2020.03.007.
Hayford, C. E., D. R. Tyson, C. J. Robbins III., P. L. Frick, V. Quaranta, and L. A. Harris. An in vitro model of tumor heterogeneity resolves genetic, epigenetic, and stochastic sources of cell state variability. PLoS Biol.19(6):e3000797, 2021. https://doi.org/10.1371/journal.pbio.3000797.
Zellmer, V. R., and S. Zhang. Evolving concepts of tumor heterogeneity. Cell Biosci. 4(1):1–8, 2014. https://doi.org/10.1186/2045-3701-4-69.
Acknowledgements
This work was supported in part by grants from the National Institutes of Health (CA187692, CA214292), the New Jersey Health Foundation, and a Faculty Scholars Award from the Howard Hughes Medical Institute. SEL was supported in part by the National Center for Advancing Translational Sciences of the National Institutes of Health (TL1TR003019, UL1TR003017). MCB was supported in part by the Ludwig Princeton Branch and by a New Jersey Commission on Cancer Research Predoctoral Fellowship (COCR22PRF009).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Associate Editor Michael R. King oversaw the review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Leggett, S.E., Brennan, M.C., Martinez, S. et al. Relatively Rare Populations of Invasive Cells Drive Progression of Heterogeneous Tumors. Cel. Mol. Bioeng. 17, 7–24 (2024). https://doi.org/10.1007/s12195-023-00792-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12195-023-00792-w